1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Solvent extraction
2.3. Stripping the solvent
2.4. Synthesis of lithium Carbonate powder
3. Result and Discussion
3.1. Exaction of Lithium ion of various solvents at different pH
3.2. Exaction of Lithium ion from different Solvents
3.3. Exaction of Lithium ion at different O/A ratio using the mixture of Solvent TBP + D2EHPA
3.4. Exaction of Lithium ion at different extraction steps
3.5. Time for phase separation during Lithium ion extraction using the solvent mixture of 20% by volume of TBP + 20% by volume of D2EHPA at pH 4.21 but at different O/A ratio
3.6. Stripping of Lithium Solution from the Solvent
3.7. Making high purity of lithium carbonate
4. Conclusion
1. Introduction
In the early stages lithium was applied in many industrial applications in ceramic industry as fluxing agent and in the metallurgical industry as dichlorination agent1). Recently Lithium is highly used in making Lithium-ion batteries (LiBs) which are extensively used in the vehicle because of its good tolerance of wide range of temperature and high energy density2,3).
US Geological survey revealed that the lithium market about 39% is hold by the lithium battery industry, 30% by ceramics and glass industry and 8% by the lubrication industry4,5,6,7,8). Even LIBs has many advantages, it can run only for 2 or three years for consumer electronics and about 10 years for EV batteries which cause to produce large amount of waste battery system9). In this regards, the high demand of lithium ion battery is only fulfilled if the LIBs production is set to increase up to 12.7 million tons by 203010,11). The production of lithium ion from brine solution12,13,14) and minerals15) is insufficient to meet the globally demand of lithium ion in the future. This is only possible if lithium ion is recycled effectively from LIBs.
The recycling of LIBs has been done by mainly two steps: pyrometallury and hydrometallurgy. The pyrometallurgical process requires high energy because LIBs scrap is subjected to heat treatment and smelting at high temperature. Also, this process produces hazardous gasses and make environment polluted. Main problem of this method to date is unable to recover all lithium as light metal Li and aluminum are lost in slag. On the other hand, hydrometallurgical process require less energy and make environment less polluted than pyro metallurgical16).
Lithium, cobalt (Co), nickel (Ni), and manganese (Mn) are the valuable materials in the LIBs that are required to extract. In hydrometallurgical process, LIBs are generally first leached with mineral acids and NCM (Ni, Co and Mn) are recovered as their hydroxide salt adding NaOH to the leaching solution. The filtrate so obtained after this process contains considerable amount of Lithium along with high concentration of sodium ion. Similarly, solvent extraction methods can be used to extract the valuable metals (Ni, Co, Mn) from the acid leached solution using the non polar solvents like D2EHPA, PC-88A, Cynex- 27217). After extraction, the raffinate solution also contains lithium and sodium ion. The waste solution so produced should be treated with recycling process to extract the lithium ion. The separation of lithium ion from sodium ion is big challenge for this process.
Lithium is generally recycled in the form of lithium carbonate which has become a very essential substance in recent years. The uses of lithium carbonate has increased day by day due to its significant character and so the market value will go high in the future18). Beside the application of lithium carbonate in the cathode materials in the LIBs, it is also used to make other lithium compounds, like lithium chloride (LiCl), lithium hydroxide (LiOH), lithium bromide (LiBr) and lithium oxide (Li2O).
Generally lithium carbonate is produced by precipitation reaction adding saturated sodium carbonate to the lithium solution. This is the simple methods to recover the lithium as lithium carbonate but the Li2CO3 so produced is contaminated with sodium ion and it has low purity17,19). In order to remove the impurities especially Na ion from the lithium solution the lithium solution should be treated with solvent extraction methods.
Solvent extraction is generally recognized methods for the separation and purification of various metal ions. In solvent extraction, several extractants such as PC88A20), D2EHPA21), Cyanex 272/Cyanex 301/Cyanex 30222,23) are used to separate the cobalt nickel and manganese from other source. The literature revealed that, extractant, Cyanex 272 can easily separate nickel from cobalt where D2EHPA has strong separating effects between cobalt and manganese19). Similarly, the solvent extraction process had been done to recover the lithium ion from the brine solution using various commercial extractants. The brine solution contains lithium ion 0.01% - 0.2% by wt. with considerable amount of chloride salt of sodium, potassium calcium and magnesium are present. The above chloride salts are removed from the brine solution using the solar evaporation and crystallization of these salts24). Then the solution containing lithium ion is sent into the solvent extraction to produce the highly pure lithium product25,26) investigated the recovery of lithium from the geothermal water congaing impurities like Na, K, Mg, and Ca using the single extrants like D2EHPA(di-(2-ethylhexyl)phosphoric acid) and MEHPA(mono-2-ethylhexyl-phosphonic acid). Moreover, he further revealed that addition of neutral solvents like TBP to D2EHPA/MEHPA make more effective for the selective recovery of lithium ion from the geothermal water.
