1. General Introduction
2. Experimental
2.1. Materials
2.2. Pretreatment
2.3. Leaching
2.4. Extraction procedure
2.5. Precipitation
2.6. Analytical method
3. Result and discussion
3.1. Thermal treatment for eliminating EVA resin
3.2. Hydrochloric acid leaching process
3.3. Copper extraction by Lix984N
3.4. Co-precipitation for separating tin and lead
4. Conclusion
1. General Introduction
The PV technology is one of the greenest and most promoting green energy-generating technologies as it generates electricity directly from the solar energy and therefore avoid fossil energy consumption during energy production process. By the statistic, 106GW of solar PV capacity added in 2018 and the accumulation of PV modules is now increased to 508GW1). More solar PV was installed than the net capacity additions of fossil fuels and nuclear power combined. However, the rapidly increasing production and installation of PV modules will cause a huge problem in the future due to the life of PV modules (15-20years)2). According to the research, the accumulation of waste modules will reach to 8600 tons in 2030. Moreover, Crystalline-silicon Photovoltaic modules account for more than 90% of the waste and there is no critical commercial way to recover the valuable metals from the PV modules. The solar module is assembled by tempered glass which allows light penetrates the c-Si cells and protects the cells; EVA resin is the adhesive of glass, solar cells and back sheet. The target of this study, PV ribbon, which looks like the wire what carried the electrical energy produced by c-Si solar cells.
As the importance of recycling PV modules increasing, variable researches were published to purify the valuable materials and reduce the mass and volume of the waste. The research from Korea separated PV ribbon by melting the coating layer composed of tin and lead and separate copper layer; then used the thermal process under a low atmosphere with CH4 to purify Copper, and the purity of Cu was 99.5%3). Another research oxidized the Pb-Sn alloy layer of PV Ribbon and used ball milling to separate copper and oxide layer. The other research leached Pb-free PV ribbon by HCl and HNO3 then separated it by precipitation and electrochemical4). The research from KIGAM showed a recycling process by leaching Sn ions by HCl and separated Cu by adding Sn powder to reductive copper ions to copper metals. After the leaching process, this research used TBP to extract Sn from the leach solution, and <1% of Pb was extracted5).
This study provided the leaching process to separated tin, lead and copper from silver by using hydrochloric acid. After the leaching process, this study extracted Cu by Lix984N and stripped by H2SO4. Tin and lead were separated by adjusting pH value and precipitation.
2. Experimental
2.1. Materials
Poly-crystalline silicon PV modules used in this study are from the waste PV modules recycling factory. Table 1 shows that PV modules are composed of aluminum frame (14.7%), tempered glass (68.4%), Ethylene-vinyl acetate resin and back sheet (11.2%), PV cells (3.1%), Ribbon (1.3%), and Junction Box (1.3%), Table 2 shows the chemical composition analyzed by ICP-OES (Inductively Coupled Plasma-Optical Emission Spectrometry, PerkinElmer optima 2100DV), and Fig. 1 shows the SEM-EDS figure of PV ribbons. The chemical composition of commercial PV ribbons is mainly composed of 83% of Cu, 9% of Sn, 4.5% of Pb and 3% of Ag.
Table 1.
Chart of PV modules mass fraction
| Aluminum frame | Tempered glass | EVA and backsheet | PV cells | Ribbon |
| 14.7% | 68.4% | 11.2% | 3.1% | 1.3% |
2.2. Pretreatment
PV ribbons were dismantled between the layers of EVA resin and c-Si PV cells. After dismantling, some EVA resin would attach on the surface of the PV ribbon. Pretreatment was employed to eliminate the resin from PV ribbons by heating the resin to evaporation.
2.3. Leaching
Hydrochloric acid was employed to leach PV ribbon. Ag was separated from other metals by halogenating to AgCl. To optimize the experimental condition for effective leaching, this study was made carrying different process parameters viz. time (0.5-4hr), oxidation rate, temperature (25-85℃), liquid-solid ratio, and acid concentration (0.5M-6M).
Leaching efficiency of metal was calculated by the equation below:
| $$\mathrm X\%=\left(\frac{{\mathrm V}_{\mathrm c}C_x}{{\mathrm{Mw}}_{\mathrm x}}\right)\times100\%$$ | (1) |
X%=leaching efficiency (g/L), M=the weight of the sample (g), Vc=the volume of the liquid (L), Wx=target metals weight fraction (wt%).
2.4. Extraction procedure
After the leaching process, the sample was extracted by Lix984N and stripped by H2SO4 in the optimal condition. This study also carried the parameters of extraction process viz. pH value (0.5-3), O/A ratio, extractant concentration, stripped solvents concentration, and reaction times.
The extraction efficiency of Lix984N and stripping efficiency were calculated by equations below:
| $$\mathrm E\%=\left(\frac{\sum_{}^{}\lbrack\mathrm M\rbrack_{\mathrm{org}}}{\sum_{}^{}\lbrack\mathrm M\rbrack_{org}+\sum_{}^{}\lbrack\mathrm M\rbrack_{aq}}\right)\times100\%$$ | (2) |
[M]org=the concentration of metal in the organic phase after extraction, [M]aq=the concentration of metal in the aqueous phase after extraction.
| $${\mathrm E}_{stripping}\%=\left(\frac{\sum\lbrack\mathrm M\rbrack_{\mathrm{after}\;\mathrm{stripping}}}{\sum\lbrack\mathrm M\rbrack_{\mathrm{before}\;\mathrm{stripping}}}\right)\times100\%$$ | (3) |
[M]after stripping=the concentration of metal in the aqueous phase after stripping from organic phase, [M]before leaching= the concentration of metal in the organic phase before the stripping process.
