1. Introduction
2. Experimental
3. Results and discussions
3.1. Selection of extractant system
3.2. Effect of commercial based extractants
3.3. Third phase studies
3.4. Water washings of the loaded organic
3.5. Scrubbing studies
3.6. Stripping studies
4. Conclusions
1. Introduction
Selective catalytic reduction (SCR) catalysts were used to control the NOX reagents in air. These NOX reagents are the cause for air pollution. Mainly NOX regents will generate from coal power plants. Due to strict environmental regulations implementation in Korea, all of the coal power plants are utilizing the SCR catalyst to prevent the NOX pollution1-3).
In new millennium, universal demand for SCR catalyst has been increased progressively due to strengthening environmental regulatory for NOx emission from stationary and mobile applications. In an effort to develop a better SCR catalyst, it is considered that V2O5-WO3/TiO2 catalyst is the most effective and widely employed to reduce NOx emissions.
Many methods were applied and developed for spent catalyst processing to recover the valuable metals, successfully in reported literature4-12).
V2O5-WO3/TiO2 catalyst having properties such as its high NOx conversion efficiency, durability from poisons and thermal, sulfur resistance, and wide operation temperature window has been considered to be the most commercial and excellent NOx conversion efficiency.
Our ongoing research on spent SCR catalyst processing, we have been developed the soda-roasting followed by water leaching, alkali leaching, pressure leaching were reported, recently13-16).
The present study feed stock was collected from Samcheonpo thermoelectric power plant, Korea and the spent SCR catalyst structure was honeycomb. This is V2O5-WO3/TiO2 type catalyst. After pretreatment roasting, spent SCR catalyst was dissolved by alkali. The generated leach liquor was followed by solvent extraction processing for vanadium and tungsten possible separation and metal loading enrichment.
2. Experimental
iCAP 6000 Series, Thermo Scientific, USA ICP-OES are used for metal ion analysis. Aqueous feed solutions were generated from alkali fusion of spent SCR catalyst followed by water leaching process.
In the alkali fusion, Na2CO3 (99.8%, Kanto Chemicals Co., Inc., Japan) was used as an alkali fusion agent for the soda-melting reactions. Before it used, further dehydration of Na2CO3 was not conducted. Mixing ratio of the spent SCR catalyst and Na2CO3 was 1 : 1.2 by weight (MOx/MOx+Na2O = 0.5, where M = Ti, Si, W, and V). Temperature of soda-melting reactions was set up to 1000°C. Dense alumina scrucible was use for alkali fusion reactions. Alkali fused melt dropped from the crucible was cooled and then pulverized. Before the water leaching of alkali fusion product, solidified alkali fusion melts (bulk) was grinded using a pin mill. Particle size distribution of pulverized soda melts was mainly ranging from 1 to 200 μm.
Water leaching of alkali fusion product was conducted under following conditions: temperature, 80°C; S/L (Weight of solid / Volume of liquid) ratio, 0.2; leaching time, 1 h; stirring speed, 350 rpm. During the water leaching, vaporized water was collected by water-cooled condenser. Water leaching slurry was filtrated and separated into solution and residue by vacuum filtration. In the further process, filtrated water leaching solution was used as feed solution in solvent extraction process.
The physical appearance of the spent SCR catalyst was presented in Fig. 1. It majorly contains 70.9 wt% of TiO2, 7.73 wt% of WO3 and 1.23 wt% of V2O5 etc. After pulverization of the spent SCR catalyst, EDS-SEM analysis was carryout and pictures were presented in Fig. 2.
The present study leach liquor obtained from spent SCR catalyst and initially it is having 1238 mg/L of vanadium and 8635 mg/L of tungsten at pH of the liquor was 14. Further studies diluted with mineral acids such as HCl and H2SO4 to reach lower pH conditions. Each experiment analyzed the fresh feed solution and applied for data analysis.
