1. Introduction
2. Mechanochemistry
2.1. Mechanochemical process
2.2. Mechanochemistry in LIBs recycling
3. Mechanochemical process in LIBs recycling
3.1. LiFePO4
3.2. LiCoO2
4. Summary
1. Introduction
Lithium (Li) is an essential element in most battery applications and cannot be easily substituted for other elements1,2,3). The composition of cathode electrodes can be varied in the forms of Li-transition metals compounds, including lithium iron phosphate (LFP, LiFePO4), lithium cobalt oxide (LCO, LiCoO2), lithium manganese oxide (LMO, LiMn2O4), and lithium nickel/cobalt/manganese oxide (NCM, LiNixCoyMn(1-x-y)O2)4). The lithium-ion batteries (LIBs) technologies have different chemistries, electrochemical performances, and applications4).
The skyrocketed LIBs demand in recent years is driven primarily by the electric-vehicle (EV) revolution to achieve decarbonization in the transportation sector and improve the air quality in urban areas1,4). According to the data reported by International Energy Agency (IEA), the total Li demand is projected to increase from 80 kiloton (kt) in 2021 to 330–500 kt in 2030, and EV batteries account for 70 %–80 % of the total Li demand in 2030 depending on the scenarios5). At the same time, this promotion of EVs generates a massive number of spent LIBs containing valuable metals, including cobalt (Co), nickel (Ni), manganese (Mn), iron (Fe), or Li, even with a higher grade than the mined ores6). Recycling for moderating the soaring mineral demand is estimated to have a minor impact until 2030, but this would contribute significantly after 20305).
Many countries established waste LIBs collection and announced the strategic recycling policy to resolve the environmental concerns caused by landfilling and depletion of critical minerals6,7,8). Spent LIBs recycling through the conventional process significantly reduce the environmental impact compared to disposal without proper treatment8). However, the pyrometallurgical process has disadvantages, such as high energy consumption and gas emission, as well as the hydrometallurgical process, such as large consumption of strong acid/base and wastewater generation9). Also, the existing spent LIBs recycling has been frequently focused on extracting Co and Ni, but the development of Li recovery processes from many types of LIBs is the recent interest due to the high Li price10). Newly reported recycling processes are designed to recover Li before Co and Ni11), which seems to be due to the high loss of Li that occurred by many routes in the conventional process10,12). Therefore, developing a sustainable and high Li recovery recycling process from spent LIBs is important, and research on the mechanochemical processes to overcome the above challenge is addressed in this paper.
2. Mechanochemistry
2.1. Mechanochemical process
Mechanochemistry refers to inducing physicochemical changes in solid materials by using mechanical forces such as compression, shear, and impact12,13,14). In addition to the size reduction and specific surface area increase, which are commonly observed in the comminution process, the accumulation of mechanical energy in solids should result in irreversible changes in solid materials by transforming the structural arrangements with the defects generation and dislocations during the mechanochemical process15). This energy transfer frequently occurs under high-energy milling conditions with the application of special mills, including planetary ball mill (PBM), vibration mill, and disk mill13). The mechanochemical process can be distinguished depending on the grinding result between mechanical activation, usually an increase in reactivity while remaining unchanged, and mechanochemical activation, when changes occur in the chemical composition of the product13,15).
The mechanochemical process can be connected or applied to properly further mineral and/or metallurgical processing steps by considering the chemical and physicochemical changes in solids material13). The mechanochemical processing generally applied in extractive metallurgy is either (1) grinding (dry or wet grinding) and leaching in a separate process or (2) simultaneously grinding and leaching (wet grinding), which is the so-called mechanochemical leaching process13,16). In general, dry grinding is a solvent-free process that introduces the solid state feed only or together with the solidified reagent to induce the direct solid-solid reaction. As a result, the process is less generative of wastewater and avoids generating undesirable products12,13). In addition, the poorly soluble or insoluble reagents can also be utilized in the solid-solid reaction system14). On the other hand, wet grinding is expected to enhance the grinding efficiency and homogenous result by reducing the dead angle in the grinding chamber and mixing the feed uniformly17,18,19). In addition, it can exploit highly excited state materials for the chemical reaction, including new phase formation or leaching, before the relaxation of damaged crystalline structures13).