2.5. Precipitation
This study provided the separation process to separate Lead and Tin through precipitation. In this process, the concentration of Lead and Tin from the liquid after extraction in different pH value would show. And this study would discuss the difference of concentration between Lead and Tin. After all, this study chose the right point to effectively separate them.
2.6. Analytical method
The sample after leaching, extraction process, and precipitation were filtered by 0.45μm of membrane filter and were diluted by 1% HNO3 solution for ICP-OSE (Inductively Coupled Plasma-Optical Emission Spectrometry, PerkinElmer optima 2100DV) analyst. The ICP-OSE analyst was calibrated with ICP multi-element stander and tin standard solutions. Each aqueous solution was analyzed for three times and averaged for the reported.
3. Result and discussion
3.1. Thermal treatment for eliminating EVA resin
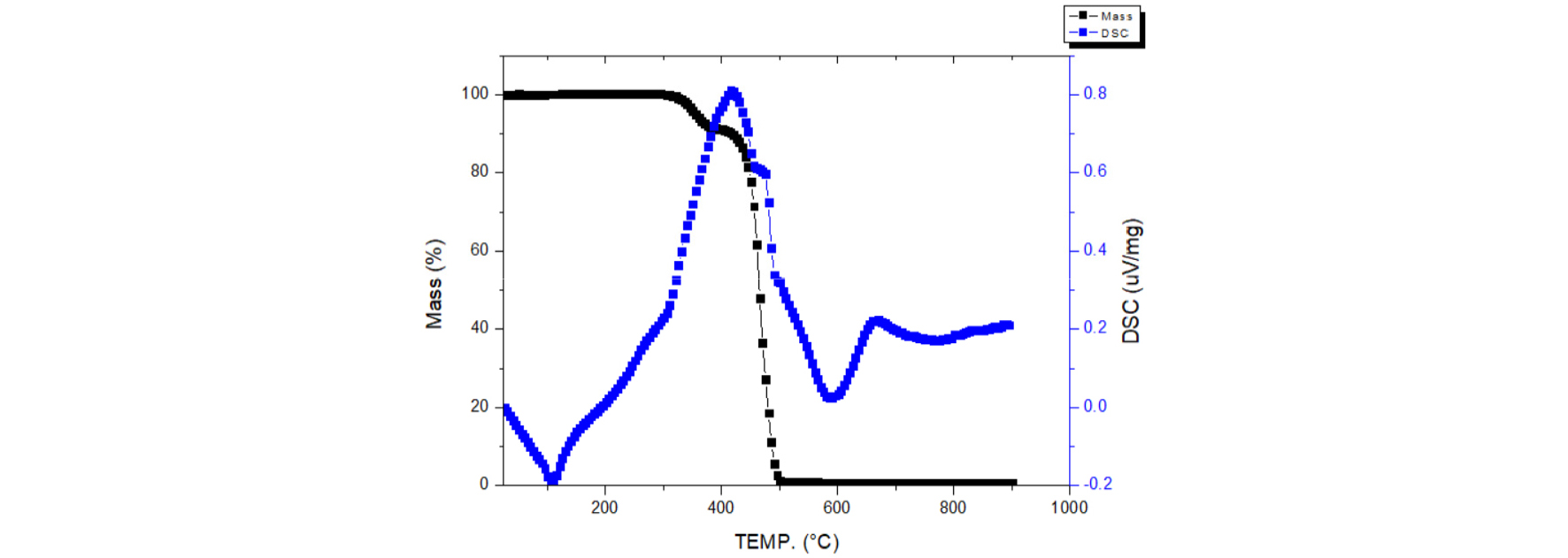
Fig. 2 shows the TG analyst of EVA resin6). After heating over 500℃, the mass decline of EVA resin approach to zero. This study used the thermal process to eliminate EVA resin by heating to 500℃ for 5 hours in the atmosphere. This process can eliminate 99.97% of EVA resin attaching on PV ribbons.
3.2. Hydrochloric acid leaching process
This study provided an acid leaching process to separate and leach valuable metals into the water as the state of Cu2+, Sn4+, and Pb2+ ions.
3.2.1. Effect of the concentration of hydrochloric acid
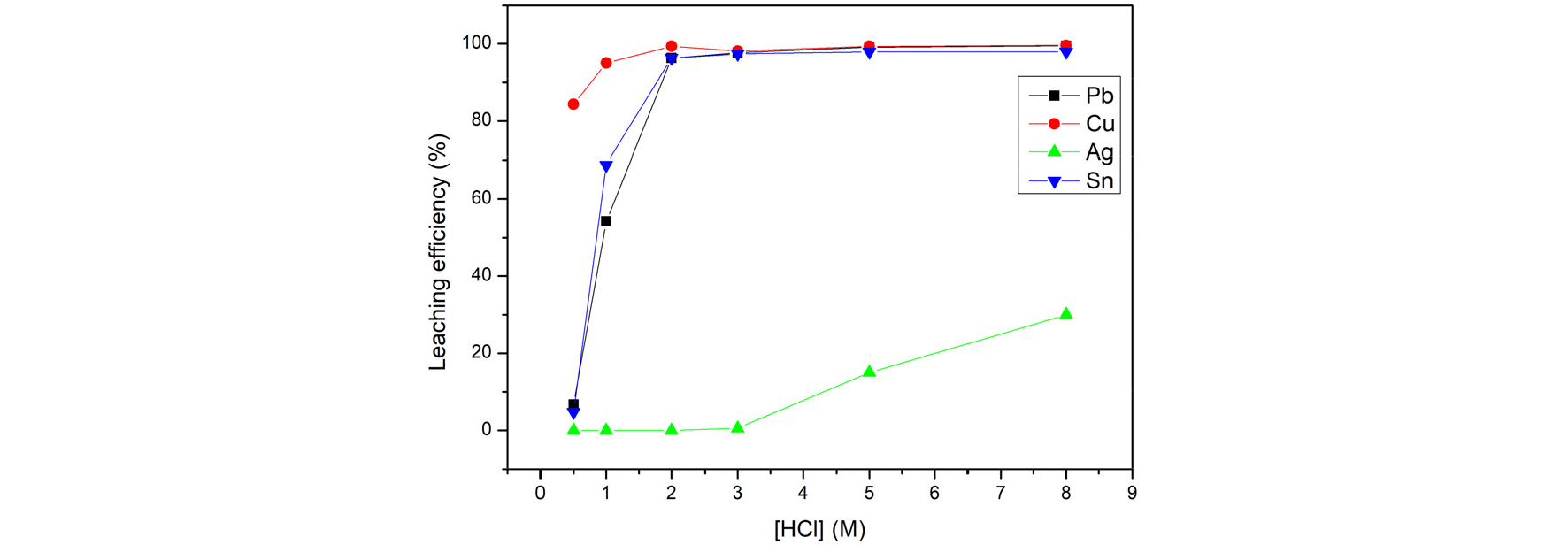
Fig. 3 shows the leaching efficiency from adjusting the concentration of HCl in 3 hours, 10% of H2O2, 20 of L/S ratio, and 60 degrees of temperature. Lead, Tin and Copper get a good leaching efficiency, up to 97.5% on 3M of hydrochloric acid. After rising the concentration of HCl, the leaching efficiency of silver would gradually increase but not over 30%. To separate silver from the other metals. This study chose 3M as an optimal condition.
