1. Introduction
2. Experimental
3. Results and Discussion
3.1. Effect of HCl concentration
3.2. Effect of extractant concentration
3.3. Comparison of extraction performance between Alamine 336 and Aliquat 336
4. Conclusions
1. Introduction
The development of lithium-ion batteries (LiBs) is currently one of the hot topics and has received the 2019 Nobel Prize in Chemistry1). LiBs are rechargeable power storage devices and have been widely employed in many applications such as mobile phones, electric vehicles and laptop computers2). In addition, lithium metal is also an important component in the alloy of advanced materials3,4). A large number of spent LiBs have been generated with the rapid expansion of electrical products5). The spent LiBs can be hazardous to the environment and are considered as secondary resources6). The main composition of spent LiBs is lithium (5-7 wt.%), aluminum (1-3 wt%), cobalt (5-20 wt%), copper (3-12 wt%), manganese (10-15 wt%), and nickel (5-10 wt%)7,8). Since the ores containing cobalt and nickel are depleting, the recovery of metals from spent LiBs is very important in terms of environmental protection and resource conservation.
Several hydrometallurgical processes such as acid leaching, bioleaching, precipitation, solvent extraction, and ion exchange have been applied for the recovery of valuable metals from spent LiBs9,10,11,12). There are two common routes for metal recovery from leach liquor of spent LiBs. The first way is to selectively separate the primary metals (Co, Li, Ni), leaving the undesirable metals (Fe, Mg, Mn) in the raffinate8). The other route is to remove the impurities (Al, Mn, Fe) from the leach liquor by precipitation or solvent extraction, leaving Co, Li, and Ni in the solution13). Although many research papers on spent LiBs are available in previous literature, very few studies have investigated the solvent extraction of Co(II) and Cu(II) from hydrochloric acid solution of spent LiBs. Among these methods, solvent extraction is often preferred by researchers due to its high metal recovery efficiency, simple equipment, and environmentally friendly.
Generally, spent LiBs are pretreated by discharging, dismantling, drying, calcination, and grinding to remove nonmetallic ingredients as well as to obtain materials in powder form. Then, either leaching or bioleaching is employed to dissolve the target metals (cobalt, lithium, nickel) from the resulting powders. Finally, the separation and purification of metals from the leaching solution could be obtained by either solvent extraction or ion exchange.
Many works have been done on the recovery of metals from acidic sulfate solutions of spent LiBs using solvent extraction10,14). Since most of the divalent metal ions exist as cationic form in the acidic sulfate leaching solution of spent LiBs, cation extractants are usually used for the separation of valuable metals15,16). Compared to sulfuric acid leaching solutions of spent LiBs, few works have been reported on the separation of metals from the hydrochloric acid leaching solutions of spent LiBs. In strong hydrochloric acid solutions, Co(II), Cu(II), Mn(II) can form anionic complexes, while Li(I), Ni(II) and Al(III) exist as cationic form. The difference in the predominant species of metal ions in the hydrochloric acid leaching solutions of LiBs can be utilized in separation by using amine extractants. Therefore, in the present work, separation of the metal ions in the hydrochloric acid leaching solutions of spent LiBs was investigated by amine extractants (Alamine 336 and Aliquat 336). The extraction behavior of the metals was investigated as a function of the concentration of hydrochloric acid and extractants. The obtained results can provide some information on the leaching condition of spent LiBs by employing HCl solution as well as the ability to selectively extract Cu(II) or Co(II)/Cu(II) by controlling the acid concentration.
2. Experimental
Cobalt(II) chloride hexahydrate (CoCl2∙6H2O, 97%) and lithium(I) chloride anhydrous (LiCl, 98%) were purchased from Junsei Chemical Co., Japan. Copper(II) chloride dihydrate (CuCl2∙2H2O, 97%) and manganese (II) chloride tetrahydrate (MnCl2∙4H2O, 98%) was supplied by Daejung Chemicals & Metals Co., Korea. Nickel(II) chloride hexahydrate (NiCl2·6H2O, 96%) was obtained from Yakuri Pure Chemicals Co., Japan. A synthetic leach solution of spent lithium-ion batteries was prepared by dissolving the required amount of these chemicals in distilled water. Generally, the main components of LiBs are composed of LiCoO2, LiNiO2, and LiMnO217,18,19). In this work, the composition of hydrochloric acid leaching solution of spent LiBs was obtained from the literature20). The composition of the synthetic solution is shown in Table 1. The acidity of the synthetic solution was controlled by adding HCl (Daejung Co., 35%) or NaOH (Duksan Co., 93%) solution. The commercial extractants like Alamine 336 (tri-octyl/dodecyl amine) and Aliquat 336 (N-Methyl-N,N,N-trioctylammonium chloride) were purchased from BASF Co. They were used without any further purification. Kerosene (Samchun Pure Chemical Co.) was used as a diluent and all the other chemicals used were of analytical grade.