On the other hand D2EHPA has been used to remove Mg impurities from the brine solution extracting magnesium in the solvents27). Even though there is effective extraction of magnesium ion, the loss of lithium was also significantly observed due to the co extraction of lithium. The removal of small quantity of calcium and magnesium ions from the brine solution was also observed using the binary extractants of D2EHPA and Versatic acid10 where loss of lithium ion co extraction is not much found28,29). Li et al.30) performed the removal of magnesium ion from the lithium solution extracting the magnesium ion in the solvent using mixture of base solvent (Aliquat 336) and acid solvent (Versatic acid10).
In this paper, we tried to recover the lithium ion from the waste solution that has been produced after the separation of valuable metals (NCM) from waste the LIBs in Sebit Chem recycling chemical company. The waste solution is highly alkaline (pH = 10.5 ~ 11.5) and contains 0.5% ~ 1% lithium ion along with contaminated with high content of sodium ion (3 ~ 6.5%) and trace amount of Ni, Co, Mn, Fe, Al, Cr, Cu etc. The metal ions content of waste lithium solution has been shown in the Table 1. The solvent extraction method has been tested with several acidic extracts and mixture of acidic and neutral extractants like TBP to extracts the lithium ion in the suitable solvents and stripped with different acids to recover the pure and concentrated amount of lithium solution. Unlike the research work in lithium ion is extracted in the solvent at certain equilibrium pH and impurities like Na ions has been sent to the raffinate27,29). The addition of TBP with acidic extracts increases the lithium extraction effects because TBP forms the dimer structure in organic solvents and dimer form is breakdown into small monomers providing more molecules for extraction30,31).
The extracted lithium ion in the solvent has been stripped with different concentration of mineral acids to recover the lithium ion. Finally, the pure stripping lithium solution free from sodium ion has been treated with ammonium bicarbonate to form the highly pure lithium carbonate.
Table 1.
ICP data of the various elements present in the waste lithium solution
2. Materials and Methods
2.1. Materials
The lithium solution as a raw materials was taken from the waste solution which has come after the valuable materials (NCM) were extracted from the waste LIBs. The contents of lithium solution have been shown in Table 1. The solvents D2EHPA (≥97%) was obtained from the Sigma Aldrich (USA), Trin-n-butyl phosphate, Methyl isobutyl ketone (98%) from samchun chemicals South KoreaTBP (≥99%) from samchun chemicals (South Korea), PC88-A (≥98%) fro sigma Aldrich (USA). Similarly Kerosene (95%), NaOH (50%), hydrochloric acid (37%) and sulphuric acid (98%), ammonium carbonate (99%) and ammonium bicarbonate (99%) were purchased from the samchun chemicals south Korea. The physical properties of the solvents mentioned from the their supplier are shown in Table 2.
Table 2.
Physical properties of the solvents used for extraction
2.2. Solvent extraction
The solvent extraction experiments were done in the 1000 ml separating funnel and equilibrium pH was measured by the pH meter DKK TOA HM-31P, Japan. In order to find the optimized condition and appropriate solvents, solvent extraction experiments were done with several times at different conditions with different mixtures of following solvents. The concentration of all solvents used in the experiments have been expressed as volume %.
1. 25%Di-(2-ethylhexyl) phosphoric acid (D2EHPA) + 75% kerosene
2. 25% 2-ethylhexyl phosphoric acid mono-2-ethylhexyl ester (PC-88A) + 75% kerosene
3. 60% Tributyl phosphate (TBP) + 40% Methyl isobutyl ketone (MIBK)
4. 25% DEHPA + 15%TBP + 60% kerosene
5. 20% DEHPA + 20%TBP + 60% by kerosene
The extraction experiments were done at different equilibrium pH and found the appropriate pH condition at which maximum extraction was observed. Then again extraction experiments were done for the different types of solvents maintaining the same pH condition to find the effective solvent mixture. The pH of the raffinate was maintained by controlling the amount of 50%NaOH during the saponification process. In saponification process, the solvent was hydrolyzed by aqueous solution of NaOH to give the sodium salt of organic compound so that sodium ion in the organic solution was easily replaced by the metal ions when metals solution are mixed with them. The saponification reaction is mentioned below.