3.2.2. Effect of the volume percentage of H2O2
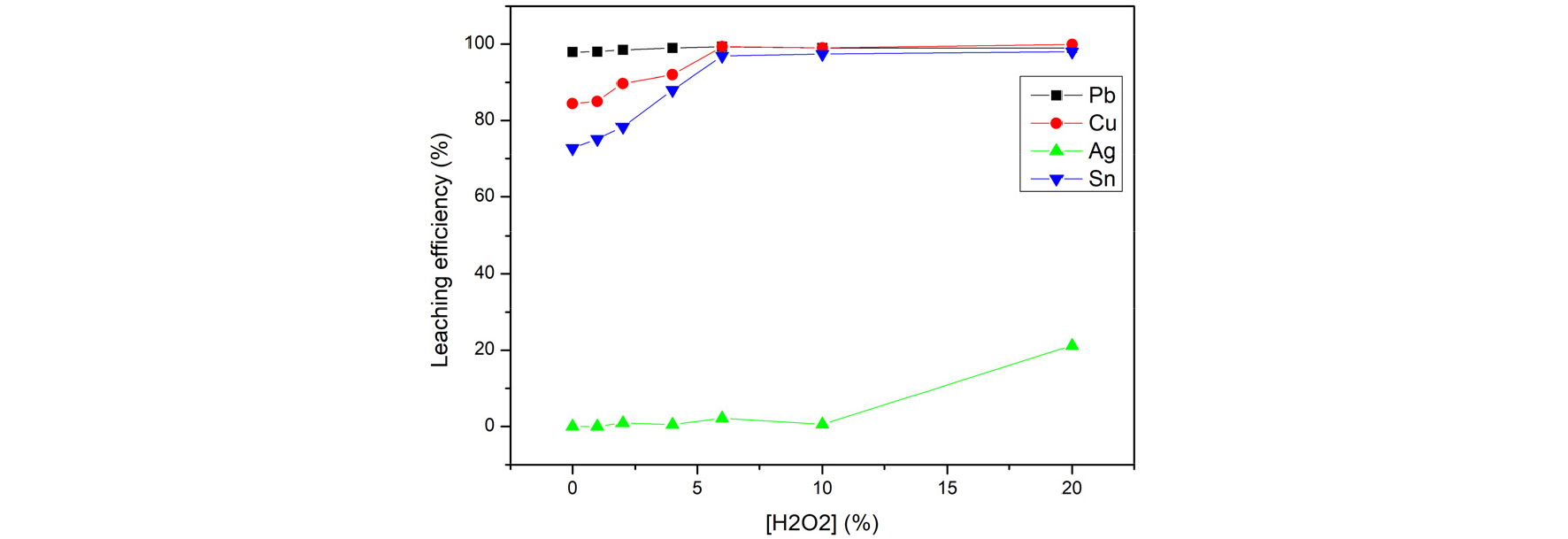
Fig. 4 shows the leaching efficiency from adjusting the volume percentage of H2O2 to increase the oxidation rate for 3M of HCl, 3hours, 20 of L/S ratio, and 60 degrees of temperature. As the volume percentage of H2O2 increased, the leaching rates of Lead, Tin and Copper increased. In the leaching tests, tin was oxidized with hydrogen peroxide as the following equation4).
| $$\mathrm{Sn}+H_2O_2+2H^+\rightarrow Sn^{2+}+2H_2O$$ |
This equation shows the significance of oxidation for tin to increase the leaching efficiency. Because that 6% of H2O2 shows effective leaching efficiency (99.3% of lead, 99.35% of copper, and 96.9% of tin) in this experiment and 6%-20% tend to balance, this study set the optimal parameters of H2O2 to 6%.