2.2. Mechanochemistry in LIBs recycling
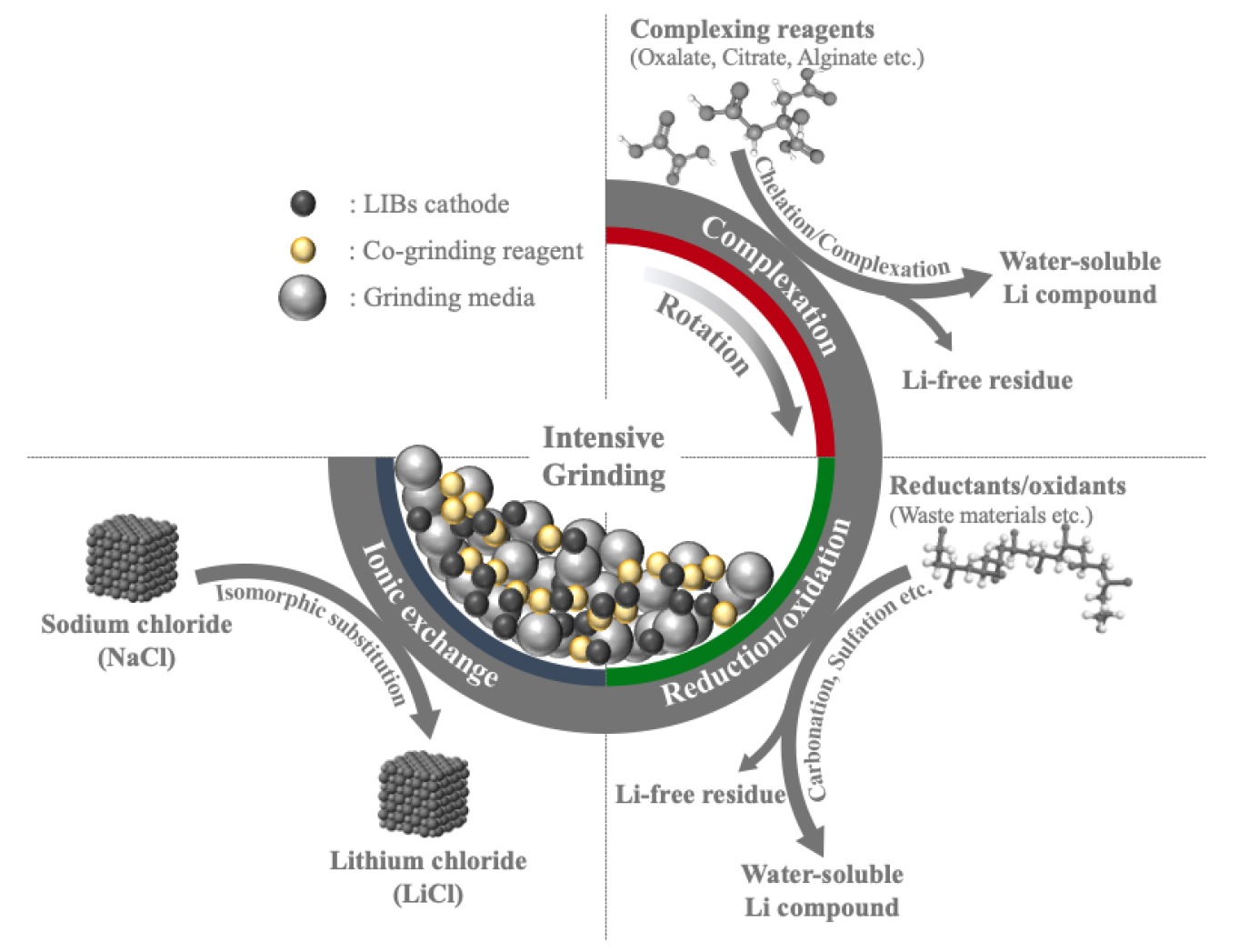
In recent spent LIBs recycling, innovative processes, either mechanochemical process only or combination with the conventional process, are proposed. In particular, the hydrometallurgical process is the most common approach to apply the mechanochemistry15) by grinding electrode materials together with the various types of reagents for redox reaction20,21,22,23,24), complexation17,18,19,25,26,27), or ionic exchange20) (Fig. 1). Then, the in-situ generated water-soluble salts can be simply separated by washing them with water. Otherwise, grinding electrode materials can be mechanically activated only and leached out the valuable metals in the following extraction process using low-concentrated leachate and milder conditions28,29).
The other key point in the mechanochemical process in proposed LIBs recycling methods is a sustainable process that does not introduce hazardous solvents, so this process is relatively safe and greener12). The mechanochemical process does not require much water (or not at all) to achieve the desired reaction, and it will reduce the wastewater stream and simplify the process flow15,16,24). Various environmentally-friendly reagents are generally employed for effective metal extraction, and furthermore, some processes include the regeneration step to minimize the reagent consumption20,22,24).
In the following section 3, the mechanochemical or mechanical activation processes are highlighted by the main reaction mechanisms for Li recovery from spent LIBs, focused on LiFePO4 and LiCoO2, in which Li has a significant economic value. The optimal experiment conditions and leach efficiencies of valuable metals from LiFePO4 and LiCoO are summarized in Table 1 and 2, respectively.
Table 1.
Mechanochemical process for metal extraction from spent LiFePO4 cathode
|
Mechano- chemical Reaction |
Conditions of Mechanochemical Process |
Conditions of Leaching Process |
Leach Efficiency |
Final Productd) | Ref. | |||||||||
|
Co-grinding reagent |
Grinding method |
Mixture mass ratio (Pa) : Rb)) |
BTR (Bc) : Pa)) |
Grinding speed (rpm) |
Time (min) |
Leach solution |
Temp (°C) |
Leach time (min) |
Li (%) |
Fe (%) | Li | Fe | ||
| Oxidation | Ce) | Dry | - | N/A | 500 | 360 | H2O | N/A | N/A | 27.0 | - | Li2CO3 | - | [20] |
| Ce) + NaClO | Dry | 1:2f) | N/A | 600 | 5 | H2O | N/A | N/A | 93.9 | 0.06 | Li2CO3 | FePO4 | [21] | |
| Na2S2O8 | Dry | 1:2 | N/A | 600 | 5 | H2O | N/A | N/A | 99.7 | 19.7 | Li3PO4 | FePO4 | [22] | |
| Chelation | H2Citg) |
Wet (H2O2) | 1:20 | 25:1 | 300 | 120 | H2O | N/A | 0h) | 99.35 | 3.86 | Li2CO3 | FePO4 | [18] |
| Na3Citi) |
Wet (H2O2) | 1:10 | 20:1 | 500 | 300 | H2O | N/A | N/A | 98.5 | 5.1 | Li2CO3 | FePO4 | [19] | |
| H2Oxj) |
Wet (H2O) | 1:1 | 20:1 | 500 | 120 | H2O | RTk) | 30 | > 99 | > 94 | Li3PO4 |
FeOx× 2H2Ol) | [17] | |
| Na2EDTA | Dry | 3:1 | 60:1 | 550 | 120 |
0.6M H3PO4 | RTk) | 20 | 97.67 | 94.29 | Li3PO4 |
FePO4× 2H2O | [25] | |
|
Isomorphic substitution | NaCl | Dry | 1:2 | N/A | 500 | 360 | H2O | N/A | N/A | 96.03 | - | Li2CO3 | NaFePO4 | [20] |
Table 2.
