1. Introduction
2. Experimental
2.1. Preparation of feedstock by deoxidation for HCl leaching
2.2. HCl leaching of Ti mixture obtained by deoxidation
2.3. Analysis
3. Results and discussion
3.1. Influence of leaching time on leaching behavior of Ti mixture
3.2. Influence of temperature on leaching behavior of Ti mixture
3.3. Influence of HCl concentration on leaching behavior of Ti mixture
3.4. Influence of gas for bubbling on leaching behavior of Ti mixture
4. Conclusions
1. Introduction
Currently, the Kroll process is used to produce titanium (Ti) sponges commercially1,2,3,4,5). The Kroll process is costly because of the low productivity arising from the highly exothermic reaction for reduction and batch-type operation3,4,5,6). In addition, approximately 10 – 20 % of Ti sponge production is low-grade scrap, known as off-grade Ti sponge, owing to contamination by oxygen (O) and iron (Fe)7). Off-grade Ti sponges are inexpensively utilized in the Fe industry instead of being remelted for Ti ingot production4,8). Therefore, efficient recycling of off-grade Ti sponges is important to reduce the cost of Ti metal and compensate for its low productivity.
To recycle off-grade Ti sponges for the production of Ti, their O concentration should be decreased to below 0.18 mass% (1800 ppm) to satisfy the American Society for Testing and Materials (ASTM) Grade (Gr.) 1 for Ti9). Additionally, the removal of Fe is required when considering the reuse of off-grade Ti sponges as a pure Ti metal4,8). However, direct removal of Fe from off-grade Ti sponges during deoxidation is impossible. Fortunately, because Fe serves as a β stabilizer for β-Ti alloys10,11), off-grade Ti sponges can be utilized for the production of β-Ti alloy ingots even if Fe remains after deoxidation. Therefore, the deoxidation of off-grade Ti sponges is important for Ti recycling.
Many studies involving the removal of O in Ti from various Ti feedstocks using calcium (Ca)12,13,14,15,16,17,18,19,20,21,22,23,24), magnesium (Mg)25,26,27,28,29,30,31,32,33,34,35,36), rare earth metals37,38,39,40,41,42,43,44,45,46,47,48), and electrons49,50,51) as reducing agents have been extensively conducted. Among these investigations, metallothermic reaction is one of the effective deoxidation method12,13,14,15,16,17,18,19,20,21,22,23,24,25,26,27,28,29,30,31,32,33,34,35,36,37,38,39,40,41,42,43,44,45,46,47,48). In this method, the O in Ti reacts with a metallic deoxidant that has a stronger affinity for O than Ti does, thereby removing O as a metal oxide. After the reaction, leaching is typically employed to remove the metal oxide and recover the deoxidized product.
Leaching contributes significantly to the removal of the metal oxides after Ti deoxidation. This is because when leaching is not sufficiently and appropriately conducted, the remaining metal oxide causes defects in the final Ti product. In addition, when leaching is excessively conducted, deoxidized Ti is significantly contaminated by O during leaching owing to the high affinity of Ti for O24). Therefore, it is important to carry out the leaching under suitable conditions to completely remove the metal oxide from deoxidized Ti and simultaneously minimize the contamination of Ti by O.
In the past, however, leaching was simply treated as a post-treatment. For example, in many studies, the hydrochloric acid (HCl) leaching was conducted at a temperature lower than room temperature without considering important factors of leaching, such as pulp density, acidity, and bubbling gas. However, recently, a few systematic studies on the HCl leaching of deoxidized Ti have been conducted.
Sarkar reported the leaching mechanism of Mg in titanium hydride (TiH2) obtained after the magnesiothermic reduction of titanium dioxide (TiO2) under hydrogen gas (H2). The influence of the dissolution kinetics as a function of temperature, particle size, and pH of the HCl solution on the concentration of Mg in the product was investigated52). However, the concentration of O in the TiH2 product as a function of the leaching conditions was not considered. Choi reported the influence of HCl leaching on the O concentration in Ti obtained after the magnesiothermic reduction of TiO2 via self-propagating high-temperature synthesis53). However, the O concentration in Ti obtained after leaching was 1 mass%, which exceeded 0.18 mass%. Suzuki reported the influence of type of bubbling gas, such as argon (Ar), air, and oxygen gases (O2), during acetic acid leaching on the dissolution behavior of calcium oxide (CaO) and O concentration in titanium aluminide (Ti3Al) products obtained after the calciothermic reduction of a mixture comprising aluminum oxide (Al2O3) and TiO254). However, no significant differences were observed.
As mentioned previously, to recycle off-grade Ti sponges into high-value products, it is essential to understand the behavior of O concentration in Ti during leaching after deoxidation. Specifically, contamination by O should be minimized and the O concentration should be below 0.18 mass% in the final Ti product to satisfy the criteria of the ASTM Gr. 1. In addition, understanding the behavior of Fe, Ti, and Mg during leaching is also important when a Ti mixture is obtained after the deoxidation of off-grade Ti sponge using Mg. However, such investigations have not been reported to date.