3.2.3. Effect of Liquid-solid ratio
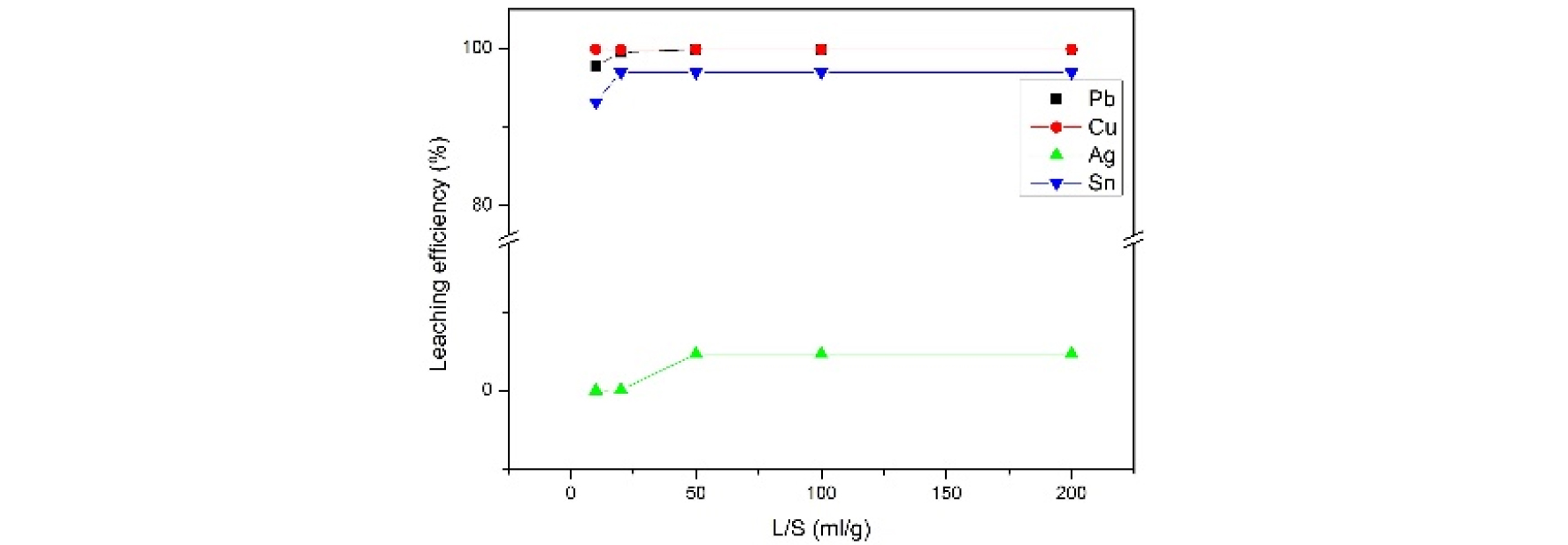
Fig. 5 shows the leaching efficiency from adjusting the liquid-solid ratio. The LS ratio was investigated by verifying the ratio from 10 to 1000. As the result, PV ribbon has effective leaching efficiency (99.5% of lead, 99.85% of copper, and 97% of tin) after 20. Hence, the optimal condition of the L/S ratio was chosen as 20ml/g.
3.2.4. Effect of the reaction times
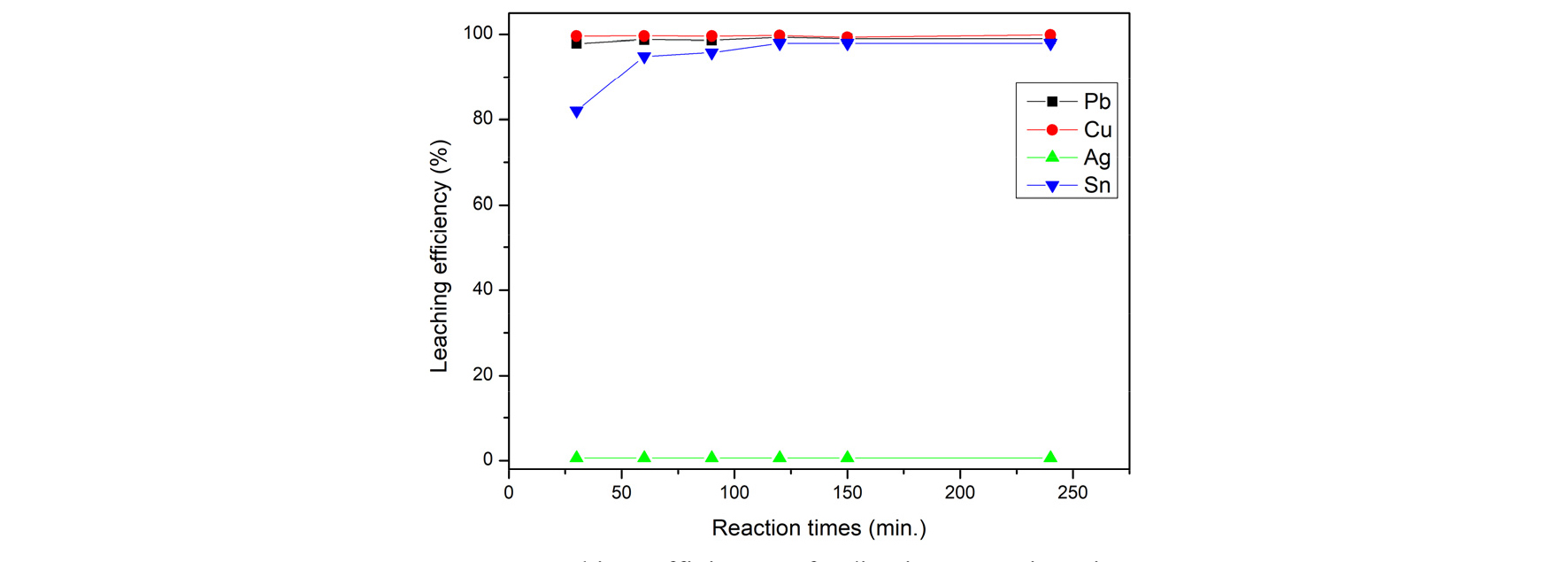
Fig. 6 shows the leaching efficiency from adjusting the reaction times for 3M of HCl in 20 of LS ratio, and 60 degrees. Reaction times was investigated by verifying from 30 minute to 240 minutes. As the result showed, tin needed the time to leach. After 120 minute, PV ribbons has effective leaching efficiency (99.3% of Lead, 99.74% of Copper, and 98.3% of Tin) and tend to balance. Hence, the optimal condition of Reaction times was chosen as 120 minutes.