Aliquat 336 received from Cognis, Ireland was used as such. D2EHPA and TBP received from Aldrich and PC 88A received from Daihachi Chemicals Japan were used as such. All other chemicals were Analar grade. Solvent extraction experiments were carried by contacting aqueous and organic phases in separating funnels and shake by mechanical incubator at room temperature. After experiment, metals in aqueous solution were analyzed to determine distribution coefficient (DE) and percentage extraction (% E) values.
| $$Extraction,D_E=\frac{\lbrack M\rbrack in\;itorganic\;phase}{\lbrack M\rbrack in\;aqueous\;phase}$$ | (1) |
| $$\%Extraction=D_E\times100/(D_E+1)$$ | (2) |
| $$Stripping\;\mathrm{or}\;scrubbing,\;D_s=\frac{\lbrack M\rbrack in\;aqueous\;phase}{\lbrack M\rbrack in\;organic\;phase}$$ | (3) |
| $$\%Stripping\;\mathrm{or}\;\%scrubbing=D_s\times100/(D_s+1)$$ | (4) |
3. Results and discussions
3.1. Selection of extractant system
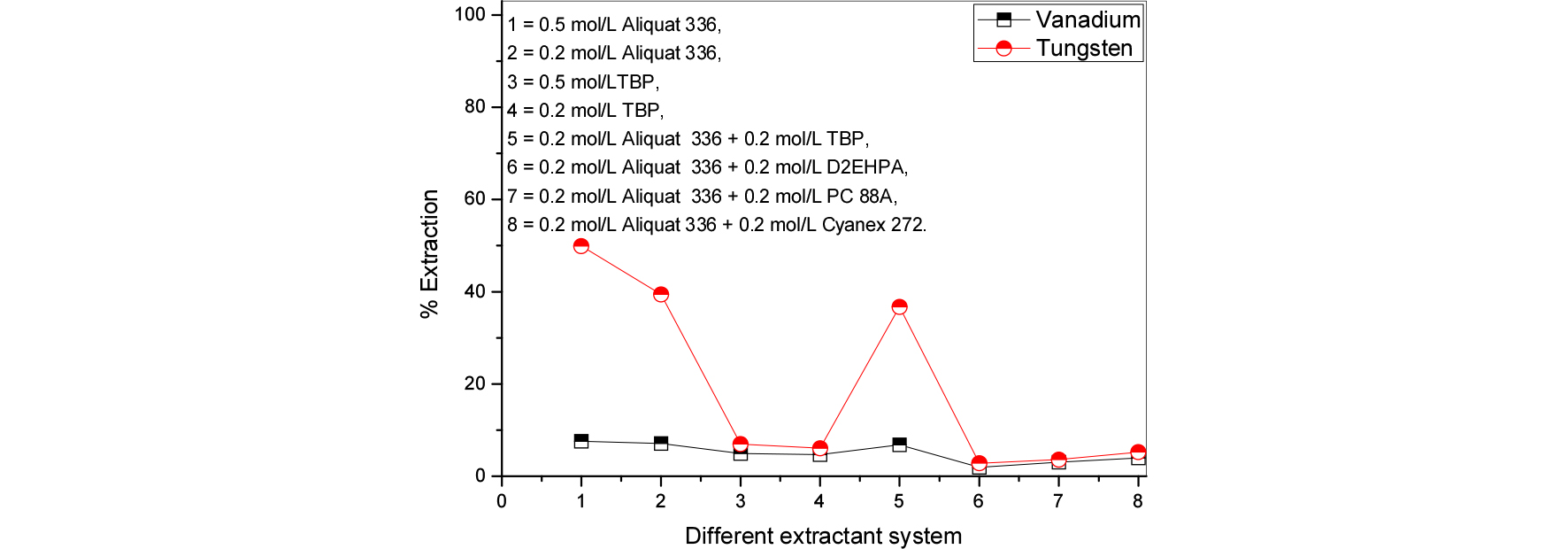
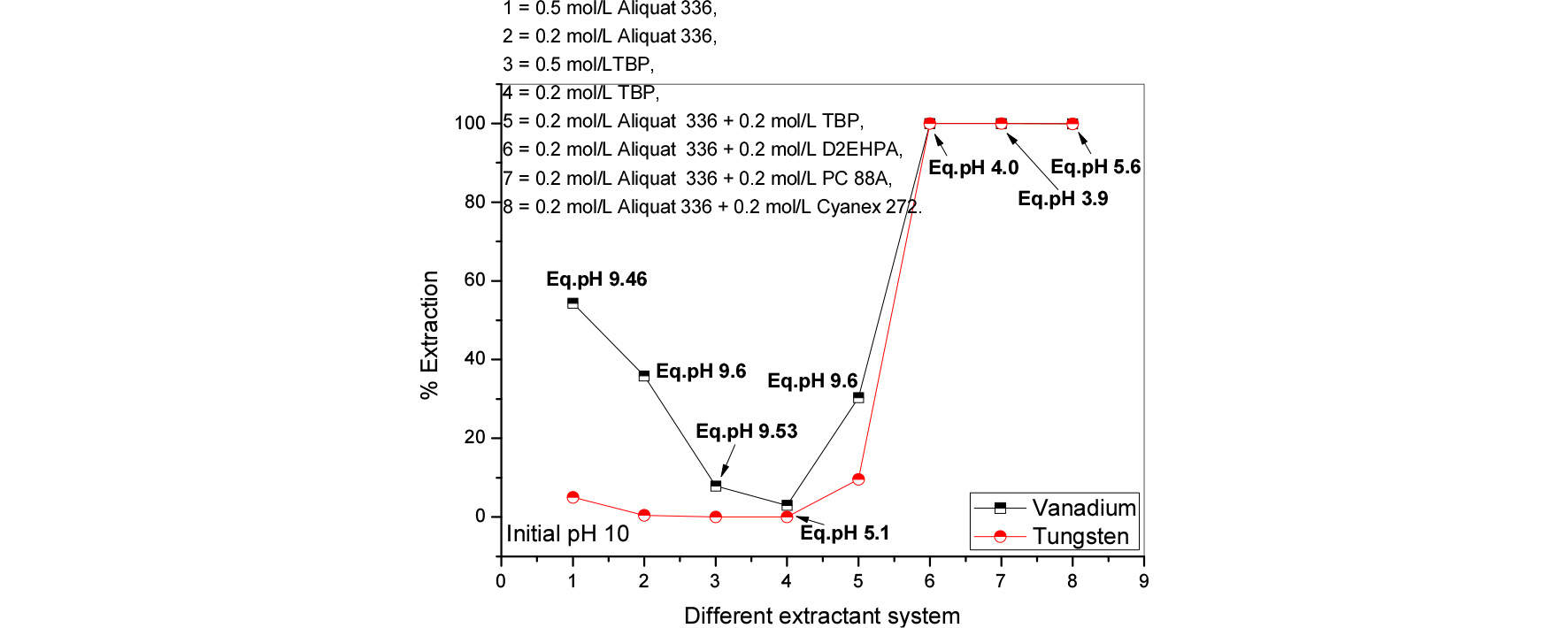
Various phosphorous and nitrogen based extractants were utilized for better extraction and possible separation of the tungsten and vanadium from spent SCR leach liquors. Three initial pH conditions were tested and obtained data were presented in Figs. 3 to 5. The results make known that; higher pH condition is not good for extraction of both metals (less than 10% extracted). The other pH conditions 7.6 to 10 are good for to achieve quantitative extraction of both the metals. And this study further indicated that, nitrogen