Mechanochemical process for metal extraction from spent LiCoO2 cathode
|
Mechano- chemical Reaction |
Conditions of Mechanochemical Process |
Conditions of Leaching Process |
Leach Efficiency |
Final Productd) | Ref. | |||||||||
|
Co-grinding Reagent |
Grinding method |
Mixture mass ratio (Pa) : Rb)) |
BTR (Bc) : Pa)) |
Grinding speed (rpm) |
Time (min) |
Leach solution |
Temp (°C) |
Leach time (min) |
Li (%) |
Co (%) | Li | Co | ||
|
Reduction- Oxidation | PVC | Dry | N/A | N/A | N/A | 1,800 | H2O | RTe) | 60 | > 99 | > 90 | LiCl | CoCl2 | [23] |
| PVC + Fe | Dry | 1:1:2g) | 50:1 | 600 | 720 | H2O | N/A | N/A | > 99 | 8.1 | LiCl | CoFe4O6 | [24] | |
| Ch) | Dry | - | N/A | 500 | 30 |
20 vol % HAci) + 5 vol % H2O2 | RTe) | 15 | 99.8 | 99.7 | Li2CO3 | Co(OH)2 | [28] | |
| SiO2j) | Dry | - | 1:1k) | 500 | 30 | H2Citl) | 80 | 30 | 97.22 | 94.91 | - | - | [29] | |
| CO2(s) | Dry | 1:20 | N/A | 700 | 90 | H2O | RTe) | N/A | 95.04 | N/A | Li2CO3 | Co3O4 | [34] | |
| Chelation | EDTA | Dry | 1:4 | 80:1 | 600 | 240 | H2O | N/A | 30 | 99 | 98 | Li2CO3 | Co3O4 | [26] |
| Na2EDTA | Dry | 1:4 | 80:1 | 600 | 240 | H2O | N/A | 30 | 74 | 71 | Li2CO3 | Co3O4 | [26] | |
|
Complexa- tion | HAlgm) |
Wet (H2O2) | 1:10 | N/A | 500 | 240 | H2O | RTe) | 5 | 97.6 | 98.6 | - | - | [27] |
3. Mechanochemical process in LIBs recycling
3.1. LiFePO4
3.1.1. Oxidation process
The carbon (C) in spent cathode materials is mostly organic binder or graphite (anode), and it is utilized as a reducing agent while thermal processing of cathode materials or burned off at high temperatures to detach cathode materials from other battery components effectively30,31). In contrast, in the hydrometallurgical process in which a trace amount of C is remaining, the agglomeration of particles could be occurred in the aqueous solution because of its hydrophobic characteristics and hinder the efficient metal dissolution and results in extra acid consumption29,32,33). During the mechanochemical process, however, many researchers found that the organic materials involved in the reactions either the formation of water-soluble Li compound20,21) or reduction of the high-valency metals ion23,28,29,34). In the mechanochemical process of LiFePO4, the formation of lithium carbonate (Li2CO3) was reported by Liu et al.20,21).
Liu et al.20) reported that 27 % of Li dissolved in the water from mechanochemically activated LiFePO4 cathodes without co-grinding reagents under the following conditions: 500 rpm of rotary speed and 6 hours of grinding time in PBM. They observed the changes in binding energy before and after mechanochemical reaction using X-ray photoelectron spectroscopy (XPS). From XPS results of C 1s, they confirmed the peaks of the binding energy of CO32- (289.3 eV) on the surfaces of LiFePO4 as well as change in the peaks of the binding energy of Li 1s and Li2CO3 (55.03 eV). From the result, they explained the changes in chemical characteristics with solid-solid reaction (between LiFePO4 and carbon) under the intensive mechanical stress condition reduced some of Li in LiFePO4 structure, and the carbon absorbed carbon dioxide (CO2) in the product and converted Li into Li2CO3. They noted that although the trace carbon enables the Li extraction through mechanochemical reaction, using the co-grinding reagents is more desirable to extract Li efficiently rather than exploiting the trace carbon only.
In the study by Liu et al.21), the intervention of C in the mechanochemical oxidation process of LiFePO4 with sodium hypochlorite (NaClO) was investigated. They also conducted direct leaching of LiFePO4/NaClO mixture in deionized water, which showed a different Li pathway by interacting with hydrogen and generating lithium chloride (LiCl), but the only mechanochemical process part is addressed below. During the mechanochemical process, they changed grinding variables, including the LiFePO4/NaClO molar ratios (2:1–5), grinding speeds (0–800 rpm), and grinding times (2.5–10 minutes), then leached mechanochemically activated samples in deionized water without agitation. They proposed the possible mechanisms of oxidation of Li from LiFePO4 and C by NaClO to generate Li2CO3, as follows:
From the X-ray diffraction (XRD) analysis, they found that the peaks of LiFePO4 disappeared from the LiFePO4/NaClO molar ratio of 1:1, which indicates the NaClO oxidized LiFePO4 to FePO4 through the mechanochemical reaction. In addition, they observed clear peaks of Li2CO3, FePO4, and NaCl from the LiFePO4/NaClO molar ratio of 1:2, and the Li leach efficiency reached a plateau. To support the proposed mechanism, they observed the oxidation of carbon and its involvement in forming Li2CO3 through XPS analysis. From XPS results of C 1s, the peaks of the binding energy of C-O species shifted from 285.6 eV to 286.4 eV, implying the oxidation, and CO32- (289.8 eV) gradually appeared as the grinding speed increased. However, when the grinding speed increased from 600 to 800 rpm, NaClO directly decomposed before fully interacting with LiFePO4, and Li leach efficiency decreased. They could selectively leach Li with 93.9 % of the highest Li leach efficiency under optimal conditions of a LiFePO4/NaClO mass ratio of 1:2 and grinding speed at 600 rpm for 5 minutes while maintaining 3.5 % of P and 0.06 % of Fe minimal leach efficiency.