3.2.5. Effect of the temperature
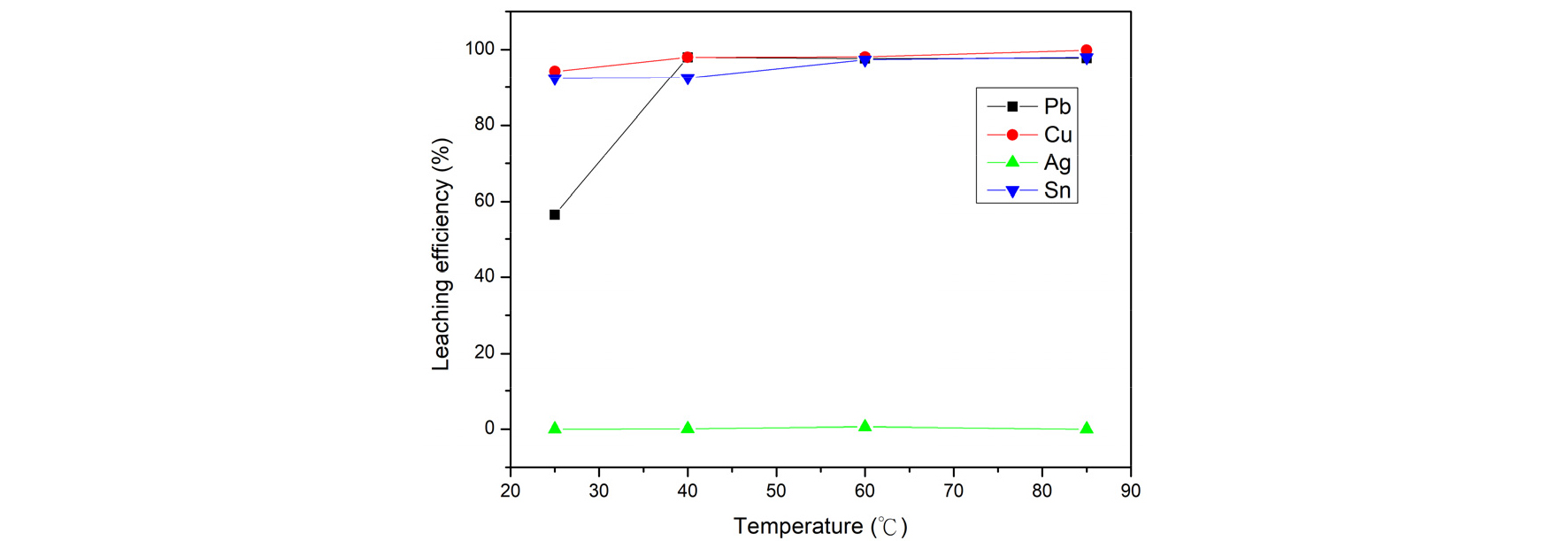
Fig. 7 shows the leaching efficiency from adjusting temperature. Temperature was investigated by verifying the temperature from 25 degrees to 85 degrees. As the result showed, set the reaction temperature to 60 degrees has effective leaching efficiency. Hence, the optimal condition of reaction temperature was chosen as 60°C.
Hydrochloric acid can leach most of the metal inside the PV ribbons and nearly no effect on silver due to the reaction equation below.
| $$Ag_{(aq)}^++Cl_{(aq)}^-\rightarrow AgCl_{(s)}$$ |
The solubility product (Ksp) of AgCl is 10-9.82 at 25°C4). That’s the reason indicated Ag ion precipitate easily and rapidly as AgCl. This study used this chemical property to separate Ag from other metals.
3.3. Copper extraction by Lix984N
After the leaching process, Lix984N was employed to extract Copper from Lead and Tin. Lix984N was diluted by kerosene.