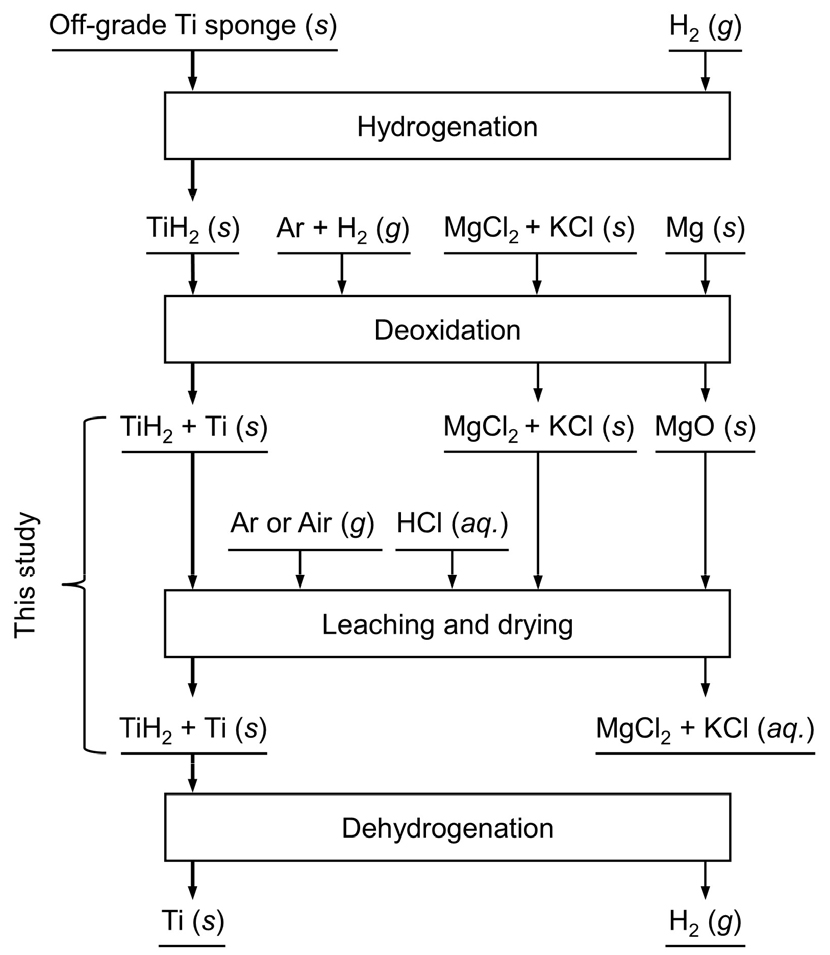
Fig. 1 shows a flowchart for the deoxidation of an off-grade Ti sponge using Mg under an Ar and H2 mixed gas atmosphere32). As shown in Fig. 1, the concentration of O in Ti decreases after deoxidation and is affected by HCl leaching. Therefore, in this study, the influences of HCl leaching conditions on the O concentration in Ti were investigated. In addition, the leaching behaviors of Mg, Ti, and Fe in the mixture obtained by the deoxidation of an off-grade Ti sponge using Mg in an H2 mixed atmosphere were also investigated.
2. Experimental
2.1. Preparation of feedstock by deoxidation for HCl leaching
To prepare the feedstock for HCl leaching, an off-grade Ti sponge was deoxidized in advance. The off-grade Ti sponge (VSMPO-AVISMA Corp.) was converted into TiH2 by hydrogenation at 973 K for 2 h before deoxidation, as shown in Fig. 1. The concentration of O in the produced TiH2 was 1.175 mass%33). In addition, magnesium chloride (MgCl2, anhydrous, purity > 97.0 %, Wako Pure Chemical Industries, Ltd.) and potassium chloride (KCl, anhydrous, purity > 99.0 %, Wako Pure Chemical Industries, Ltd.) were dried in advance at 453 K for more than 72 h using a vacuum oven (VOS-601SD, EYELA, Co.) to remove any H2O from the reagents.
A mixture comprising 500 g of the produced TiH2 with a mean particle size of 162 μm33), 125 g of Mg metal (pellet, ϕ = 6 mm, diameter, h = 6 mm, height, purity: 99.99 %, RNDKOREA Co.), 750 g of MgCl2, and 250 g of KCl were placed in an Fe crucible and reacted at 933 K for 12 h under a 10 % H2 and Ar mixed gas atmosphere. After deoxidation, the Ti mixture consisting of the salts, unreacted Mg, the magnesium oxide (MgO) produced, TiH2, and Ti was obtained. This mixture was pulverized using a clean hammer, followed by a mortar and pestle. The mixture was then sieved using a sieve shaker (J-PVS, JISICO, Co.). Particles of the mixture less than 1.18 mm in size were collected and uniformly mixed before being used as feedstock for HCl leaching. Fig. 2(a) shows a photograph of the pulverized mixture obtained after deoxidation.
2.2. HCl leaching of Ti mixture obtained by deoxidation
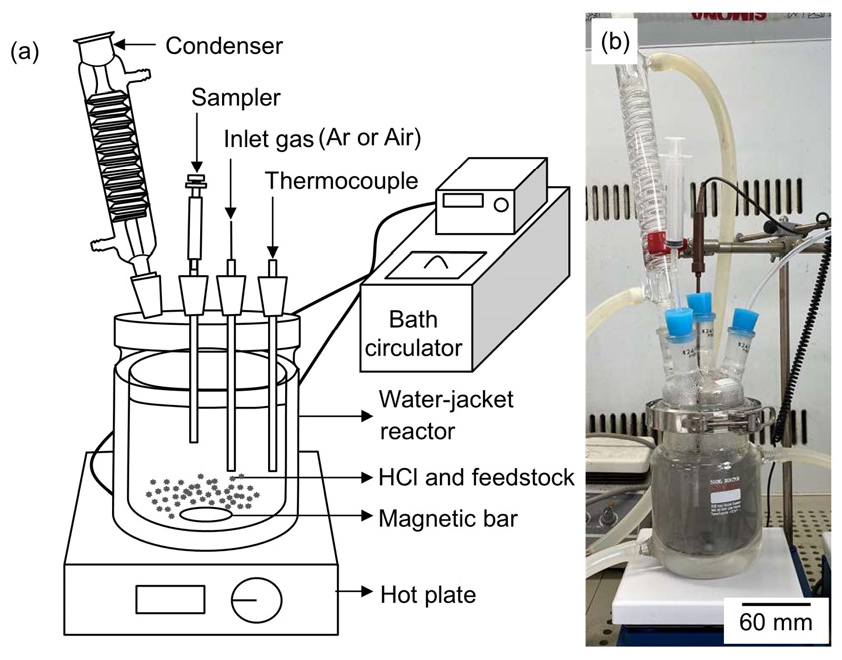
After collecting the Ti mixture with a particle size of less than 1.18 mm, HCl leaching, filtration, washing, and drying were conducted to remove salts, unreacted Mg, and the MgO produced, thereby yielding a Ti product composed of TiH2 and Ti. Fig. 3 shows a schematic and photograph of the experimental apparatus used for HCl leaching. Table 1 lists the experimental conditions used for HCl leaching.