On the other hand, Liu et al.22) introduced sodium persulfate (Na2S2O8), an oxidant, as a co-grinding reagent in the mechanochemical process followed by water leaching and precipitation-filtration process to recover Li as lithium phosphate (Li3PO4). They targeted further development of the oxidation leaching process of spent LiFePO4 using Na2S2O8 conducted by Zhang et al.35), which should maintain the solution pH between 3 and 4 with the addition of acid for high metal recovery. Liu et al.22) assumed the solid phase oxidation mechanisms during the mechanochemical process of LiFePO4/Na2S2O8 as follows: (1) Na2S2O8 forms the oxidation environment in solid-solid interfacial reaction and (2) mechanical force acts as the driving force of oxidation system similar to electrical energy. The combination of these mechanisms allows Li to be released from the FePO4 structure, and results in the formation of a new compound, LiNaSO4, with the decomposition of Na2S2O8 as described in Eq. (4).
Liu et al.22) studied the Li extraction trend with different grinding speeds (0–600 rpm), grinding times (5–40 minutes), and mass ratios of LiFePO4/Na2S2O8 (1:0.5–4) while maintaining a ball-to-powder ratio (BTR). LiFePO4 reacted with Na2S2O8 rapidly during the mechanochemical process, and they could achieve 99.7 % of Li and 19.7 % of Fe leaching efficiency under optimal conditions of mass ratio of LiFePO4/Na2S2O8 of 1:2 and grinding at 600 rpm for 5 minutes in PBM. Prolonged grinding time rather deteriorated the leaching efficiency of Li due to the partial decomposition of the product of the mechanochemical reaction. They noted that the mechanochemical process provides a promising approach for achieving an environmentally-friendly process, such as acid/alkali-free and wastewater discharge-free, while maintaining high extraction efficiency. Furthermore, the price of Na2S2O8 is relatively high and is unfavorable for scaled-up process plant35,36), but advantages of the proposed process, including regeneration of Na2S2O8 and less pH adjustment for maximal Li recovery, could balance the operational cost36).
3.1.2. Chelation process
3.1.2.1. Citrate chelation
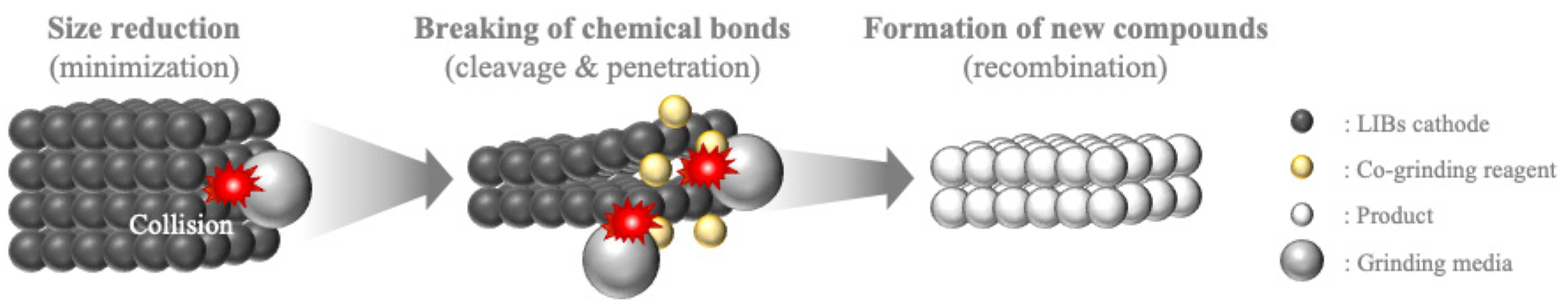
Li et al.18) and Zhang et al.19) studied the mechanochemical reaction of LiFePO4 with solidified citric acid (H3Cit) and sodium citrate (Na3Cit), respectively, but equally extracting Li by chelating Li+ with organic ligands (Cit3-). Both research groups proposed a similar extraction mechanism with three stages which are particle size reduction and specific surface area increase (minimization), penetration of grinding additives into the LiFePO4 structure and breaking of its chemical bond (cleavage), and formation of new compounds (recombination) (Fig. 2). Besides, those research groups ground mixture under the wet grinding condition using a small amount of liquid, either hydrogen peroxide (H2O2) or water.