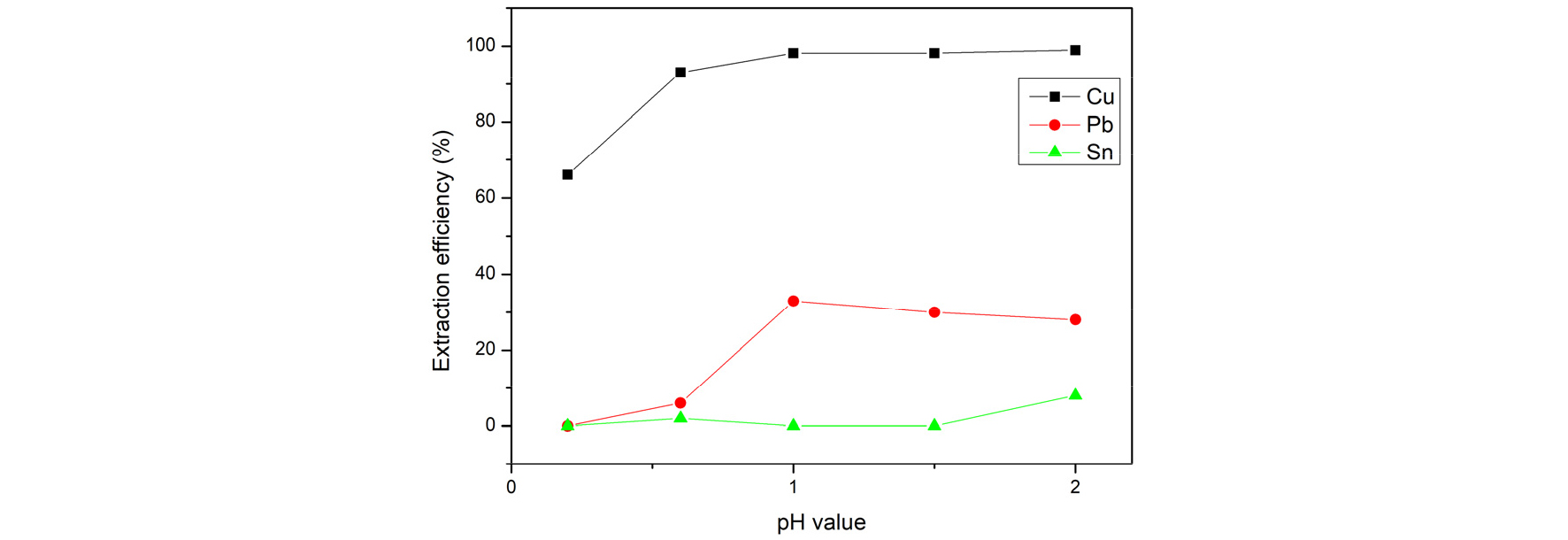
3.3.1. Extraction efficiency of adjusting initial pH value
Because Tin ion started to precipitate at pH value=2.1, the initial pH value in this study was adjusted within 0.2 to 2 in the condition of A/O=1, 30%(v/v) of Lix984N, shaking frequency of 500 min-1 and reaction times of 10 minute. Fig. 8 shows the effect of aqueous phase pH value on extraction percentage. The optimal parameter was found. The result showed that the extraction percentage of copper and lead increased when the aqueous phase pH value increased. Lix984N is an acidic extractant. Extraction reaction of Copper with Lix984N could be represented by7,8)
| $$\mathrm{Cu}_{(\mathrm{aq})}^{2+}+2{\mathrm{HA}}_{\mathrm{org}}\rightarrow{\mathrm{CuA}}_{2,\mathrm o}+2\mathrm H_{\mathrm{aq}}^+$$ |
As the equation above shows, Lix984N in the high acidity of aqueous phase is a disadvantage of releasing hydrogen ion from Lix984N. However, in order to separate copper and lead from extraction, the optimal parameter of pH value was chosen as 0.6. Because the Lix984N exchanges hydrogen ion selectively, the tin and lead ions mostly remain in the water phase. Copper, tin, and lead of extract phase can be separated.
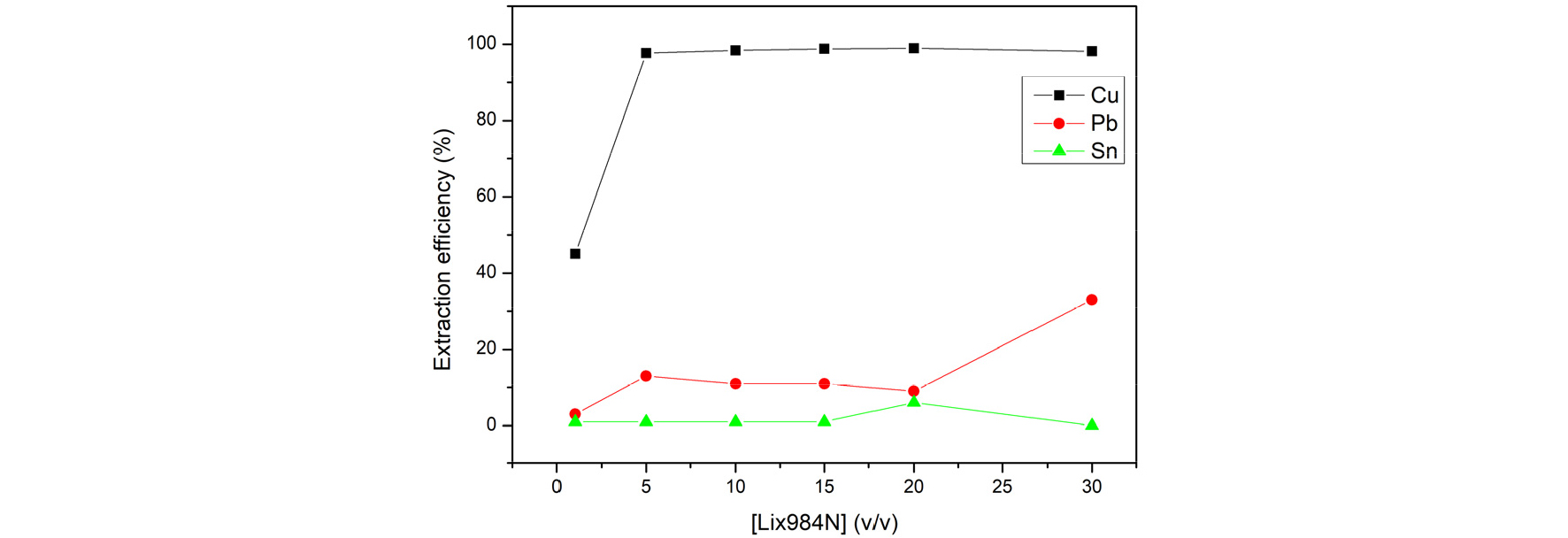
3.3.2. Extraction efficiency of adjusting the concentration of Lix984N
The effect of A/O ratio was examined, and the results are shown in Fig. 9 The result showed that at the v/v(%) =20% has the highest extraction efficiency for copper (99%). The concentration of Lix984N=20% was fixed for subsequent experiment.