based extractants were better than phosphorous based extractants. Further experiments the aqueous pH solutions were good have initial >10. And nitrogen based extractants were chosen for further studies.
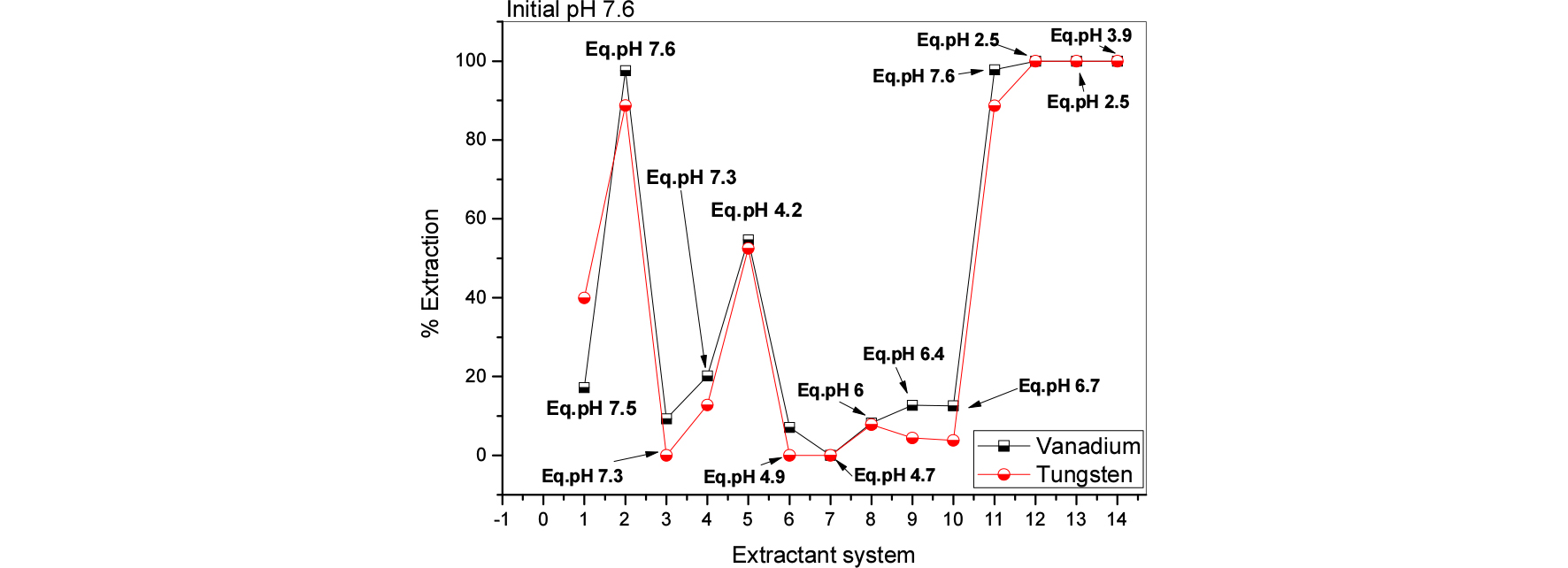
Fig. 5.
Effect of different extractant systems on vanadium and tungsten extraction from spent SCR catalyst leach solutions at initial pH 7.6 (1 = 0.5 mol/L Aliquat 336, 2 = 0.2 mol/L Aliquat 336, 3 = 0.5 mol/L TBP, 4 = 0.2 mol/L TBP, 5 = 0.5 mol/L D2EHPA, 6 = 0.2 mol/L D2EHPA, 7 = 0.5 mol/L PC 88A, 8 = 0.2 mol/L PC 88A, 9 = 0.5 mol/L Cyanex 272, 10 = 0.2 mol/L Cyanex 272, 11 = 0.2 mol/L Aliquat 336 + 0.2 mol/L TBP, 12 = 0.2 mol/L Aliquat 336 + 0.2 mol/L D2EHPA, 13 = 0.2 mol/L Aliquat 336 + 0.2 mol/L PC 88A, 14 = 0.2 mol/L Aliquat 336 + 0.2 mol/L Cyanex 272).