Table 1
Experimental conditions for HCl leaching
| Exp. noa |
Weight of feed, wfeed / g |
Time, t' / min |
Temp., T / K |
HCl concentration, CHCl (mol·L−1) |
Gas for bubbling |
| 1 | 5 | 360 | 293 | 3.0 | Ar |
|
2 3 4 5 6 |
5 5 5 5 5 |
60 60 60 60 60 |
283 303 313 323 333 |
3.0 3.0 3.0 3.0 3.0 |
Ar Ar Ar Ar Ar |
|
7 8 9 |
5 5 5 |
60 60 60 |
313 313 313 |
0.1 1.0 2.0 |
Ar Ar Ar |
| 10 | 5 | 60 | 313 | 3.0 | Air |
Leaching experiments were performed using a four-necked double-jacket reactor connected to a bath circulator. The reactor was equipped with a condenser, a sampler using polytetrafluoroethylene (PTFE) tubing, a gas inlet using PTFE tubing, and a Teflon-coated thermocouple. The reactor was positioned on a hot plate and filled with 500 mL of 0.1 – 3 mol/L HCl solution (35 – 37 %, Junsei Co., Ltd.). The condenser was used to prevent the evaporation of the solution. The reaction temperature was controlled using a bath circulator and was accurately measured using a thermocouple. The solution was stirred at 300 rpm with a magnetic bar. Ar gas (purity = 99.999 %, 1000 ccm) was supplied to the solution through a submerged PTFE tube for bubbling before and during the experiments to remove dissolved oxygen from the solution as well as O2 from the atmosphere of the reactor. In a certain experiment, air (1000 ccm) was used for bubbling instead of Ar gas to compare its effect with that of Ar gas bubbling. The feedstock was added to the solution with 1 % pulp density (w/v) after the temperature of the solution stabilized at 283 – 333 K.
Throughout the leaching, 3 mL of the solution was periodically withdrawn using a sampler at intervals of 5, 15, 30, 60, 120, 240, and 360 min from the beginning of leaching and then filtrated using a membrane filter (pore size: 0.45 μm). Upon the completion of leaching, the solution was filtrated using a membrane filter (pore size: 0.45 μm) and a Ti product was obtained. Subsequently, the Ti product was washed with deionized water followed by acetone (purity > 99.8 %, Daejung Chemicals & Metals Co., Ltd.), and then dried at 298 K for 60 min.
2.3. Analysis
The O concentration in the samples was analyzed using a N/O/H determinator (TCH-600, LECO Co.). The concentrations of Mg, Ti, and Fe in the solution and samples were analyzed using an inductively coupled plasma optical emission spectrometer (ICP-OES: Optima 5300DV/iCAP 6500, Perkin Elmer/Thermo Fisher Scientific, Inc.). In addition, the crystalline phases of the samples were identified using X-ray diffraction (XRD: Smartlab, Rigaku Co.). The microstructure and elemental distribution were analyzed qualitatively using field-emission scanning electron microscopy (FE-SEM: Merlin Compact, Carl Zeiss Co. Ltd.) with energy-dispersive X-ray spectroscopy (EDS: AZTEC, Oxford Instruments Pte. Ltd.) to identify residual impurities visually.
3. Results and discussion
3.1. Influence of leaching time on leaching behavior of Ti mixture
In the Ti mixture obtained after deoxidation, MgO was produced because the O in the TiH2 feedstock reacted with Mg metal, as shown in Eq. (1). After deoxidation, the produced MgO was removed from the Ti mixture during HCl leaching to obtain the Ti product.
The concentration of O in the final Ti product after deoxidation and leaching is affected by the following three factors: the O remaining in the Ti mixture obtained after the deoxidation of the TiH2 feedstock, O contamination by the solution during HCl leaching, and the MgO remaining in the Ti product after HCl leaching. Among these three factors, the concentration of O remaining in the obtained Ti mixture is determined solely by the deoxidation conditions. In other words, its value is determined when deoxidation is finished and is not affected by leaching. Thus, to mitigate the increase in the O concentration in the final Ti product, the MgO produced should be removed as much as possible during leaching. Additionally, O contamination by the solution during leaching should be suppressed as much as possible.