After finding the appropriate solvents and equilibrium pH, the ratio of organic/aqueous (O/A) ratio was optimized on the basis of lithium extraction. In this way, after optimizing the equilibrium pH, appropriate solvent mixtures and O/A ratio, extraction of lithium ion from the waste solution was again done in several steps maintaining the above optimum conditions in order to get the maximum extraction efficiency of lithium ion. The raffinate from each solvent extraction was analyzed by a Perkin Elmer Avio 500 ICP-OES, and the composition of the organic phase was determined through a mass balance. Then Lithium extraction by the given solvent is measured by the following equation.
First of all, the mass of the lithium ion in the raffinate was measured by above mentioned ICP-OES. Then the mass of metal ion in the organic solution was calculated by subtracting mass of lithium ions in the raffinate solution from the initial mass of lithium ion in the aqueous solution.
2.3. Stripping the solvent
After the solvent extraction, the stripping of the solvent containing lithium ion was done by sulphuric acid or hydrochloric acid in several steps in order to recover the lithium ion. The appropriate weight percentage of the striping agent was determined on the basis of the lithium recovery percentage and pH of the recovered lithium solution. The stripping percentage was calculated from the relation mentioned below.
2.4. Synthesis of lithium Carbonate powder
The stripping solution which contains lithium ion recovered from the waste solution was treated with ammonium bicarbonate at 70 ~ 80°C to produce the lithium carbonate. The lithium carbonate so produced was transferred to another beaker containing water and the solution was stirred for one hour to remove the Na ions. The lithium carbonate powder was then dried at 105°C for 5 hours to get very pure and dry lithium carbonate.
3. Result and Discussion
3.1. Exaction of Lithium ion of various solvents at different pH
First of all, four types of solvents were first chosen to extract the lithium ion at different pH with same O/A ratio = 1.
1. 25% by volume of D2EHPA (diluted with kerosene)
2. 25% by volume of PC-88A (diluted with kerosene)
3. 60% by volume of TBP diluted with MIBK and
4. 20% by volume of TBP + 20% by volume of D2EPHA diluted with kerosene for the extraction of lithium ion.
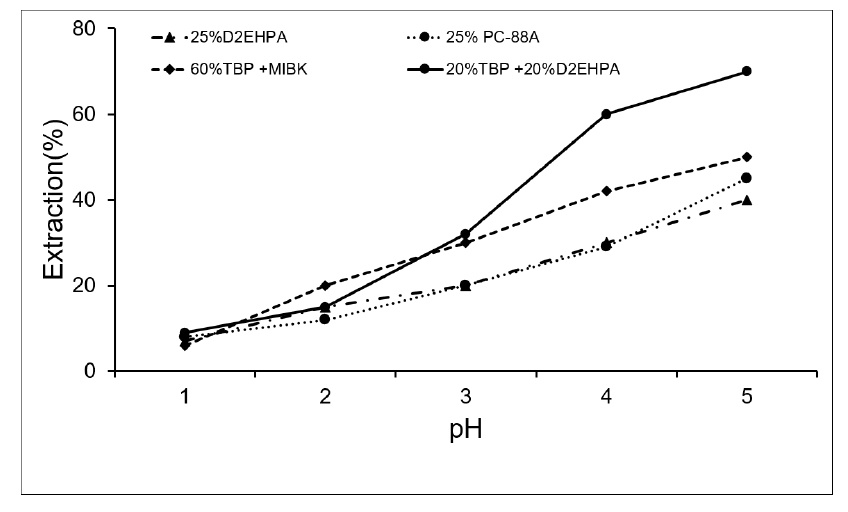
The extraction of lithium ion of different solvents at various pH are shown in Fig. 1. The pH of the raffinate at equilibrium state was maintained controlling the amount of 50% NaOH added to the solvent during the saponification. Higher the amount of sodium hydroxide added, higher was the pH of the raffinate. From the result obtained in Fig. 1 and Table 3 showed that each solvent had low extraction rate at low pH and increases with increasing equilibrium pH. This is so because lithium ion can make a strong bond with solvents at higher pH. However, the time taken for separation of solvent and aqueous solution was also higher in higher pH. Out of four set of solvents, the mixture of solvent of 20% by volume of TBP + 20% by volume of D2EPHA had the highest extraction% throughout all pH except at pH = 1 because the mixture of solvent TBP + MIBK has little higher extraction of lithium than mixture of solvent of 20% by volume of TBP + 20% by volume of D2EPHA at that pH. Based on the results, the appropriate equilibrium pH is chosen about 4.2 and the best solvent for the extraction of lithium is mixture of 20%D2EHPA and 20%TBP. TBP is well known interface modifier for extractants which help to increase the extraction efficiency and make easy for phase separation28). We noticed that addition of TBP in D2EHPA make for shortening the phase separation of time. At pH 5, the extraction of all lithium is very higher than other pH but the time taken for phase separation of solvent and aqueous is very long.
