1. Introduction
2. Experimental
2.1. Reagents and apparatus
2.2. Liquid-liquid extraction process
3. Results and discussions
3.1. Influence of extractant concentration on thorium extraction
3.2. Prieme JM-T treated with 50% sulfuric acid
3.3. Extraction isotherm (Mc-Cabe Thiele diagrams) for thorium
3.4. Stripping of thorium from loaded organic phase
4. Conclusions
1. Introduction
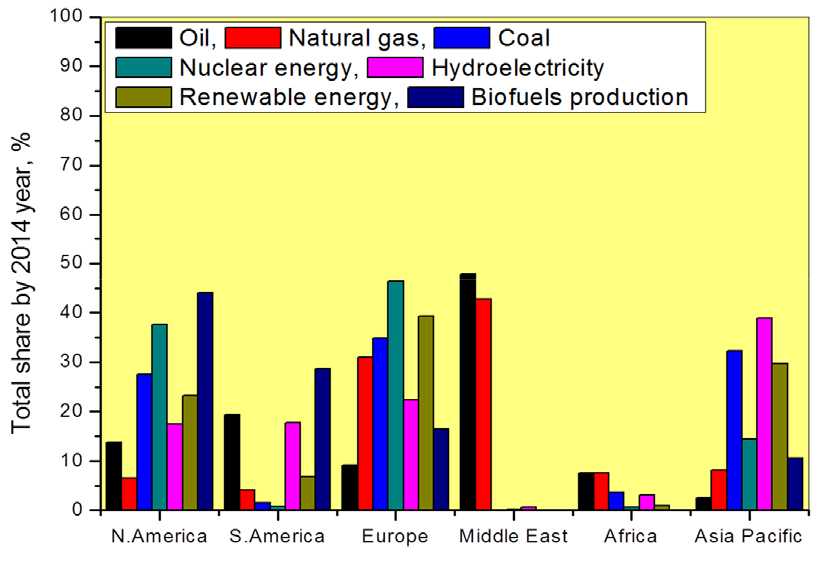
Six major issues are most concern in new millennium fourth industrial revolution development. Those are global energy resources (Fig. 1), global electricity production, global accessibility and non-accessibility of electricity, lifecycle of greenhouse gas emissions, global non-renewable energy resources and global nuclear reactors scenario. Primary energy supply was predicting 82 to 76% reduction, increasing the renewables from 10 to 16% hydro and nuclear energy utilization was not much change in year 20201). By using lignite, coal, oil natural gas etc generating the high carbon emissions whereas biomass, nuclear, hydro and wind energy sectors will generate very low carbon emissions when compared with fossil fuel energy sources1).
Nuclear power mainly depends on the uranium and thorium resources. A uranium nuclear reactor functions by flowing water over the surface of the zirconium metal tubes with uranium fuel sealed inside the fuel rods. Much radioactive material is possible to contaminate in air. Humans and all living animals get effected by this radiation. Whereas liquid fluoride thorium reactor (LFTR) are generate 0.01% of waste only as well as generate 1,000 MW of electricity from 0.9 MT of natural ThO2. Moreover, proliferation material is zero, fission products such as iodine etc can chemical process at on-site. In addition, actinides such as neptunium etc will generated up to 1 kg, which can be separated and recycled onsite. Three major factors are exposes that, thorium is better nuclear sources; those are thorium is more abundant in nature than uranium, it is a fertile rather than fissile and chemically distinct (easy to removal the radioactive materials). Finally, molten salt reactors are well suited to thorium fuel as normal fuel fabrication is avoided2,3,4,5).
For thorium processing from monazite, one of the better technique is hydrometallurgy. Various reports are available in past literature on thorium extraction from monazite or synthetic liquors6,7,8,9,10,11,12). The present study is focused mainly on as much as thorium extraction with less interfere metals and experimental conditions for liquid-liquid extraction processing was optimized.
2. Experimental
2.1. Reagents and apparatus
Aqueous sample analysis of metals such as thorium, cerium and others were analyzed by using in ICP-OES manufactured by Thermo Scientific, USA model iCAP 6000 Series. The commercial grade amine based extractant such as Primene JM-T amine was supplied by Cognis Corporation USA and used as it is without purification. The present study we were used diluent as A150 (commercial form of kerosene) supplied by Samsung, Korea and all other reagents used were analytical reagent grade. The aqueous feed of monazite was used.