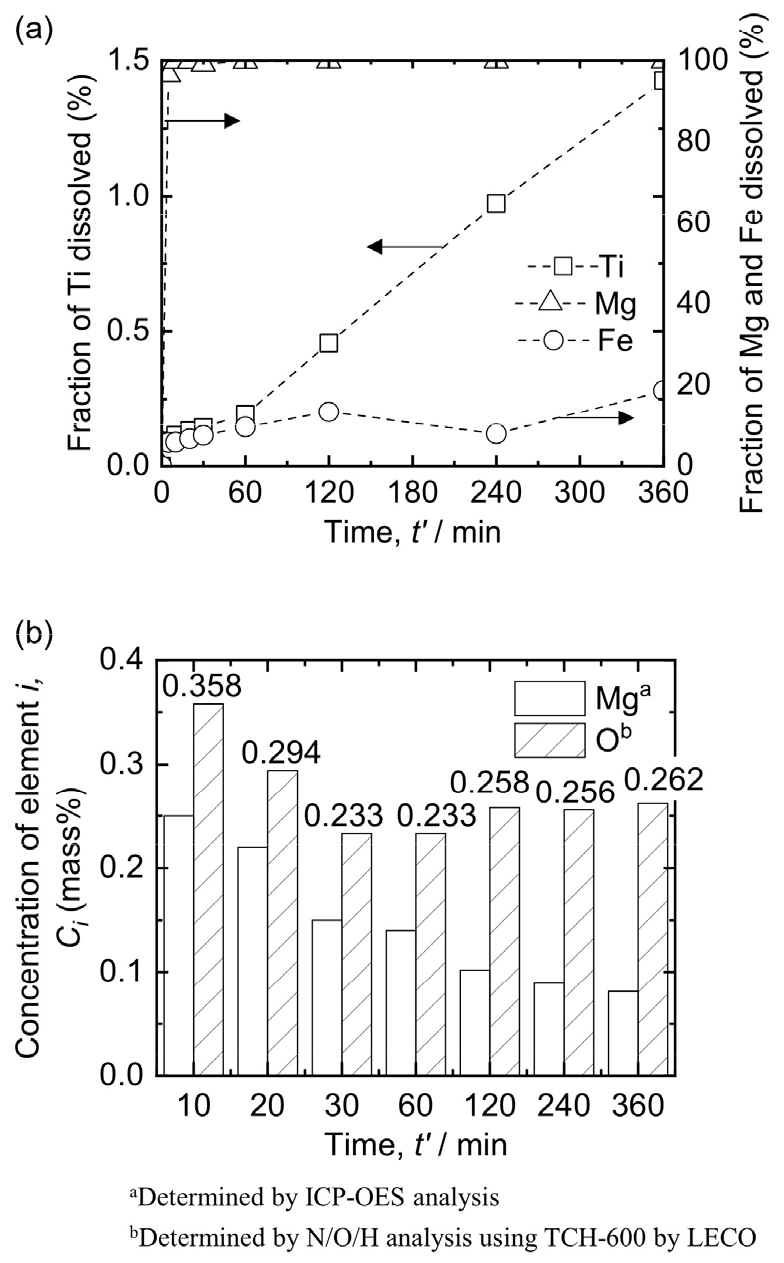
When Ti metal powders are exposed to HCl solution for a long time, O contamination occurs. This indicates the importance of determining the appropriate leaching time for removing impurities from the Ti mixture obtained after deoxidation. Fig. 4(a) shows the influence of leaching time on the dissolution of Ti, Mg, and Fe in the Ti mixture when leaching was conducted using 3 mol/L HCl solution at 293 K with 1 % pulp density (w/v) under Ar gas bubbling. The fraction of each dissolved element was calculated using Eq. (2)55).
In Eq. (2), Ciin solution represents the concentration of element i in the solution sampled at preset times, Ciin final solution represents the concentration of element i in the solution sampled after leaching is finished, and Ciin residues represents the concentration of element i in the Ti product after leaching.
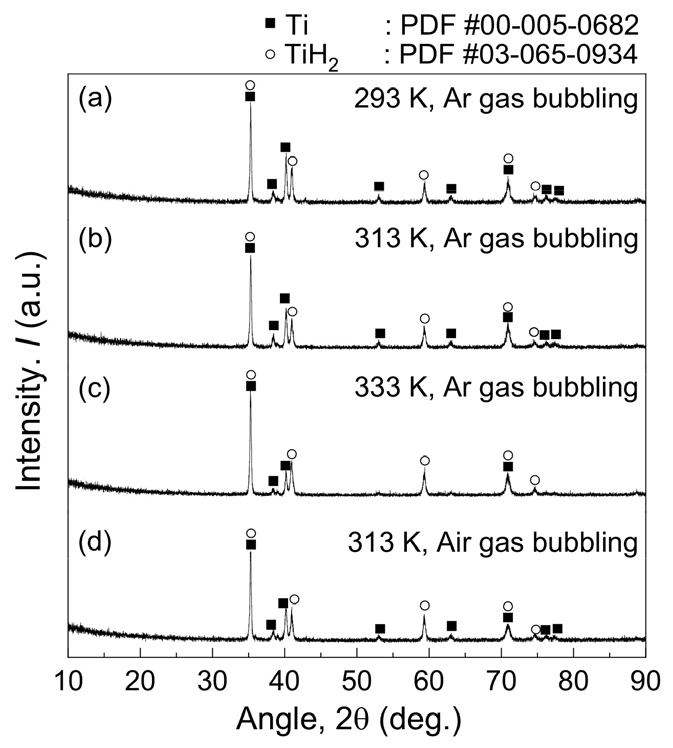
As shown in Fig. 4(a), the fractions of dissolved Ti and Fe increased gradually with the leaching time and were 1.43 % and 18.73 %, respectively, after HCl leaching was conducted for 360 min. However, the fraction of dissolved Mg increased significantly and reached 99.83 % after leaching was performed for only 10 min. This is because most of the Mg compounds in the Ti mixture obtained after deoxidation were MgCl2 and residual Mg metal, which dissolved easily in the 3 mol/L HCl solution. As a result, the leaching efficiency of Mg reached 99.83 % after leaching was performed for 10 min. In addition, after leaching was performed for 360 min, the leaching efficiency of Fe reached 18.73 % even though the leaching efficiency of Ti was limited to 1.43 %. As shown in Fig. 5(a), the Ti product obtained after leaching comprised Ti and TiH2. Additionally, Fig. 2(b) shows a photograph of the Ti product obtained after leaching.