Table 3.
Effect of equilibrium pH on the extraction of Li (25% D2EPHA, 25%PC-88A, (60%TBP + MIBK), (20%TBP + 20% D2EPHA), O/A = 1)
3.2. Exaction of Lithium ion from different Solvents
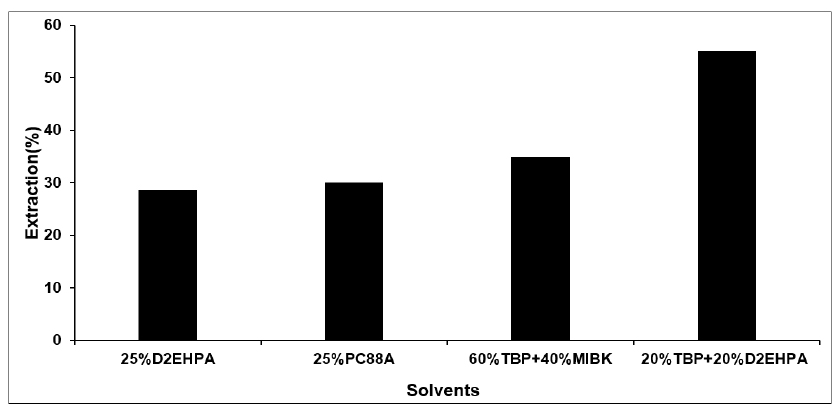
After optimizing the appropriate pH for different solvents, if we observe the extraction % of the different solvent (O/A = 1) at the same pH about 4.2, the fourth solvents 20%TBP + 20%D2EHPA give the highest extraction of lithium ion, which has been shown in the Fig. 2 and Table 4. The results showed that when TBP solvent was added to the acidic solvents like D2EHPA lithium extraction was increased. The Table 4 and Fig. 2 showed the mixture of solvent of TBP (20% by volume) + TBP (20%) diluted with kerosene gave 55% lithium ion extraction from the lithium solution while other solvents gave lower lithium extraction % by wt (D2EHPA 25% = 28.7, 25% PC-88A = 30%, 60% TBP + 40%MIBK = 35%) under the same condition (pH = 4.2, O/A = 1).
Table 4.