Table 1.
Chemical composition of synthetic leach solution of spent LiBs
| Element | Co(II) | Cu(II) | Li(I) | Mn(II) | Ni(II) |
| Concentration, mg/L | 24000 | 780 | 3000 | 15 | 35 |
In solvent extraction experiments, the aqueous and organic phases were mixed in 50 mL screwed cap bottle with a wrist action shaker (Burrell model 75, USA) for 30 mins. All of the experiments were carried out at ambient temperature and the O/A ratio was fixed at unity. After equilibrium, the two phases were separated from a separation funnel. The concentration of metal ions in the raffinate and feed solution was measured by ICP-OES (Spectro Arcos, Germany). Metal ion concentrations in the organic phase were determined by mass balance. The extraction percentage (E%) of metals was calculated as the ratio of the mass of metal extracted into the organic to that of metal in the aqueous before extraction.
3. Results and Discussion
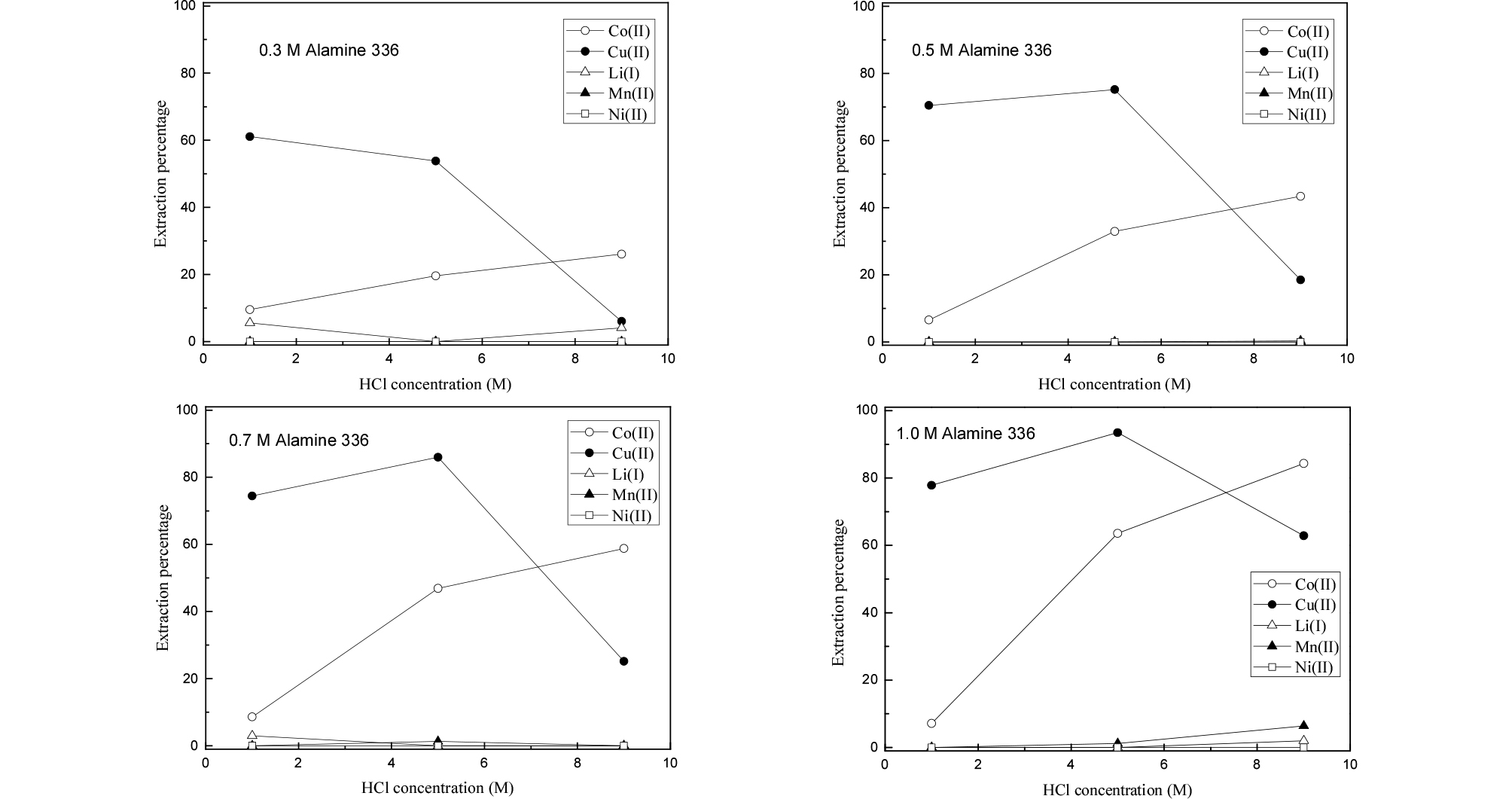
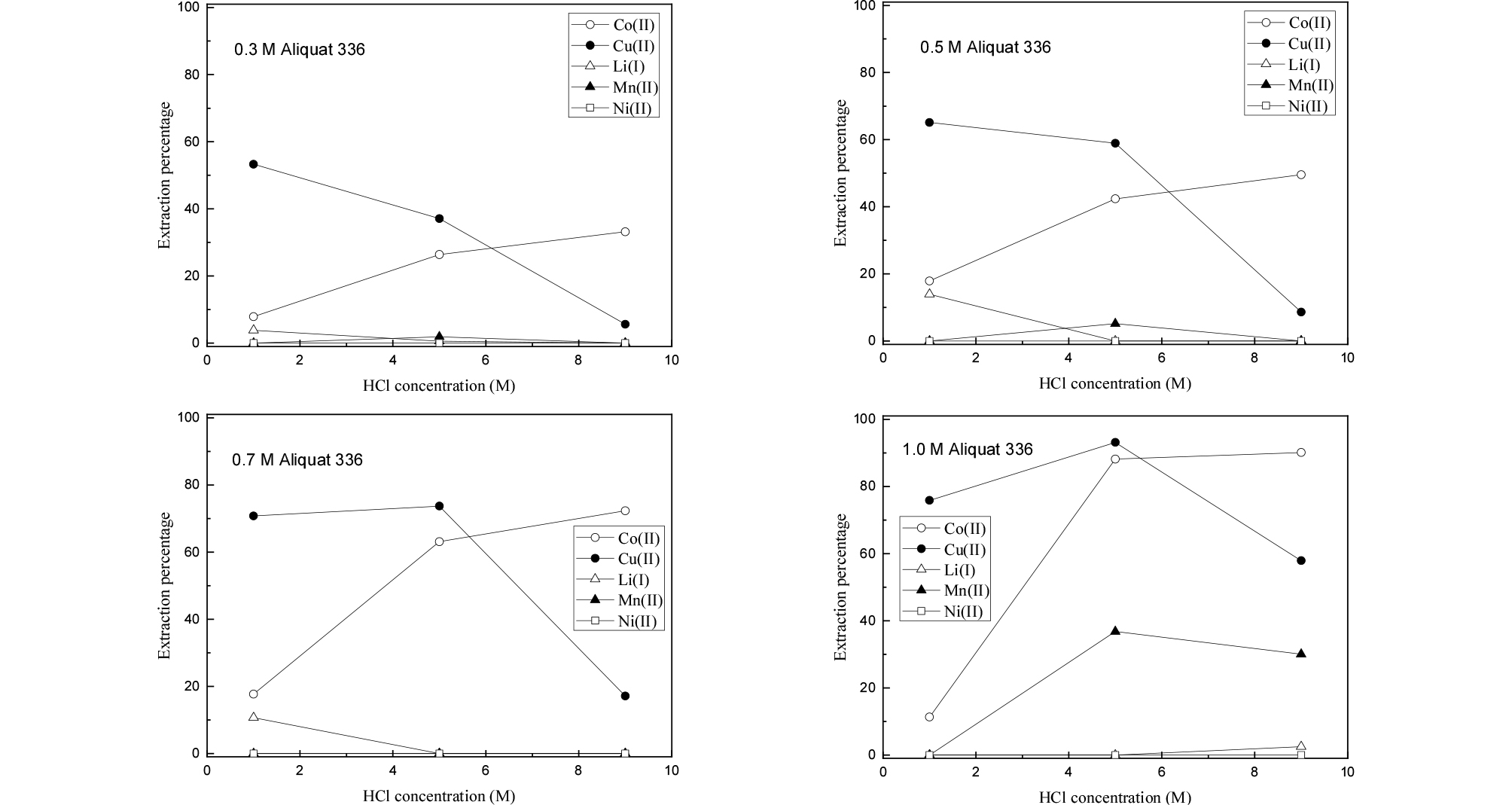
In hydrochloric acid solution, Co(II), Cu(II) and Mn(II) have a strong tendency to form complexes with chloride ion. In extraction with amines, the existence of anionic complexes and their valence state is very important. The nature of the complexes of these three metal ions depends on the concentration of chloride ion. Therefore, the effect of HCl concentration on the separation of the metals from the synthetic leaching solution was investigated by employing Alamine 336 and Aliquat 336.