Fig. 6(a) shows the SEM-EDS results of the Ti product obtained after HCl leaching was performed for 20 min. As shown in Fig. 6(a), the bright microstructure is identified as Ti. However, the result shown in Fig. 5(a) suggests that the bright microstructure comprises Ti and TiH2. H was not analyzed owing to the limitations of EDS. Notably, high concentrations of Mg and O were identified at dark points on the surface of the microstructure. In addition, based on a comparison of the microstructures of the Ti products obtained after 20 min and 360 min of HCl leaching, as shown in Fig. 6(b) and (c), the number of dark points decreased as the leaching time increased. This indicates that Mg compounds in the form of oxides remained in the Ti product when HCl leaching was conducted for an insufficient time, thus affecting the O concentration in the Ti product.
Fig. 4(b) shows the changes in the concentrations of Mg and O in the Ti product obtained after HCl leaching was conducted for 10 – 360 min. The concentration of Mg was less than 0.25 mass% for all samples, which satisfies the ASTM B299/B299M-18 standard for commercial pure Ti56), i.e., below 0.5 mass% of Mg. Interestingly, the concentration of Mg decreased continuously until 360 min, whereas the concentration of O decreased until 30 min and then increased after 60 min. During HCl leaching, the O concentration in the Ti product was influenced by the remaining Mg compounds in oxide form and the contamination by O from the solution. As leaching progressed, the remaining Mg compounds in oxide form decreased, thus causing a decrease in the O concentration in the Ti product. Conversely, contamination by O in the HCl solution increased with the leaching duration, thus increasing the O concentration in the Ti product. Because Ti is easily oxidized, the oxide layer consisting of Ti oxides on the surface of the Ti product gradually increases as the leaching time increases, further contributing to the increase in O concentration. Considering these two factors, the increase in the O concentration in the Ti product after 60 min indicates that the influence of contamination by O from the solution during leaching on the O concentration in the Ti product was greater than the influence of the amount of Mg compounds removed. These results suggest that conducting HCl leaching for 60 min is preferred.
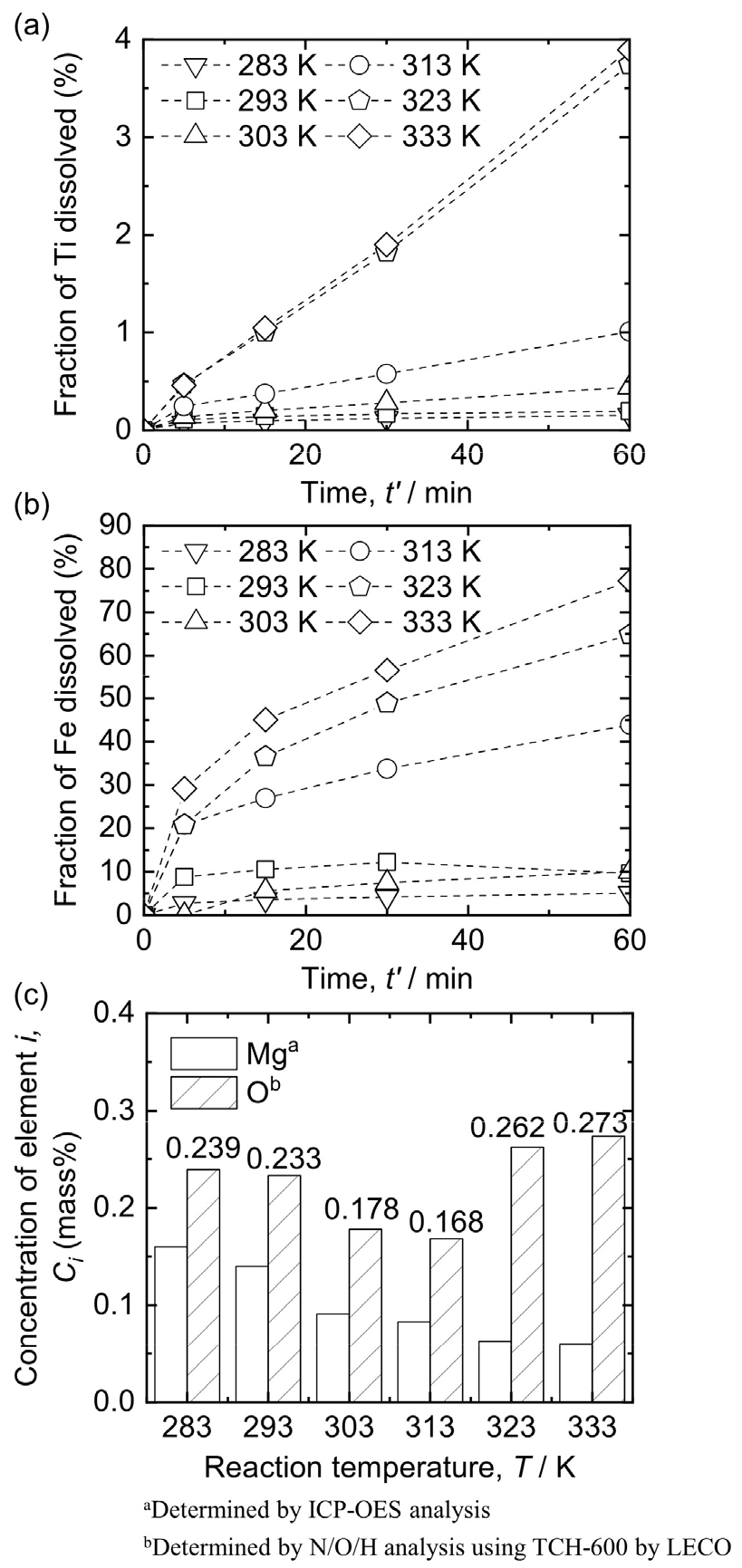
3.2. Influence of temperature on leaching behavior of Ti mixture
The leaching efficiencies of the impurities increased with the leaching temperature. However, the effect of the leaching temperature on the concentration of O in the Ti product and the loss of Ti by dissolution should be considered.