3.3.3. Extraction efficiency of adjusting aqueous-oil volume ratio
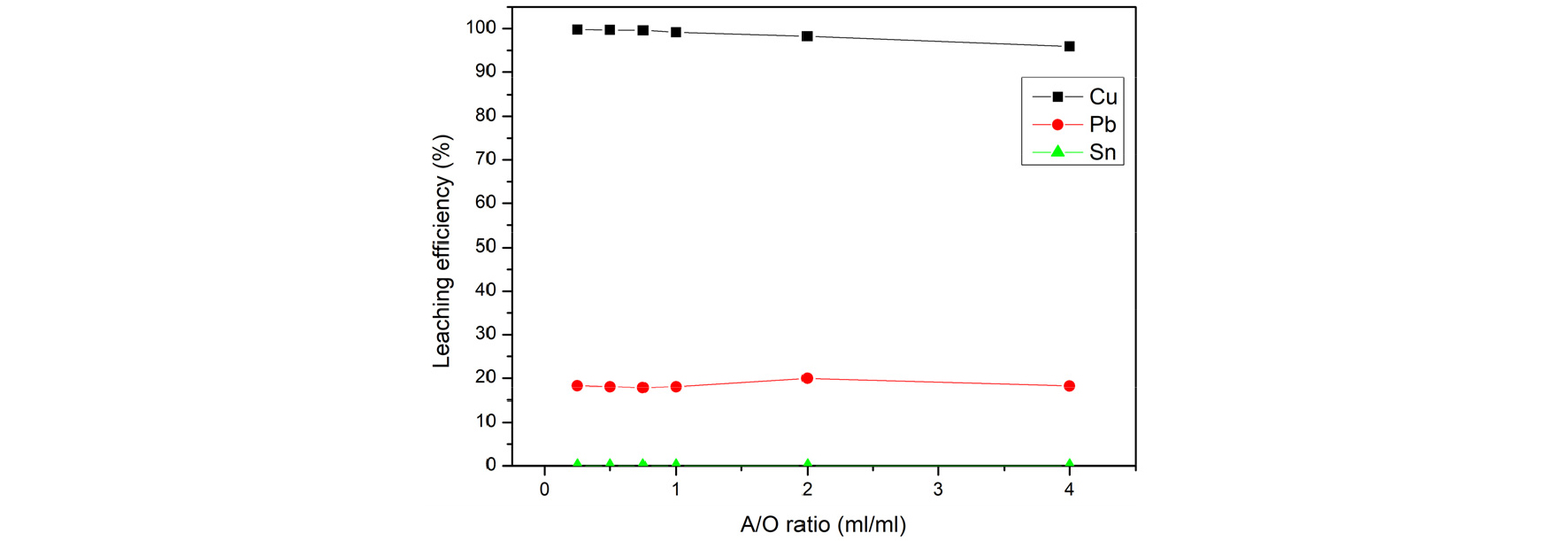
The effect of A/O ratio was examined, and the results are shown in Fig. 10 The result shows that no significant change in the extraction percentage of copper. The A/O ratio=1/1 was fixed for subsequent experiments.
3.3.4. Extraction efficiency of adjusting reaction times
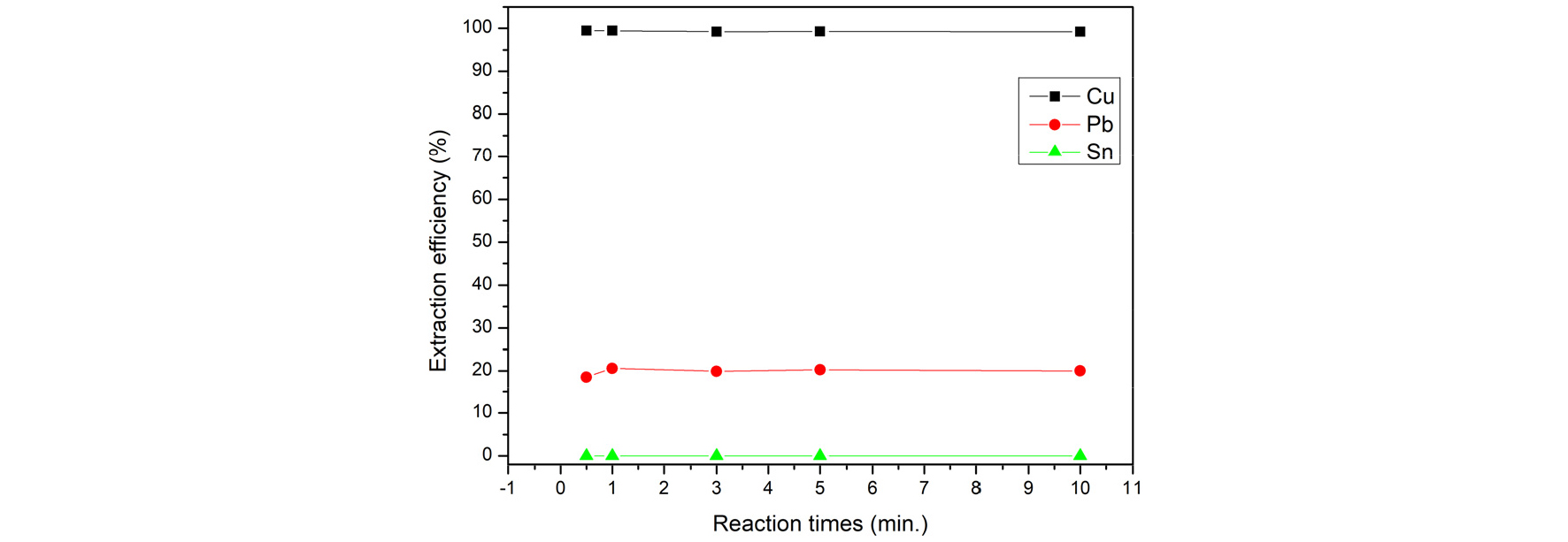
Under the condition of fixed solution pH value=0.6, O/A ratio=1, 20% of Lix984N and shaking frequency of 500min-1, the shake time was changed to 0.5, 1, 3, 5, and 10, respectively. The result is shown in Fig. 11. The results showed that reaction times has less influence the extraction percentage of copper. The extraction efficiency can easily reach to 99% in 1 minute. Hence, the reaction times=1 min. was fixed for the experiments.