2.2. Liquid-liquid extraction process
Metal distribution ratios were determined by shaking equal volumes (30 mL each) of aqueous and organic phases for 30 min in a separating funnels using a mechanical shaker at 25±0.5°C. Preliminary experiments showed that the extraction equilibrium was attained within 5 min for thorium. The solutions were then allowed to settle, the phases separated, and thorium and other metals in the aqueous phases determined by ICP-OES. The concentration of metal ion in the organic phase was then obtained by mass balance. The distribution ratio, D was defined as the ratio of the concentration of metal ion in organic phase to that in the aqueous phase (D = metal in organic phase / metal in aqueous phase). The percentage extraction (% E) calculated by the following equation: % E = (D x 100 / D +1) A:O. The general agreement between the distribution ratios values obtained was within ± 5%. The present investigations for thorium separations using amine based extractants, following experimental conditions are constant for the all experiments: time = 5 min, temperature = 25°C and aqueous: organic (A:O) = 1:1 that means unit phase ratio.
3. Results and discussions
3.1. Influence of extractant concentration on thorium extraction
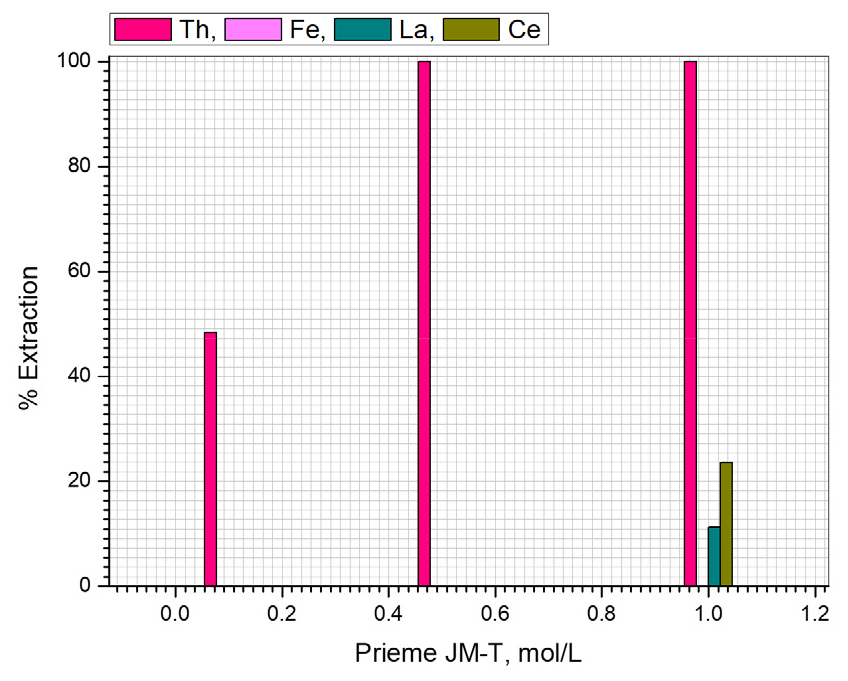
Monazite leaching solution contained 2720 mg/L of thorium, 150 mg/L of iron, 1603 mg/L of lanthanum and 1913 mg/L of cerium. The first screening test was conducted with commercial extractant Primene JM-T; it was varied from 0.1 mol/L to 1.0 mol/L. The other experimental conditions such as phase ratio (Aqueous (A) / Organic (O)) was one and temperature is 25°C. The obtained extraction was presented in Fig. 2. The data was make known that, with 0.1 mol/L of Prime JM-T 48.3 % of Th was extracted, after that both the cases 0.5 and 1.0 mol/L of Prime JM-T, Th was quantitatively complete extracted (100%). Whereas, other associated metals were not extracted with Prime JM-T up to 0.5 mol/L concentration and with 1.0 mol/L Prime JM-T 11.2 % of La and 23.5 % of Ce were co-extracted with thorium.