Fig. 7(a) and (b) show the influence of the leaching temperature on the dissolution of Ti and Fe in the Ti mixture when leaching was conducted using 3 mol/L HCl solution at 283 – 333 K with 1 % pulp density (w/v) under Ar gas bubbling. The fraction of dissolved Fe increased from 5.07 % to 77.27 % as the temperature increased from 283 K to 333 K when leaching was conducted for 60 min. However, the fraction of dissolved Ti was below 1.01 % until the leaching temperature reached 313 K and was 3.90 % at 333 K when leaching was conducted for 60 min. Fe readily dissolves in acidic chloride solutions, and its dissolution kinetics are enhanced by increasing temperature. In contrast, Ti surfaces are protected by a stable passive film composed of Ti oxides, which are resistant to HCl. Consequently, Ti dissolution remains limited, leading to only a slight increase in Ti loss compared to the fraction of dissolved Fe.
The Ti products obtained after HCl leaching at 313 K and 333 K were mixtures of TiH2 and Ti, as shown in Fig. 5(b) and (c), respectively. Some researchers reported the dissolution of a low fraction of Ti during HCl leaching of TiH2. Sarkar reported that 1.49 – 2.56 % of Ti was dissolved when 1 mol/L HCl was used for leaching of a mixture comprising residual Mg, MgO, MgCl2, and TiH2 particles with an average diameter of 44.3 µm at 303 – 333 K for 1 h52). In addition, Cho reported a Ti loss of less than 0.50 % and a leaching efficiency of Fe of larger than 55.01 % when 0.1 mol/L HCl solution was used for leaching of a mixture comprising TiH2, ferric oxide (Fe2O3), and MgO at 363 – 463 K for 4 h57).
Fig. 7(c) shows the concentration change of Mg and O in the Ti product obtained after HCl leaching was performed for 60 min at 283 – 333 K. When the leaching temperature increased, the concentration of Mg in the Ti product decreased because the leaching efficiency of the Mg compounds increased. The concentration of O in the Ti product decreased from 0.239 mass% to 0.168 mass% when the leaching temperature increased from 283 K to 313 K. However, the concentration of O in the Ti product increased from 0.168 mass% to 0.262 – 0.273 mass% when the leaching temperature increased from 313 K to 323 – 333 K. These results indicate that, during leaching at 323 – 333 K, the influence of Ti product contamination by O from the solution on the O concentration in the Ti product was greater than the influence of the amount of Mg compounds dissolution. As a result, leaching is preferable to be conducted at 313 K when considering the leaching efficiencies of Ti and Fe and the O concentration in the Ti product.
In previous studies, the leaching of feedstock in Ti form was strictly controlled at room temperature to decrease the contamination of Ti by O from the leaching solution19). However, in this study, the optimal leaching temperature for a mixture comprising Ti and TiH2 was 313 K when leaching was conducted for 60 min using 3 mol/L HCl solution with 1 % pulp density (w/v) under Ar gas bubbling. TiH2 has been reported to possess higher resistance to air as compared with Ti because it is a ceramic and not a metal. Zhang showed that when Ti and TiH2 powders were exposed to air at room temperature, the O concentration in TiH2 powder increased more slowly than that in Ti powder58). These results suggest that the contamination of TiH2 by O was hindered during leaching, even when the leaching temperature was 313 K, as in the experiments.