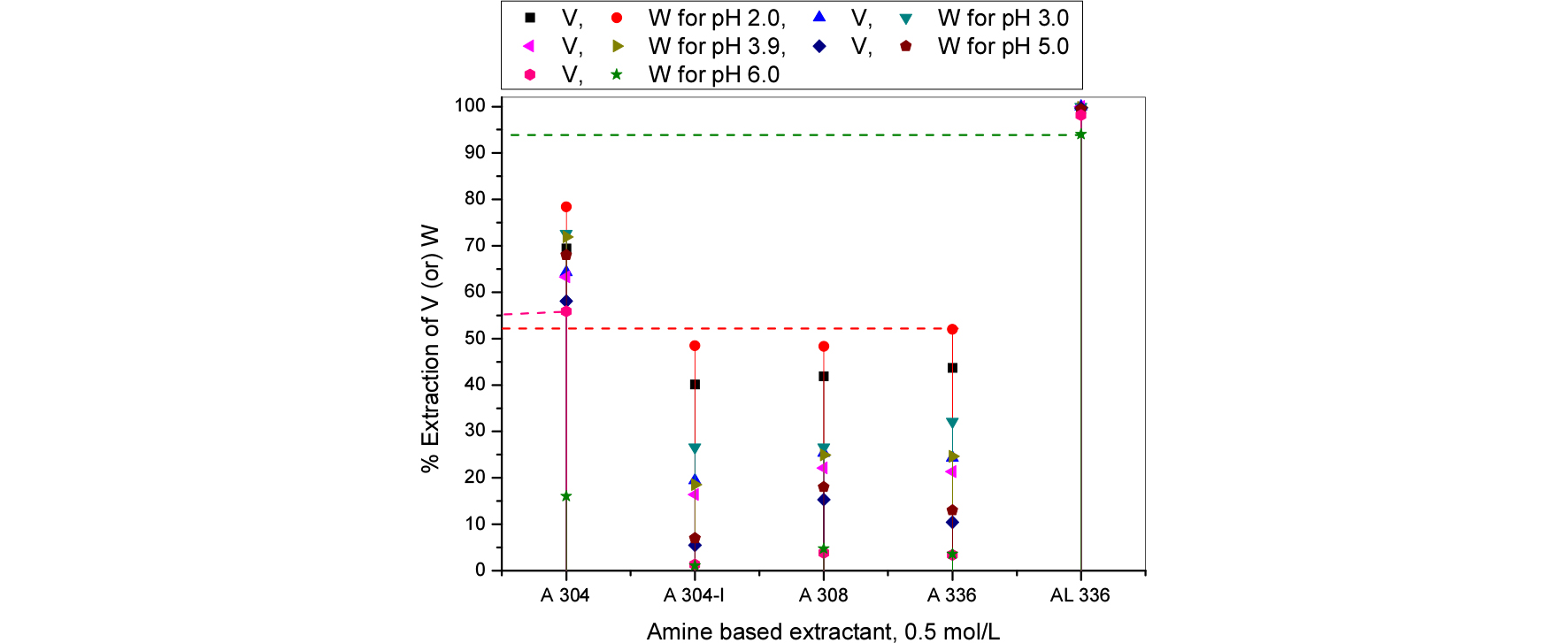
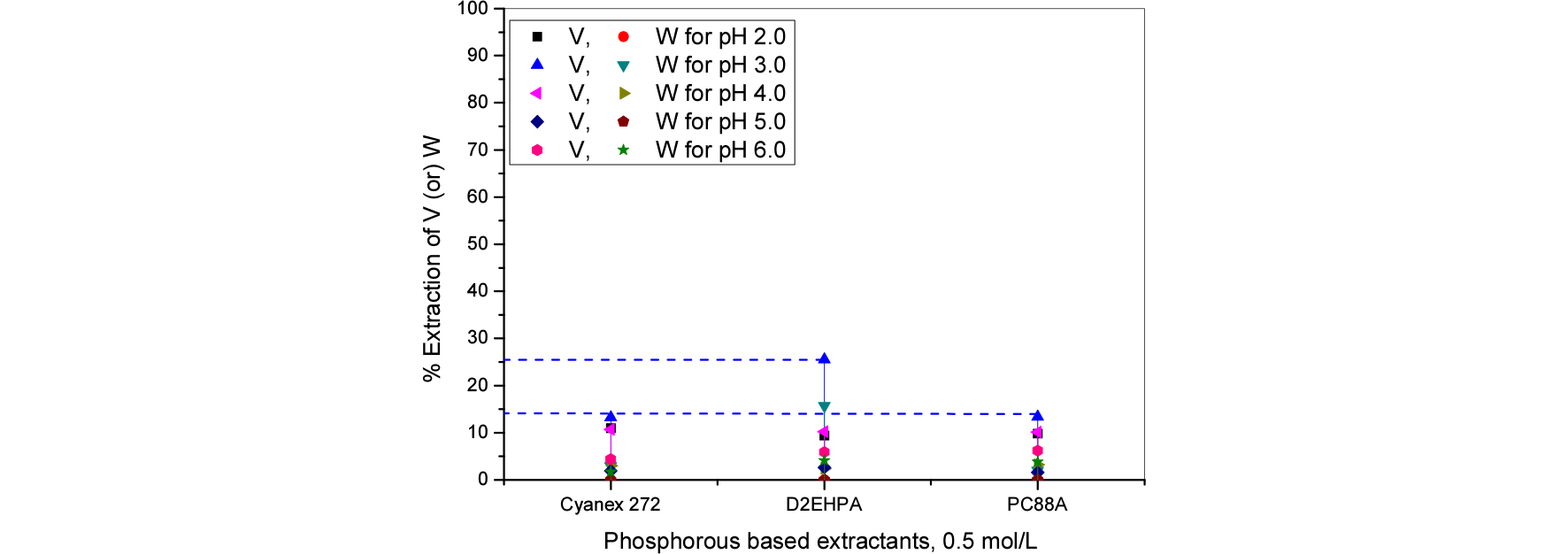
3.2. Effect of commercial based extractants
Based on previous experiments nitrogen based extractants were used for further study. For comparision purpose phosphorous based extractants are also studied. Extractants influence with pH varied from 2 to 6 for vanadium tungsten extraction from spent SCR catalyst. The gained experimental results were presented in Figs. 6 and 7. Amines are proved that, better than phosphorous based extractants for vanadium and tungsten extraction and possible separation process. The results reveals that, the extraction efficiencies were as follows the order: Aliquat 336 > Alamine 304 > Alamine 336 > Alamine 308 > Alamine 304-I.