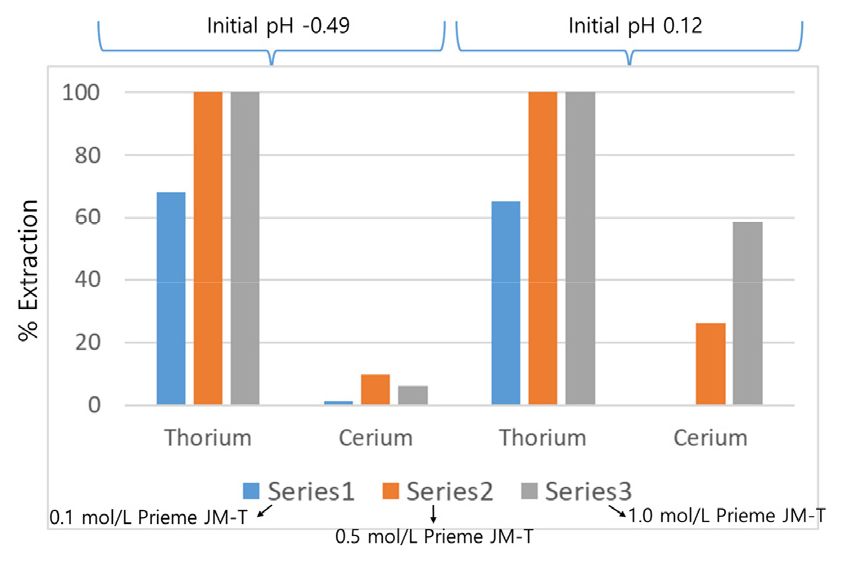
Based on the first experiment, Primene JM-T was selected for further thorium extraction process development. The extractant concentration was varied from 0.1 to 1.0 mol/L concentration. The significant extraction was observed mainly of thorium and cerium (Fig. 3). With 0.1 mol/L of Primene JM-T up to 68% of thorium was extracted, whereas cerium was extracted less than 1.2%. On the other hand, from 0.5 mol/L to 1.0 mol/L of Primene JM-T concentration thorium was 100% extracted, whereas cerium was extracted up to 9.7% at initial pH of feed was -0.49, at pH 0.12 cerium was extracted 26.2% with 0.5 mol/L of Primene JM-T and 58.7% with 1.0 mol/L of Primene JM-T. The data indicates that lower acidic region (-0.49 pH) is the much promising to thorium, in addition cerium co-extraction was very nominal (less than 1.2%).
3.2. Prieme JM-T treated with 50% sulfuric acid
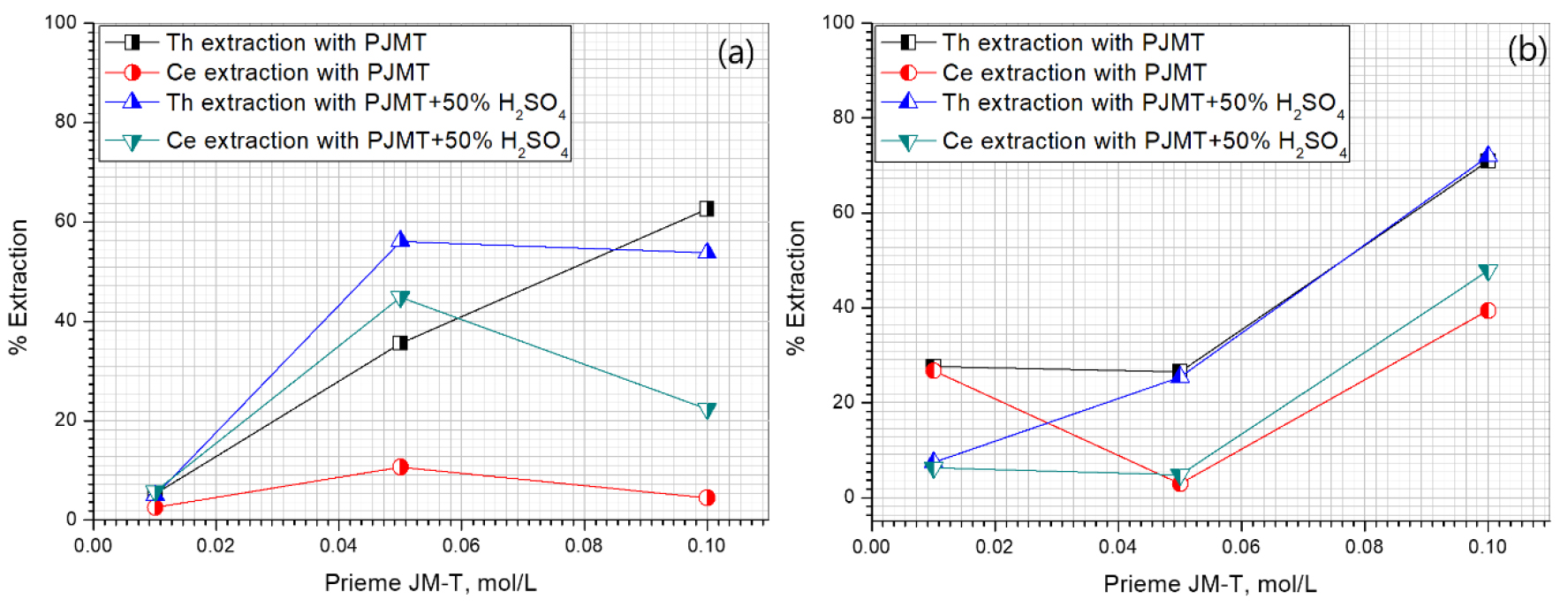
Prieme JM-T was treated with 50% sulfuric acid to neutralize the extractant basic nature. Further lower concentration of the Primene JM-T and 50%H2SO4- Primene JM-T were tested. Both the extractants were varied from 0.01 to 0.1 mol/L of concentration, at two different initial pH conditions such as -0.49 and 0.12, the obtained data was presented in Fig. 4. Cerium extraction was more favorable with acidified extractant and pH 0.12 feed condition.