Effect of different Solvent on the extraction of Li at PH = 4.2, O/A = 1
| solvent | extraction % |
| 25%D2EHPA | 28.7 |
| 25%PC88A | 30 |
| 60%TBP + 40%MIBK | 35 |
| 20%TBP + 20%D2EHPA | 55 |
3.3. Exaction of Lithium ion at different O/A ratio using the mixture of Solvent TBP + D2EHPA
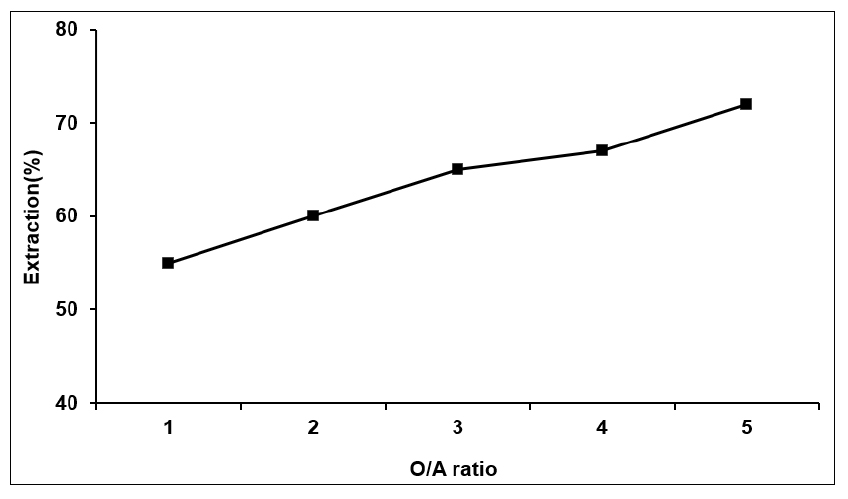
From the above experiment, the mixture of solvent of 20% by volume of TBP + 20% by volume of D2EHPA has the higher extraction of lithium ion than other solvents when O/A ratio was 1 at the equilibrium pH = 4.2. In order to increase the more extraction efficiency of this solvent more and more, the extraction of Li+ was done at higher O/A ratio at the same pH 4.21. The result showed that extraction of lithium ion was slightly increases with increase in O/A ratio and maximum extractions was found about 72% by wt. at O/A = 5. The results were shown in Table 5 and Fig. 3. It was therefore chosen O/A ratio = 5 for the further experiment during extraction of lithium ion.
Table 5.
Effect of O/A ratio on the extraction of lithium 20% D2EHPA + 20% TBP by wt at pH = 4.21)
| O/A ratio | Extraction% |
| 1 | 55 |
| 2 | 60 |
| 3 | 65 |
| 4 | 67 |
| 5 | 72 |
Table 6.
Effect of extraction steps on the extraction of lithium for 20% D2EHPA + 20% TBP by wt at pH = 4.21, O/A = 5
| Nof extraction steps | Extraction% |
| 1 | 72 |
| 2 | 85 |
| 3 | 95 |
| 4 | 97 |
3.4. Exaction of Lithium ion at different extraction steps
From the above experiment, extraction of lithium ion was higher (72%) from the one step extraction for the solvent mixture 20%TBP + 20%D2EHPA when O/A ratio was 5 at the equilibrium pH = 4.2. But the extraction of lithium ion can be further increased up to more than 90% by wt. when extraction steps were increased in more than one step. So, extraction of lithium ion from the above solvent mixture at the above optimum condition was done by increasing the extraction steps and found that extraction of lithium is more than 97% when extraction steps was four. The results were shown in the Table 6.
3.5. Time for phase separation during Lithium ion extraction using the solvent mixture of 20% by volume of TBP + 20% by volume of D2EHPA at pH 4.21 but at different O/A ratio
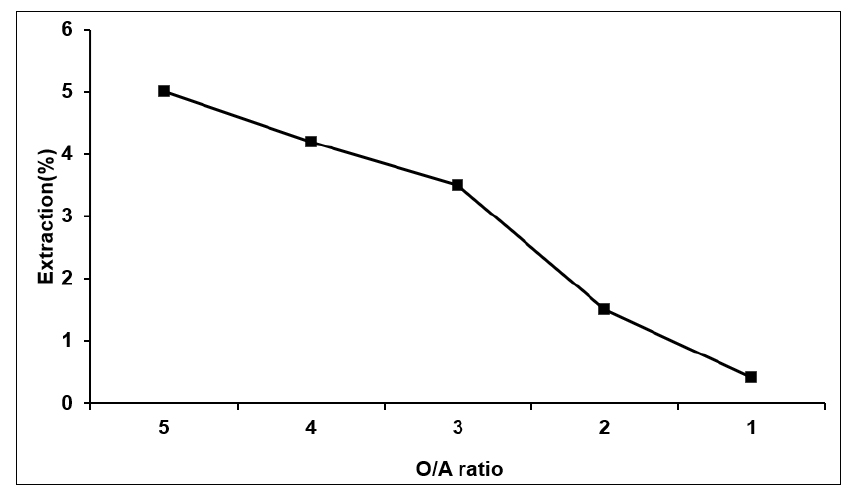
Time of phase separation is also important factor for the solvent of extraction. If time of separation is very slow efficiency of the process is decreased because it takes long time to complete the process. It is therefore important to find the accurate O/A ratio which is occurred at very short time. However, very short separation time is also not appropriate because extraction of lithium ion is decreased. So we need to maintain the balance of separation time and extraction of lithium ion. We measured the separation time for different O/A ratios for the 20% by volume of TBP + 20% by volume of D2EHPA at the same pH = 4.21. The results were shown in the Fig. 4 and Table 7. The result showed that separation time increased with decrease in O/A ratio. The short time was about 40 minutes for the separation of two phases at O/A = 5 when the separation of time was very long about 5 hour when O/A ratio is 1. The results were shown in graph and table.