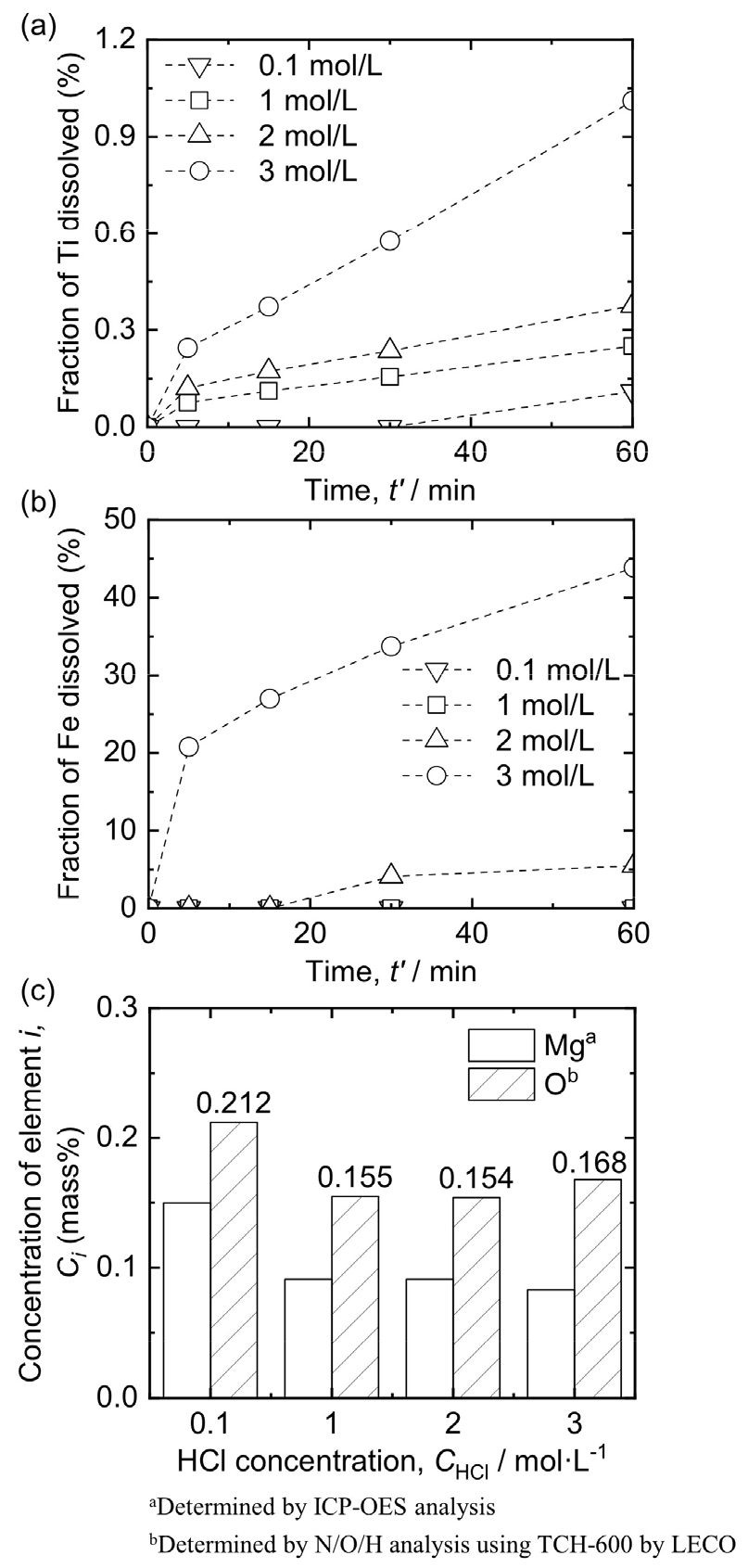
3.3. Influence of HCl concentration on leaching behavior of Ti mixture
Fig. 8(a) and (b) show the influence of the concentration of HCl solution on the dissolution of Ti and Fe when leaching was performed at 313 K using 0.1 – 3 mol/L HCl solution with 1 % pulp density (w/v) under Ar gas bubbling. When 0.1 mol/L HCl solution was used, the fractions of dissolved Ti and Fe were 0.11 % and negligible, respectively. In addition, when the concentration of the HCl solution was increased to 3 mol/L, the fractions of dissolved Ti and Fe were 1.01 % and 43.84 %, respectively. The influence of HCl concentration on the fraction of dissolved Fe is explained by the acidity of the HCl solution. When the concentration of the HCl solution is increased, the larger hydrogen ions (H+) contribute to an increase in the fraction of dissolved Fe.
As shown in Fig. 8(c), when the concentration of the HCl solution was increased from 0.1 mol/L to 1 – 3 mol/L, the concentration of Mg decreased from 0.150 mass% to 0.091 – 0.083 mass%. Notably, the O concentration in the Ti product was 0.154 – 0.168 mass%, showing no significant difference even when the concentration of the HCl solution varied from 1 mol/L to 3 mol/L. By contrast, the O concentration in the Ti product was 0.212 mass% when 0.1 mol/L HCl solution was used.
In addition to Fe dissolution, H+ ions were used to dissolve MgO and Mg, as shown in Eqs. (3) and (4)59), respectively. The dissolution of MgO and Mg compounds requires sufficient H+ ions. When the HCl concentration is 0.1 mol/L, the insufficient supply of H+ ions limits the dissolution rate, which is expected to leave a certain amount of Mg compounds in the Ti product and result in a higher O concentration. In addition, the O concentrations in the Ti products, ranging from 0.154 mass% to 0.168 mass%, suggest that no significant O contamination occurred when the concentration of the HCl solution was 1 – 3 mol/L. Therefore, 3 mol/L HCl solution is suitable when considering the O concentration and the removal of Fe and Mg impurities from the Ti product.
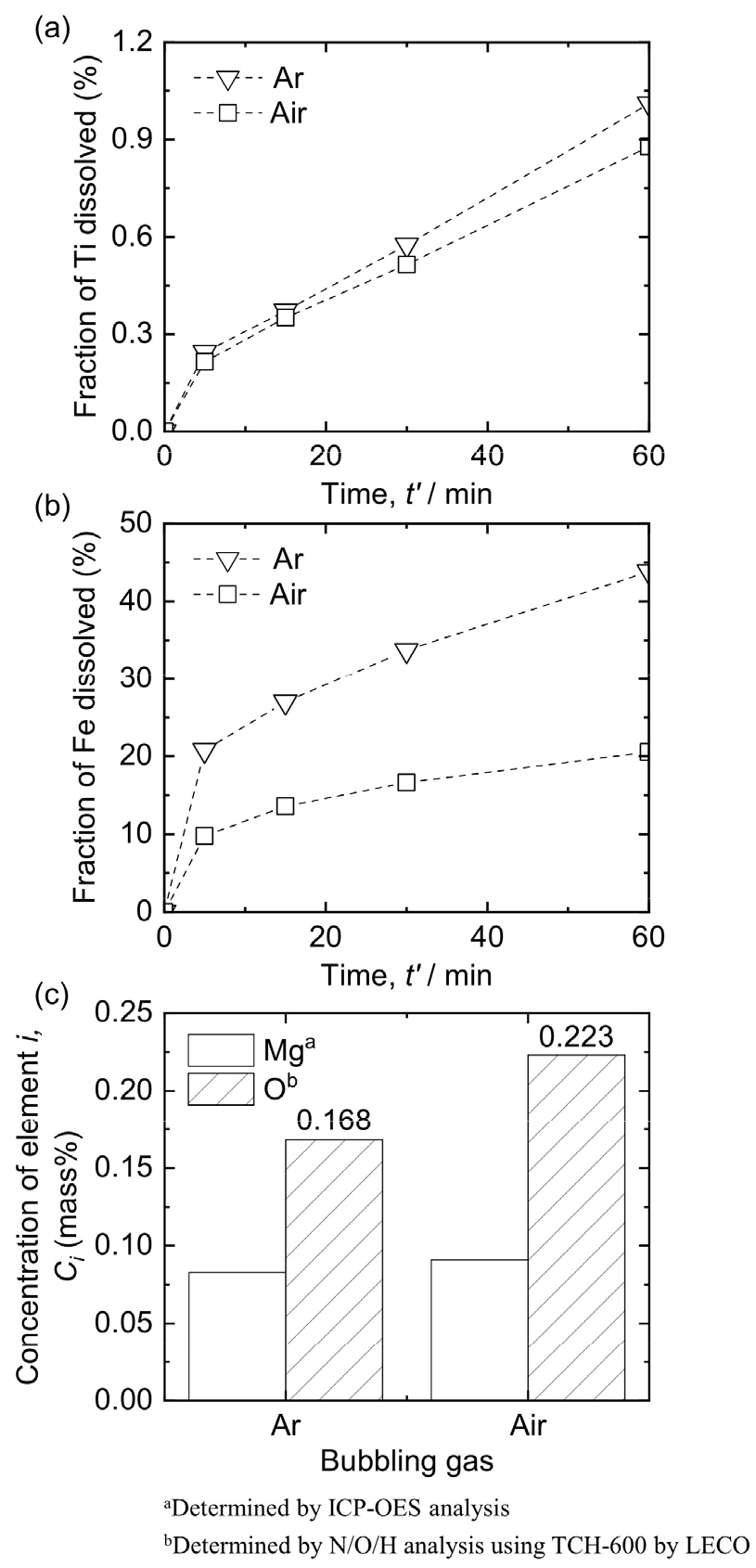
3.4. Influence of gas for bubbling on leaching behavior of Ti mixture
Fig. 9(a) and (b) show the influence of the type of gas used for bubbling on the dissolution of Ti and Fe when leaching was conducted at 313 K using 3 mol/L HCl solution with 1 % pulp density (w/v). As shown in Fig. 9(a), the fractions of dissolved Ti after leaching was conducted for 60 min were 0.88 % and 1.01 % under Ar gas and air bubbling, respectively. This indicates that the type of gas used for bubbling did not significantly influence the fraction of dissolved Ti. In addition, Fig. 5(d) shows that a mixture comprising TiH2 and Ti was obtained after HCl leaching was performed at 313 K using a 3 mol/L HCl solution with 1 % pulp density (w/v) under air bubbling.
However, the fraction of Fe dissolved under Ar gas bubbling was 43.84 %, which was larger than that of Fe dissolved under air bubbling, i.e., 20.55 %. It has been reported that the dissolution behavior of Fe oxide in oxalic acid solutions is accelerated with increasing ferrous ions (Fe2+) concentration in the solution60,61). In addition, the importance of removing dissolved oxygen from the solution via inert gas bubbling has been emphasized, since Fe2+ ions are readily oxidized by dissolved oxygen60,61). Accordingly, the fraction of Fe dissolved under air bubbling is expected to be lower than that under Ar gas bubbling. However, the detailed mechanism is still under investigation.