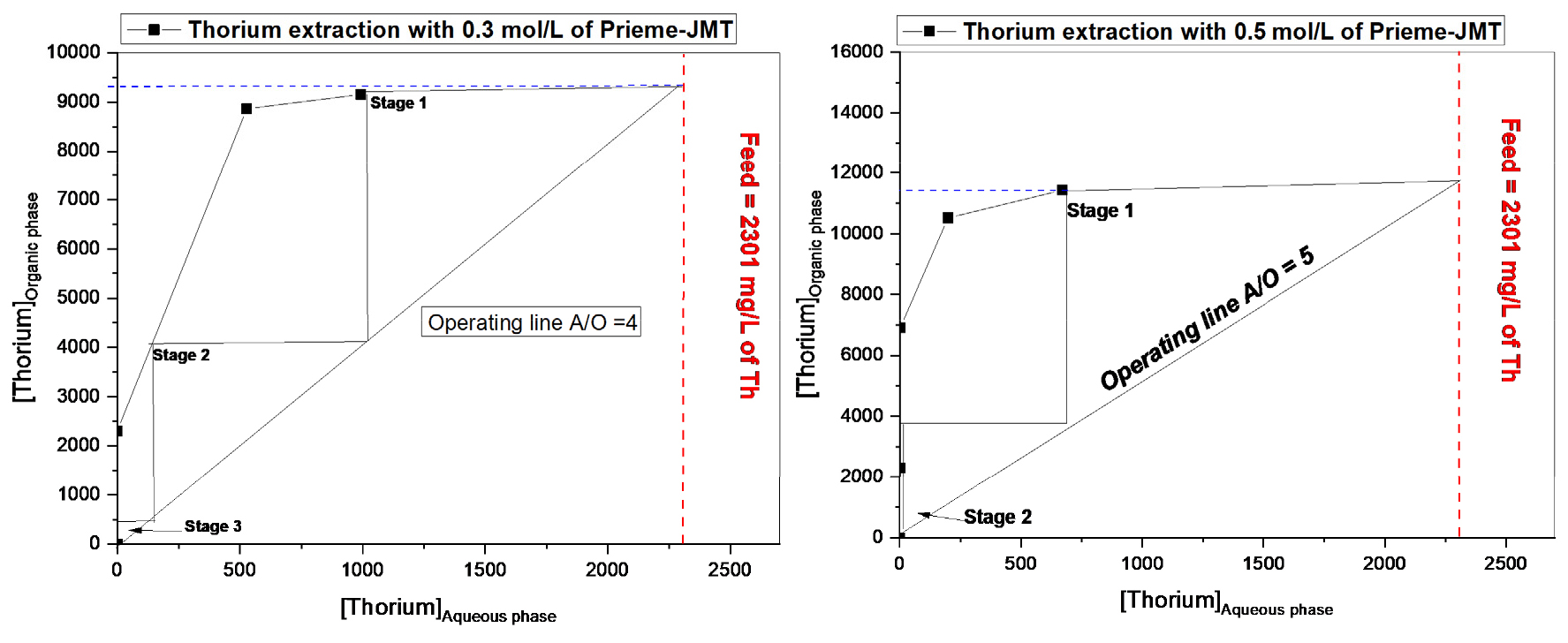
3.3. Extraction isotherm (Mc-Cabe Thiele diagrams) for thorium
Two different concentration of the extractant was tested for extraction isotherm construction for thorium extraction. With 0.3 mol/L of Primene JM-T with phase ratio (A/O) 4 needs three extraction stages to load the above 9 g/L of Th from aqueous feed containing 2301 mg/L. Whereas with same feed, above 11 g/L needs to load the two extraction stages at phase ratio 5 with 0.5 mol/L of Primene JM-T (Fig. 5).
3.4. Stripping of thorium from loaded organic phase
The stripping of the targeted metal is most significant process in hydrometallurgical process development. The present study the loaded organic phase was water washed by two or three times then followed by acidified water treatment to remove un-wanted metals from thorium then finally applied the thorium stripping by using 1 mol/L of hydrochloric acid was able to recover the thorium more than 90% at single stripping stage. It was indicated that multiple stripping stages could able to strip the loaded thorium.
4. Conclusions
The present study was concluded that, Primene JM-T is the best extractant for extract the thorium from monazite leach liquors. Moreover, very low acidified leach liquor (-0.49 pH) condition is more suitable for thorium complete extraction with very nominal cerium co-extraction. The water washings and diluted sulfuric acid solutions could able to remove the all impurities from loaded organic phase. The most suitable reagent is HCl to recover the loaded thorium, successfully.