Li et al.18) investigated mechanochemical reaction by varying the grinding factors, including the LiFePO4/H3Cit mass ratios (1:10–80), grinding speeds (100–500 rpm), grinding times (0.5–4 hours), BTRs (15–55:1), and volumes of H2O2 (0–2 mL), and washed out the mixture using deionized water after grinding. In the case of the grinding without H2O2, they added 2 mL of water in the grinding chamber, and they conducted water leaching for 0–8 hours after grinding for 2–10 hours while maintaining the other grinding variables. In this study, the liquid phase resulted in major differences in this study, as described in Eq. (5) (without H2O2) and Eq. (6) (with H2O2). The wet grinding condition without H2O2 loses Li selectivity, extracting Li and Fe more than 90 % under all experimental conditions. Furthermore, phosphoric acid (H3PO4) is produced (Eq. (5)) by mechanochemical reaction, and H3PO4 precipitates Li in the form of lithium phosphate (Li3PO4), which is a water-insoluble compound, in the subsequent water leaching. On the other hand, H2O2 minimizes the leach out of ferrous ions (Fe2+) by oxidizing it to ferric ions (Fe3+), then forming a water-insoluble ferric phosphate (FePO4) (Eq. (6)), which enables to achieve highly selective recovery of Li. They observed clear peaks of the FePO4 and some of the unreacted LiFePO4 in the residue using XRD analysis. They could achieve the selective Li extraction (99.35 %) over Fe (3.86 %) under optimal conditions of a LiFePO4/H3Cit mass ratio of 1:20, a BTR of 25:1, and grinding at 300 rpm for 2 hours in PBM with 1 mL of H2O2.
Recently, Zhang et al.19) used Na3Cit to achieve a cost-effective and acid-free process simultaneously (Eq. (7)). They investigated various grinding factors, including the LiFePO4/Na3Cit mass ratios (1:2.5–12.5), grinding speeds (200–600 rpm), grinding times (1–5 hours), and volumes of H2O2 (0.25–1.25 mL), while maintaining BTR (20:1). In contrast to study by Li et al.18), the maximum Li leach efficiency only reached around 25 % with a similar Fe extraction trend without H2O2 under this acid-free process. From the results, Zhang et al.19) noted the addition of H2O2 majorly affected Li leach efficiency unlikely the study by Li et al.18), deforming LiFePO4 to FePO4 and accelerating Li dissolution, as well as the suppression of leach out of Fe2+. Under the acid-free system, they could achieve 98.9 % of Li leach efficiency with 5.1 % of Fe leach efficiency under optimal conditions of a LiFePO4/Na3Cit mass ratio of 1:10 and grinding at 500 rpm for 5 hours with 1 mL of H2O2.
Both studies commonly selectively extracted Li from the skeleton structure of FePO4 by the oxidation effect of H2O2 assisted by Cit3+ ligand and recovered as a form of Li2CO3. In addition, Zhang et al.19) noted that the leach residue also produces economic benefits besides Li2CO3, since it can be utilized as the cathode material again.
3.1.2.2. Oxalate chelation
Fan et al.17) reported the mechanochemical process of LiFePO4 using solidified oxalic acid (H2Ox), a co-grinding reagent, followed by 30 minutes of water leaching. They recovered Li+ and PO43- in the leach solution as Li3PO4 precipitates by adjusting pH to 8 with NaOH and Fe as ferrous oxalate (FeOx×2H2O), which is poorly insoluble in water, as leach residue (Eq. (8)). They investigated the effect of mechanochemical reaction by varying the grinding factors, including the LiFePO4/H2Ox mass ratios (5:1–1:2), grinding speeds (200–500 rpm), grinding times (0.08–4 hours), and BTRs (10–40:1) with 1 mL of water in PBM.
To support the reaction mechanism and leach result, they utilized XRD analysis, and they could detect the clear peaks of FeOx×2H2O in tested variables ranges of mass ratios of LiFePO4/H2Ox. The peaks of LiFePO4 disappeared from a mass of 1:1 as well as the Fe dissolution decreased by generating FeOx×2H2O, indicating that the mass ratio of LiFePO4/H2Ox significantly influences the chelation efficiency of metal ions. They also observed oxalates with rodlike shapes from the mechanochemically activated samples using scanning electron microscope (SEM), which was not detected with activated samples without oxalic acid.
On the other hand, although the increase of the other grinding factors enhanced the Li leach efficiency and selectivity over Fe, the structure of FeOx×2H2O was decomposed and resulted in Fe dissolution when the grinding time extended more than 2 hours. They could recover approximately 99 % of Li and 94 % of Fe under optimal grinding conditions of a LiFePO4/H2Ox mass ratio of 1:1, a BTR of 20:1, and grinding at 500 rpm for 2 hours with 1 mL of water.
3.1.2.3. EDTA chelation
Yang et al.25) introduced the mechanochemical process of LiFePO4 with ethylenediaminetetraacetic acid disodium (Na2EDTA) in PBM, a well-known chelating reagent, before acid leaching. Compared to the citrate18,19) or oxalate chelation17), however, Li or Fe could not be selectively leached out, so they should be recovered in stepwise precipitation as forms of FePO4×2H2O by oxidizing Fe2+ to Fe3+ by refluxing air (Eqs. (9) and (10)) and Li3PO4 at pH 8 (Eq. (11)). Instead, they targeted to reduce acid consumption, use less strong acid, and prevent second contamination by containing the reagents in the process. As a result, they investigated the effects of grinding or leaching variables, including the LiFePO4/Na2EDTA mass ratios (6–1:1) and grinding times (0.5–6 hours) during the mechanochemical reaction and H3PO4 concentration (0.2–0.7 M), pulp densities (40–100 g/L), and leaching times (0–60 minutes) in the leaching stage, respectively, while maintaining a grinding speed at 550 rpm with a BTR of 60:1.