Table 7.
Effect of O/A ratio on separation time of organic and aqueous (TBP + D2EHPA, pH = 4.21) solution
| O/A ratio | Time |
| 5 | 40 minutes |
| 4 | 1.5 hours |
| 3 | 3.5 hours |
| 2 | 4.2 hours |
| 1 | 5 hours |
3.6. Stripping of Lithium Solution from the Solvent
Li ion captured in the solvent was stripped by first 12% by wt. of sulphuric acid and found that stripping of lithium ion was very effective. But when lithium sulfate solution obtained from stripping the solvent treated with ammonium carbonate did not give much amount of lithium carbonate. It means lithium recovery was very low as lithium carbonate. This is so because adding ammonium carbonate into the lithium sulfate solution make the reaction going into another direction where it formed little soluble double salt of Li2SO4.(NH4)2SO430) It is generally formed at high concentration of SO42- ion. Lithium ion in the double salt cannot form the lithium carbonate. As a result lithium recovery from the lithium carbonate became less.
Since HCl is stronger than sulphuric acid because pKa value of HCl is-6.3 while H2SO4 has -3. Therefore, compared to H2SO4 even lower concentration of HCl can easily strip the lithium solution. Unlike with H2SO4, ammonium carbonate cannot form any double salt with lithium chloride solution and no lithium ion is lost during the formation of lithium carbonate. We therefore tried to strip the lithium solution from the solvent mixture containing lithium solution using the various concentration of hydrochloric acids using O/A ratio = 10 in two stripping steps. The stripping results are shown in the following Table 8. The result showed that 8% by wt. of HCl is efficient for stripping the solvent to recover the lithium chloride solution because lithium recovery was about 97% and pH of the solution is 2.1. The ICP data of the lithium chloride solution obtained from the stripping of solvent using 8% by wt of HCl is shown in the Table 9. ICP-OES data of the stripping solution data showed that about 92% by wt. of Na ions were removed from the lithium solution. Compared to the waste lithium solution, the recovered lithium chloride was highly pure with less contaminated with sodium ion. Moreover, after stripping the solution, lithium concentration in lithium chloride was nearly double than raw material.
Table 8.
Effect of HCl concentration on Li stripping efficiency from the solvents (20%TBP + 20%D2HPA O/A = 10) and respective pH of the lithium chloride solution
| HCl% | Stripping % | pH (stripping solution) |
| 3 | 48 | 3.5 |
| 7 | 95 | 3 |
| 8 | 99 | 2.1 |
| 11.5 | 99 | 1.53 |
| 12.5 | 99 | -0.09 |
Table 9.
ICP analysis data of stripping solution (Lithium chloride) obtained stripping by 8% HCl, O/A = 10
3.7. Making high purity of lithium carbonate
Similarly, ammonium carbonate was first added to make lithium carbonate to synthesis of lithium carbonate. However, the ammonium carbonate is unstable even at room temperature and decomposes easily into ammonia gas. So, we tried with ammonium bicarbonate which is more stable upto 36°C.
Ammonium bicarbonate also easily reacts with Lithium chloride to give lithium bicarbonate29).
The total amount of 99% of ammonium bicarbonate was added 1.2 times of required amount as per stoichiometric equation 3. Then solution was slowly heated upto the 70°C and stirred for 30 minute at this temperature to complete the reaction. The white precipitate of lithium carbonate was separated from the solution using the vacuum filtration. The lithium carbonate so obtained from the filtration was again transferred into the beaker containing water (1.6 times wet lithium carbonate) and stirred continuously upto 30 minutes in slurry condition to remove the unwanted Na ion. Then the solution was again filtered and collected the very pure lithium carbonate. Some lithium ions are lost during the washing of lithium carbonate due to its solubility. So the washing solution containing lithium and sodium ion was again recirculated mixing with raw material for extraction in order to avoid the loss of lithium ions. The lithium carbonate was dried at 105°C over 5 hours. After five hours, very pure lithium carbonate (purity 99.2%) was obtained (shown in Fig. 5) and lithium recovery was about 55% by wt. The filtrate obtained from the lithium carbonate filtration contains lithium ion about 1600 ppm which causes the loss of lithium ion. So the filtrate obtained from the filtration of lithium carbonate is evaporated upto two times and the evaporated solution is also recirculated to avoid the loss of lithium ion, the purity and impurities of lithium carbonate has been shown in the Table 10.