As shown in Fig. 9(c), the concentration of O in the Ti product increased from 0.168 mass% to 0.223 mass% as the gas type was changed from Ar to air. When HCl leaching was conducted under Ar gas bubbling, the dissolved oxygen in the solution was depleted, thereby delaying the formation of a passive film on the Ti product during HCl leaching62). However, when air was used for bubbling, O2 from the air and dissolved oxygen in the solution facilitated the formation of a passive film on Ti. The formation of the passive film indicates the oxidation of the Ti product and an increase in its O concentration. Therefore, Ar gas bubbling is preferred over air bubbling to mitigate the increase in the O concentration in the Ti product during HCl leaching.
Overall, the optimized leaching conditions at 313 K with 3 mol/L HCl, 1 % pulp density (w/v), and Ar bubbling resulted in a Ti product that satisfied the O concentration below 0.18 mass%, ASTM Gr. 1 for Ti, while maintaining low Ti loss of 1.01 %, high Mg removal above 99.9 %, and significant Fe removal of 43.84 %. Compared with the previous study52), where Mg removal reached 99.96 % with a Ti loss of 1.8 %, the present study achieved an equally high Mg removal but with lower Ti loss. These results highlight that effective impurity removal and Ti recovery were achieved by the optimized leaching process developed in this study.
4. Conclusions
During the HCl leaching of a Ti mixture obtained after the deoxidation of an off-grade Ti sponge using Mg in a H2 mixed gas atmosphere, the O concentration in the Ti product is important. Additionally, low Ti loss and high dissolution efficiencies of Fe and Mg are essential during leaching. Therefore, the effects of leaching time, temperature, molarity of HCl solution, and gas type used for bubbling on the leaching behavior of a Ti mixture were evaluated in this study.
(1) When leaching was performed at 293 K using 3 mol/L HCl solution with 1 % pulp density (w/v) under Ar gas bubbling for 5 – 360 min, the O concentration in the Ti product decreased from 0.358 mass% to 0.233 mass% until 60 min of leaching time, owing to Mg removal. However, after 360 min, the O concentration increased to 0.262 mass% owing to O contamination by the solution.
(2) When leaching was conducted at 283 – 333 K using 3 mol/L HCl solution with 1 % pulp density (w/v) under Ar gas bubbling, the O concentration in the Ti product decreased from 0.239 mass% to 0.168 mass% as the temperature increased from 293 K to 313 K, owing to Mg removal. At 333 K, however, the O concentration increased to 0.273 mass% owing to O contamination by the solution.
(3) The O concentration in the Ti product was 0.155 – 0.166 mass% when 1 – 3 mol/L HCl solution was used for leaching at 313 K with 1 % pulp density (w/v) under Ar gas bubbling. However, the O concentration in the Ti product was 0.212 mass% when 0.1 mol/L HCl solution was used.
(4) When HCl leaching was conducted at 313 K using 3 mol/L HCl solution with 1 % pulp density (w/v) under Ar gas bubbling, the removal of Mg was larger than 99 % within 60 min. Under these optimized conditions, the fractions of dissolved Fe and Ti were 43.84 % and 1.01 %, respectively, demonstrating that low Ti loss was achieved while effectively removing impurities.