Overall, the grinding and leaching variables positively affected the leach efficiency and reached a plateau within 2 hours of grinding and 20 minutes of leaching time. In the case of the reagent consumption, they chose a LiFePO4/Na2EDTA mass ratio of 3:1, which is a slightly lower Na2EDTA addition than the theoretical ratio (2.4:1) for full chelation, and an H3PO4 concentration of 0.6 M, when pulp density is kept below 50 g/L, due to the leach efficiency showed minor differences. With the above grinding and leaching conditions, they could achieve 97.67 % of Fe and 94.29 % of Li leach efficiency, which could not achieve with a mechanically activated sample with an equal amount of acid.
3.1.3. Isomorphic substitution process
Liu et al.20) investigated the isomorphic substitution of Li in LiFePO4 with sodium (Na) in sodium chloride (NaCl) during mechanochemical process in PBM (Eq. (12)) followed by water leaching. After the completion of extraction process, they recover Li as Li2CO3 by precipitation using sodium carbonate (Na2CO3) (Eq. (13)) and regenerated NaCl return to the initial mechanochemical process.
They investigated various grinding factors, including the LiFePO4/NaCl mass ratios (1:1–4), grinding speeds (200–600 rpm), and grinding times (2–8 hours). In addition, they conducted theoretical studies on the structural stability when Na replaced Li or Fe in LiFePO4 and electron structure in LiFePO4 to support the reaction mechanism by identifying each compound’s formation energy and structural stability. The theoretical calculation found less volume expansion and significantly low formation energy when Li is replaced by Na and expected to form NaFePO4 rather than LiNaPO4. Besides, the oxygen (O) forms a stable covalent bond with FePO4 while the interaction was significantly weak with Li, and the similarity in structural chemical characteristics between Na and Li (e.g., atomic radius, coordination category, and electron arrangement) facilitated Na to substitute Li. Similarly, they referred to potassium chloride (KCl) as a co-grinding reagent, but the mechanochemical reaction of LiFePO4/KCl showed lower leach efficiency owing to its larger atomic radius than Na. Based on the theoretical study, they believed the induction of mechanical force promoted the local atom rearrangements of the defects in LiFePO4 and NaCl, and the isomorphic substitution could be occurred by solid-phase reaction accordingly because the LiFePO4 is not directly leached out using NaCl solution.
They could achieve 96.03 % of Li leach efficiency under optimal conditions of a LiFePO4/NaCl mass ratio of 1:2 and grinding speed at 500 rpm for 6 hours. However, the detailed explanation on relationships between grinding variables and leach efficiency is not available. On the other hand, they noted that the wet grinding significantly deteriorated the leach efficiency, from 96.03 % to 12.6 % of Li leach efficiency, due to the difficulty in mechanochemical reaction between NaCl dissolved in water and LiFePO4.
3.2. LiCoO2
3.2.1. Reduction-Oxidation process
3.2.1.1. Chlorination
Saeki et al.23) and Wang et al.24) investigated the mechanochemical processing of LiCoO2 and polyvinyl chloride (PVC). Both research groups commonly exploited PVC waste as a donor of chlorine (Cl) to produce metal chlorides and contribute to reducing environmental impact. Although PVC is an important material in our society, an enormous amount of PVC waste is destined for combustion facilities or landfills23). In addition, these disposal methods of PVC are environmentally concerned since it generates toxic substances (e.g., HCl and dioxins) and requires securing a massive land area respectively23,24).
Saeki et al.23) ground the LiCoO2 and LiCoO2/PVC mixture to determine the effect of the mechanochemical process on metals leach efficiencies. They ground samples with different grinding times (0–36 hours), but other grinding conditions are not described. They could achieve about 99 % of Li and 90 % of Co leach efficiencies after water leaching of the mechanochemically reacted sample, while the mechanically activated sample showed 60 % of Li leach efficiency with nearly 0 % of Co leach efficiency. They proposed the overall mechanochemical reaction as follows:
The two grinding processes showed different Li pathways, LiCl from the mechanochemical reaction of mixture and lithium hydroxide (Li(OH)) from grinding only LiCoO2, and resulted in the difference in the solubility of Li in water. Furthermore, C in PVC played an important role in reducing Co3+ in LiCoO2 into Co2+ and forming cobalt chloride (CoCl2). From XRD analysis, they could detect the peaks of CoCl2×2H2O after 12 hours of mixture grinding.
On the other hand, Wang et al.24) developed a process to selectively recover Li from solution and Co as a form of cobalt iron oxide (CoFe4O6) using zero-valent Fe powder and PVC as co-grinding reagents. During the mechanochemical process, Fe aids the dichlorination of PVC and recombination with Co as the magnetic CoFe4O6 product. In previous research, the addition of zero-valent metals (e.g., Ni and Fe) promoted the PVC’s dichlorination via reduction without producing undesired by-products compared to the other reducing reagents. In addition, zero-valent metals favor forming LiCl while showing a low conversion of Co to chloride since Li is more active than Co. They could achieve almost 100 % of Li and 8.1 % of Co leach efficiency with 96.4 % of Cl leach efficiency under optimal conditions of the LiCoO2/PVC/Fe mass ratio of 1:1:2, BTR of 50:1, and grinding speed at 600 rpm for 12 hours. The remaining Co in Li-free residue was recovered as magnetic material after calcination.
3.2.1.2. Carbonation
Wang et al.28) and Qu et al.29) focused on the leaching process after the mechanochemical process. Both groups only exploited trace C in spent cathode powders rather than introducing additional co-grinding reagents.