Table 10.
ICP-OES analysis data of metal ions impurities present in dry lithium carbonate powder manufactured from the waste lithium solution and its purity measured by titration
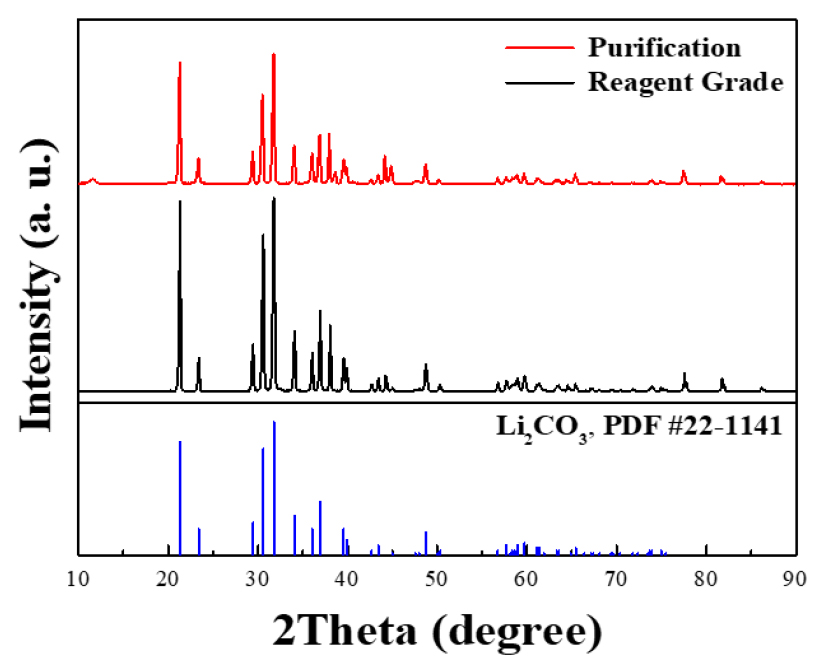
The recovered lithium carbonate was analyzed by XRD to confirm whether it was structurally consistent with high-purity lithium carbonate (Sigma-Aldrich, CAS Number 554-13-2). It was confirmed that the recovered lithium carbonate was a crystalline phase uniformly present as lithium carbonate without an impurity (Li2CO3) peak (around 2θ = ~21°). In particular, it was confirmed that all XRD peaks analyzed were highly consistent with lithium carbonate (Li2CO3, PDF no. #22-1141), which is the main peak. The lithium carbonate so obtained was also analyzed by XRD and its XRD pattern is compared with that of commercial grade of lithium carbonate (≥99%) found in the market. The XRD pattern in the Fig. 6. The lithium carbonate recovered through this result is considered to have the same structure compared to the purified lithium carbonate.
4. Conclusion
The waste lithium solution so produced after NCM separation from the waste lithium secondary battery contains lithium ion about 5000 mg/kg ~ 1% with rich in amount of Na ion. The solvent extraction has been done to extract the lithium ion from waste lithium solution using the mixture of solvent 20%by volume of D2EHPA + 20% by volume of TBP (diluted with kerosene) in four extractant steps at equilibrium pH 4.2 ~ 4.5 and O/A = 5. Under this condition, above 95% lithium solution is extracted where most of Na ion passes into the raffinate so that about 92% by wt. of Na ion was removed from lithium ion. The extracted lithium ion was stripped by 8% HCl at O/A = 10 under the equilibrium pH = 2 ~ 3 to get concentrated lithium chloride solution. The lithium chloride solution was treated with ammonium bicarbonate at 70°C to get the powder of lithium carbonate. The titration result showed that the purity of lithium carbonate so obtained is about 99.2%.