3.3.5. Stripping experiment
From the extraction experiments, it was observed that Lix984N can extract copper from Cu-Pb-Sn solution. The organic phase with copper was stripped to water phase by different concentration of sulfuric acid, OA ratio and reaction times. The results indicated that 3M H2SO4 in the condition of O/A ratio=1and reaction times=10 minutes can efficiently strip the copper from organic phase.
In terms of extraction, the optimal extraction operating condition of this technique were copper-lead-tin solution with pH 0.6, 20% of Lix984N, oil-aqueous volume ratio of 1/1 and 1minute of reaction time. The extraction efficiency of copper was higher than 99.5% as a result. The copper metal in the organic phase can be stripped by H2SO4 successfully.
3.4. Co-precipitation for separating tin and lead
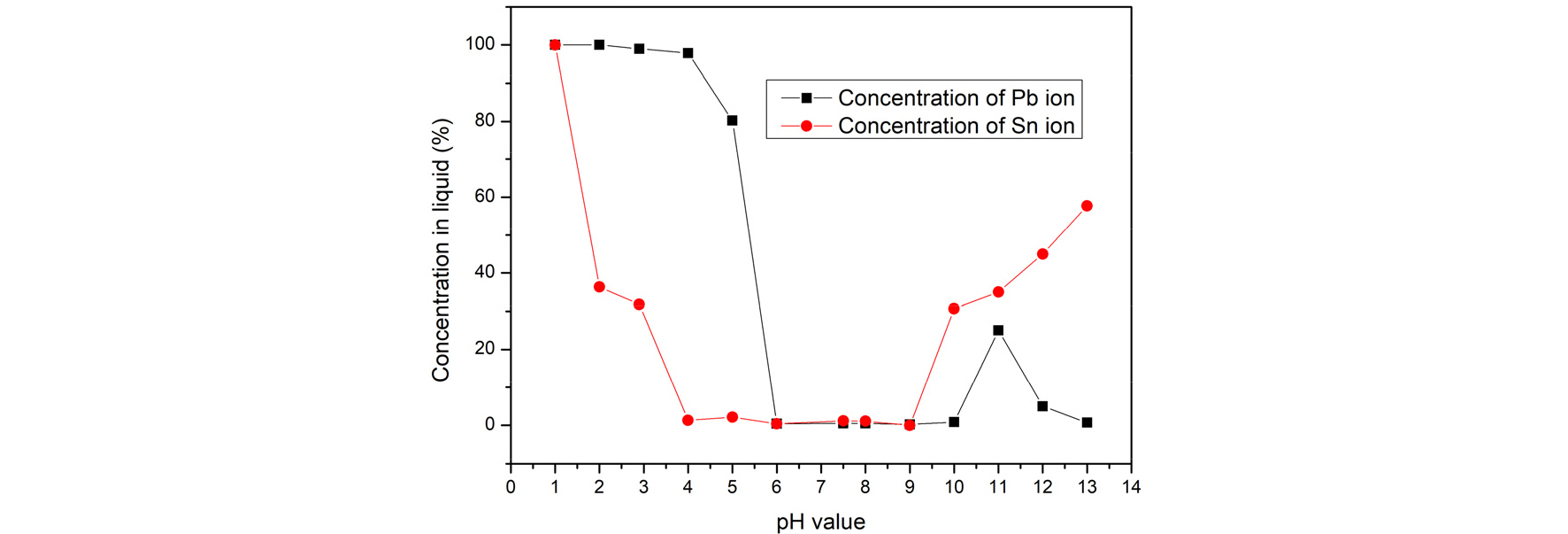
After extraction process, copper was separated from lead and tin. According to the research, Sn2+ started to precipitate to Sn(OH)2 at pH 2.1 and Pb2+ started to precipitate to Pb(OH)2 at pH 7.2 by adding NaOH9). Fig. 12 shows the concentration of metal in liquid by adding NaOH to adjust pH value. The result showed that tin was totally precipitated at pH 4 and lead ions remained in the liquid. After rising the pH value to 6, lead was totally precipitated. This study separated tin by adding NaOH to pH 4 and filtered the precipitate. Then adjust pH value to 7 to collect Pb(OH)2. As Table 3 shows, the recovery of lead and tin can reach 99%. The purity of lead was 98% and tin was 95.4%.
4. Conclusion
This study provides several processes to separate valuable metals from PV ribbons. According to the results of this experiment:
1) Ag was separated by halogenating to AgCl(s).
2) The leaching efficiency of Pb, Cu, and Sn was 99.5%, 99.8%, and 98%, individually. Cu and Sn need H2O2 to rise the Redox value for increasing the leaching efficiency.
3) The optimal condition of leaching is 3M HCl, L/S= 20, reaction times=120min., [H2O2]=6%, and reaction temperature=60 degrees.
4) In order to prevent co-extraction of lead and copper, this study chose pH=0.6 as the optimal condition to extract copper. The optimal condition of Lix984N extracting copper is pH=0.6, [Lix984N]=20%, A/O=1, and reaction times=1min. The extraction efficiency is 99.64%
5) Copper inside the organic phase was stripped by 3M of H2SO4 in O/A=1, reaction times=10 min.
6) The solvent extraction can recover low concentration metals selectively, and the separating effect of high distribution coefficient can be obtained by changing parameters. Therefore, this technique provides an effective method for separating and recovering copper metals.
7) The precipitation separation process can provide high purity (98%, 95.8%) and recovery of lead and tin.