3.3. Third phase studies
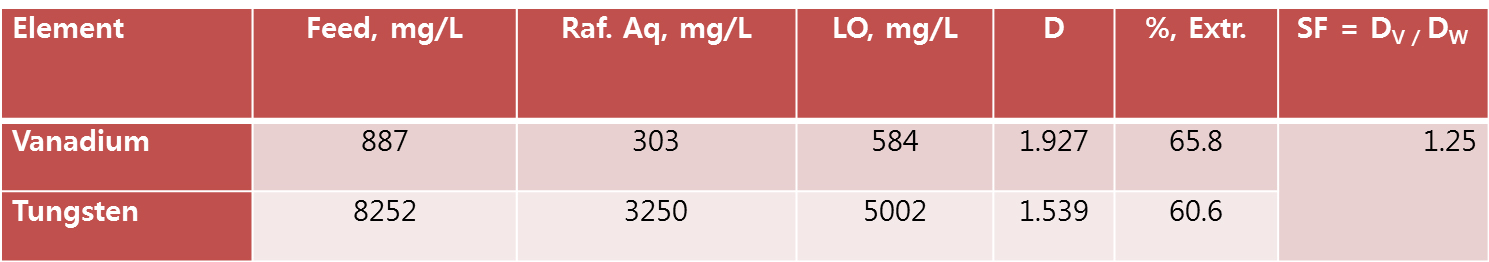
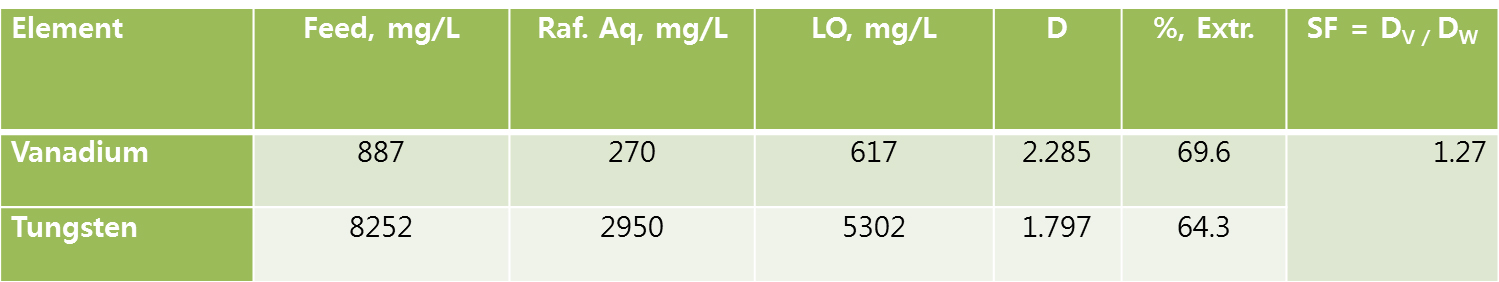
Among the all amine based extractants Aliquat 336 proved the better extraction capabilities for vanadium and tungsten. But it forms third phase, to remove third phase, this study needs to apply modifier. Two different type of the phase modifiers such as tri-butyl-phosphate (TBP) and iso-decanol (IDA) were used (Tables 1 and 2). Both the phase modifiers were suitable for to remove the third phase but the extraction efficiency was reduced ~30 to 35%.
3.4. Water washings of the loaded organic
After extraction experiment, the loaded organic (LO) phase was washed with distilled water. To possible to remove the un-wanted metals from LO and the gained data was presented in Tables 3 and 4. The extraction LOs were used for 3rd phase studies (LO data from Tables 1 and 2) and both the LOs after water washings loss nominal loss of tungsten and vanadium.