Wang et al.28) leached mechanically activated LiCoO2 using acetic acid (HAc). They widely investigated the impact of mechanical processing on the physicochemical properties of LiCoO2 powders and analyzed the effects of these changes on Li and Co leach efficiencies. They ground LiCoO2 powders by varying the grinding speeds only (0–500 rpm) and observed the changes in particle characters, including particle size, surface characteristics, and phase structure. They detected that the particle size reduced with the increasing grinding speed, but further size reduction did not occur when the grinding speed exceeded 300 rpm due to the agglomeration. Nevertheless, owing to the formation of small size pores and crack, the specific surface area consistently increased up to 500 rpm. When the rotary speed exceeded 300 rpm, they detected the carbonate formation and reduction of Co3+ to Co2+ by the presence of C in LiCoO2 powder using high-resolution XPS. These changes reduced the reagent consumption and enhanced the metal leach efficiencies by reacting faster to organic acid. From the activated spent LiCoO2 cathode, they could extract metals as lithium acetate (LiAc) and cobalt acetate (CoAc2) (Eqs. (15) and (16)) and recover in the forms of cobalt hydroxide (Co(OH)2) and Li2CO3 using NaOH and Na2CO3 respectively. In addition, HAc could be regenerated into sodium acetate (NaAc) during the precipitation process.
They could achieve 99.8 % of Li and 99.7 % of Co leach efficiencies from mechanically activated LiCoO2 under optimal grinding of a grinding speed at 500 rpm and leaching conditions of 20 vol % of HAc, 5 vol % of H2O2 for 15 minutes at room temperature.
Qu et al.29) introduced quartz (SiO2) as a grinding-aid media for the mechanical activation of LiCoO2. They studied various grinding factors, including the LiCoO2/SiO2 mass ratios (1:0–1.5), grinding speeds (0–500 rpm), and grinding times (10–30 minutes), and leached mechanically activated samples using H2Cit at 80 °C for 30 minutes. They observed the LiCoO2 particles before and after mechanical activation with/without SiO2 using SEM. From the SEM images, they detect the presence of organic binders that agglomerate the particles and hinder the penetration of leach solution, but the binder was removed, and floccular-shaped particles were produced after grinding in the presence of SiO2. In addition, SiO2 acted as a grinding media and enhanced the fineness of ground LiCoO2 particles compared to the grinding without SiO2. Similar to the study by Wang et al.28), C in the LiCoO2 powder played as the reductant, reduced Co3+ to Co2+, and subsequently produced Li2CO3. By contrast, they proposed slightly different reaction mechanisms, as described in Eqs. (17) and (18).
They could achieve 97.22 % of Li and 94.91 % of Co leach efficiencies under optimal grinding conditions of a LiCoO2/SiO2 mass ratio of 1:1, a grinding speed at 500 rpm for 30 minutes, while only 62.50 % of Li and 75.21 % of Co leach efficiencies were obtained from mechanically activated sample without SiO2.
On the other hand, Wang et al.34) who more focused on the mechanochemical reaction by introducing dry ice (CO2(s)) as a co-grinding reagent to destroy the LiCoO2 structure and recover Li and Co separately. They ground the sample by changing the LiCoO2/CO2(s) mass ratios (1:0–40), grinding speeds (0–700 rpm), and grinding times (0.5–4 hours). During the mechanochemical process, they could produce Li and Co in the forms of Li2CO3 by in-situ conversion and water-insoluble cobalt tetroxide (Co3O4) (Eq. (19)).
The mechanical forces destroyed the LiCoO2 structure, the cluster of octahedral metal oxides that Li atoms are intercalated between Co-O layers. Li atoms were released from the crystal structure and formed Li2CO3 by adsorbing CO2 molecules through their own activity. As the mechanochemical reaction time was prolonged, the products were released and carried away while producing a fresh surface for new collision, and the reaction between LiCoO2 and CO2(s) was continued28,34).
Each product is separated through the water leaching and filtration process. In the following pyrometallurgical process, they readily obtained high-value Co metal from Co3O4 in Li-free residue by exploiting C in residue, a so-called self-reducing reagent. They could achieve 95.04 % of Li2CO3 recovery with 99.75 % purity under optimal conditions of a LiCoO2/CO2(s) mass ratio of 1:20 and a grinding speed at 700 rpm for 1.5 hours.