3.5. Scrubbing studies
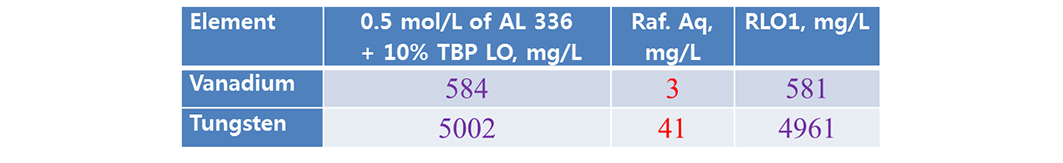
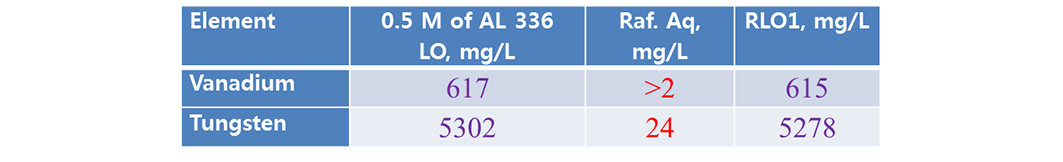
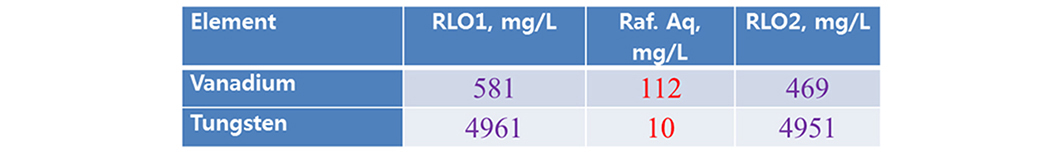
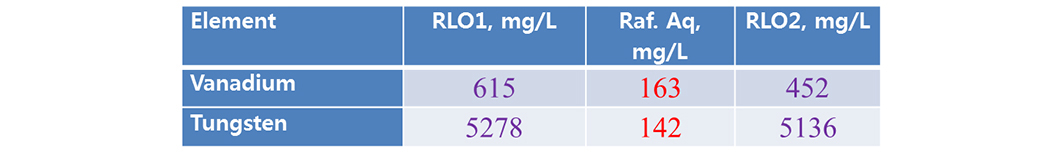
Scrubbing studies are made for possible separation of co-extracted metal. Present study acidified water (pH 1.7, acidified low diluted hydrochloric acid). The rsults are shown that (Tables 5 and 6), LO of 0.5 mol/L Aliquat + 10% TBP performed better removal of vanadium than LO of 0.5 mol/L Aliquat + 10% IDA (here both the metals are scrubbed).
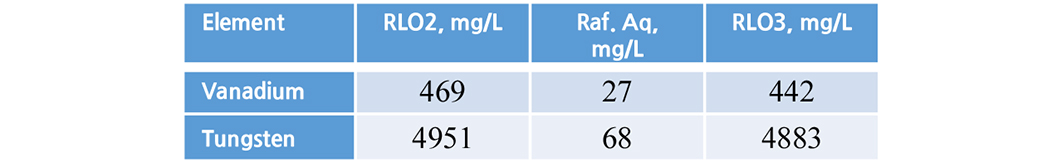
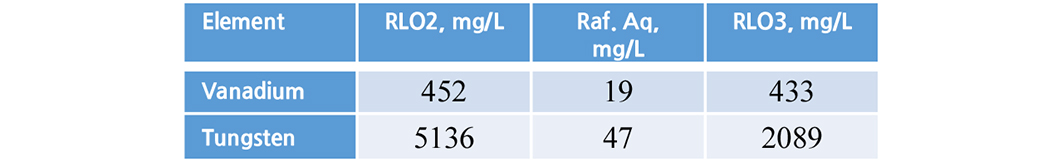
3.6. Stripping studies
Finally, the loaded metals were recovered with same acidified water pH 1.7 used for vanadium and tungsten. Both the metals more than 90% were recovered (Tables 7 and 8).
4. Conclusions
The present study concludes that, both the title metals such as vanadium and tungsten were positively extracted 60 to 65% from spent SCR catalyst leach liquors by using Aliquat 336 as an extractant system and kerosene use as a diluent. Water washings, scrubbing and stripping studies were developed and effectively up to 90% of vanadium and tungsten metals were recovered. This study reveals that, need to develop future work on metal enrichment as well as possible separation each other of vanadium and tungsten from spent SCR catalyst leach liquors.