3.2.2. Chelation process
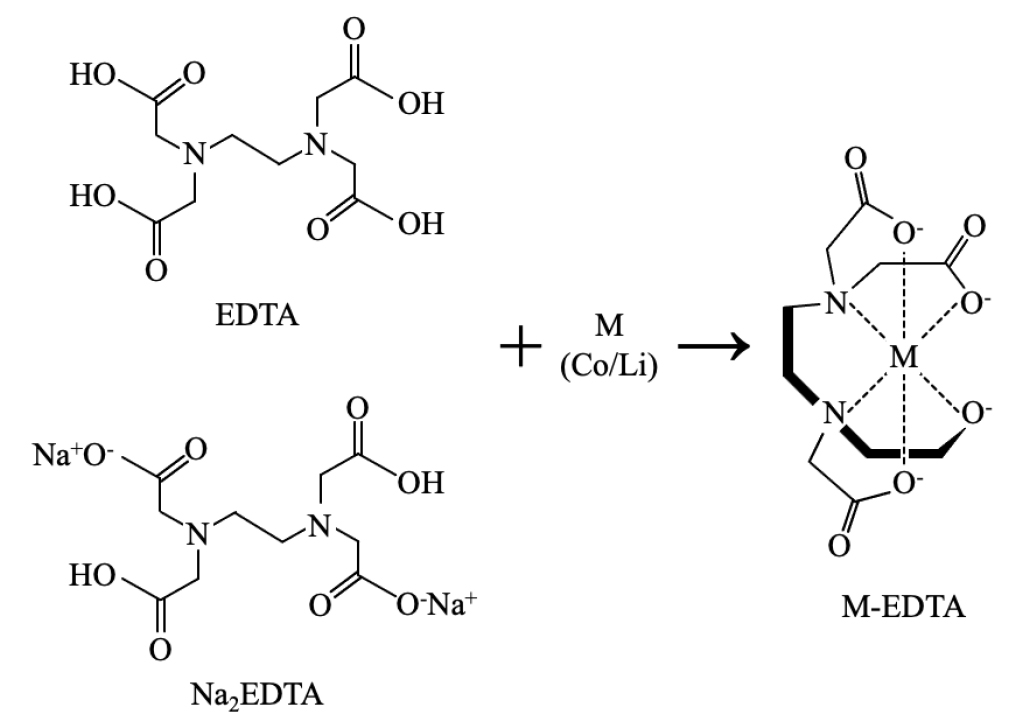
Wang et al.26) used EDTA and Na2EDTA as co-grinding reagents to chelate Li and Co from cathode powder during the mechanochemical processing. They tested various grinding conditions, including the LiCoO2/EDTA mass ratios (1:1–6), grinding speeds (300–600 rpm), grinding times (1–5 hours), and BTRs (10–125:1), with two chelating reagents, and then leached in the water for 30 minutes and recovered metal in the form of Co3O4 and Li2CO3 by chemical precipitation. They explained the extraction mechanism by conversion of metals in LiCoO2 into water-soluble compounds (Li- or Co-EDTA) by the solid-solid reaction, as shown in Fig. 3. During the mechanochemical process, they noted that EDTA having six ligands (two amines and four carboxyl groups) could coordinate with Li and Co with a 1:1 molar ratio by providing lone pair electrons and entering the empty orbit of Li and Co through the general effects of mechanical force (e.g., defect and dislocation).
The metal recovery rates steadily increased until reaching a LiCoO2/EDTA mass ratio of 1:4, which is slightly higher than the 1:1 molar ratio of metal ions/EDTA equal to the mass ratio of 1:3.6. In addition, the larger amount of energy was required to totally damage the LiCoO2 structure and efficiently extract metals so they should maintain the grinding variable at higher levels. They could achieve almost 99 % of Li and 98 % of Co leach efficiencies under optimal grinding conditions of a LiCoO2/EDTA mass ratio of 1:4, a BTR of 80:1, and grinding at 600 rpm for 4 hours. Meanwhile, they could achieve only 74 % of Li and 71 % of Co leach efficiencies under the same grinding conditions when Na2EDTA was introduced instead of EDTA. They mentioned that the number of carboxyl groups in the molecular made a difference in the chelating performance of two reagents.
3.2.3. Complexation
Cai et al.27) used solidified alginic acid (HAlg) as a reductant in the mechanochemical reaction. After the mechanochemical process, metals in the LiCoO2 are converted to water-soluble alginate complexes, which are lithium alginate (LiAlg) and cobalt alginate (CoAlg2), and recovered in the subsequent water leaching and centrifugation process. They tested various grinding conditions, including the LiCoO2/HAlg mass ratios (1:8–16), grinding speeds (200–600 rpm), grinding times (1–5 hours), and the amount of H2O2 (1.25–2.25 mL).
They explained the extraction mechanism during the mechanochemical process as follows: (1) metal ions were released from collapsed LiCoO2 crystal under the reductant-assisted condition, while breaking the polymer-structured HAlg simultaneously and then (2) liberated metal ions complexed with carboxyl groups in the broken alginate molecular chain, as forms of metal alginates. Although HAlg is directly involved in the extraction mechanism, an excessive amount of HAlg deteriorated metal recovery in centrifugation by forming the hydrogen bonding between broken and unbroken alginate molecular chains and remaining in leach residue.
They could achieve 97.6 % of Li and 98.6 % of Co leach efficiencies under optimal conditions of a LiCoO2/HAlg mass ratio of 1:10 and grinding speed at 500 rpm for 4 hours with the addition of 2 mL of H2O2.
4. Summary
The intensive mechanical forces imparted by mechanochemical processing promote unique reactions which are not observed in the conventional route, and lead to the selective metal recovery process with high recovery efficiency from spent LiFePO4 and LiCoO2 cathodes. The solvent-free (or minimal use of solvent) condition avoids forming undesirable products while generating the final product by direct solid-solid reaction. These characteristics of the mechanochemical process not only simplify the subsequent purification stage but more importantly reduces the significant amount of Li loss reported in the conventional process. In addition, many researchers introduce natural organic materials and/or regenerate reagents to establish greener and sustainable processes.
In the case of LiFePO4, where Li has a significant economic value than other cathode chemistries, selective Li recovery from the refractory FePO4 structure is the major interest. In most applications, the selective Li recovery is completed during the mechanochemical and water-leaching process by the combination of mechanical force and co-grinding reagent as follow: (1) In-situ formation of water-soluble Li compound and (2) Oxidation Fe2+ to Fe3+ ions and prevention of Fe dissolution from FePO4 structure.
Both Li and Co are important recovery targets in LiCoO2, and thereby these metals can be recovered together by the leaching-purification process or separately by leaching Li first and extracting Co from Li-free residue in the following thermal process. Besides, the trace C in spent cathode powder, which hinders the chemical reaction in the hydrometallurgical process, is utilized as the reductant for high valency Co in the mechanochemistry-assisted process.
Many studies proposed sustainable and efficient recovery processes using mechanochemistry as well as economic feasibility. However, all of the studies are conducted on laboratory scales, so further development should be completed in the scaled-up conditions for its commercialization.