3.1. Effect of HCl concentration
The concentration of HCl in the synthetic solution was varied from 1 to 9 M. The volume ratio of the two phases during extraction was kept at unity. Figs. 1 and 2 show the extraction results of the metals by Alamine 336 and Aliquat 336, respectively. As expected, Co(II) and Cu(II) were selectively extracted over the other metal ions by both extractants. This might be due to the formation of anionic complexes (CoCl3–, CoCl42–, CuCl42–) in HCl solution which can be extracted into the organic phase, while Li(I), Ni(II) and Mn(II) remained in the raffinate21,22,23,24). Mn(II) can form anionic species MnCl3- with chloride ion25). Since the concentration of Mn(II) in the solution was only 15 mg/L, its extraction percentage was low. The results also indicated that the extraction percentage of Co(II) increased as HCl concentration increased from 1 to 9 M by both extractants. Generally, Alamine 336 should be protonated in an acidic solution to act as an anion exchanger26). The protonation reaction of Alamine 336 is represented as Eq. (1). The extraction reaction of Co(II) by Alamine 336 and Aliquat 336 can be represented as Eqs. (2) and (3), respectively. The extraction efficiency of Co(II) by Aliquat 336 is always higher than Alamine 336 in our experimental ranges. The highest extraction percentage of Co(II) was 84% by 1 M Alamine 336 at 9 M HCl, while that by Aliquat 336 was 90%.
| $${\mathrm R}_3{\mathrm N}_{\mathrm{org}}\;+\;\mathrm{HCl}\;=\;{\mathrm R}_3{\mathrm{NHCl}}_{\mathrm{org}}$$ | (1) |
| $${\mathrm{CoCl}}_3^–\;+\;{\mathrm R}_3{\mathrm{NHCl}}_{\mathrm{org}}\;=\;{\mathrm{CoCl}}_3{\mathrm R}_3{\mathrm{NH}}_{\mathrm{org}}\;+\;\mathrm{Cl}^–$$ | (2) |
| $${\mathrm{CoCl}}_3^–\;+\;{\mathrm R}_4{\mathrm{NCl}}_{\mathrm{org}}\;=\;{\mathrm{CoCl}}_3{\mathrm R}_4{\mathrm N}_{\mathrm{org}}\;+\;\mathrm{Cl}^–$$ | (3) |
Unlike Co(II), the extraction of Cu(II) decreased with the increase in HCl concentration at low extractant concentrations (0.3-0.5 M). This may be ascribed to an insufficient concentration of extractants according to the molar ratio in the following reactions:
| $${\mathrm{MeCl}}_4{}^{2–}\;+\;2{\mathrm R}_3{\mathrm{NHCl}}_{\mathrm{org}}\;=\;{\mathrm{MeCl}}_4{({\mathrm R}_3\mathrm{NH})}_{2\;\mathrm{org}}+2\mathrm{Cl}^-$$ | (4) |
| $${\mathrm{MeCl}}_4^{2-}\;+\;2{\mathrm R}_4{\mathrm{NCl}}_{\mathrm{org}}\;=\;{\mathrm{MeCl}}_4{({\mathrm R}_4\mathrm N)}_{2\;\mathrm{org}}+2\mathrm{Cl}^-$$ | (5) |
where Me denotes both Co(II) and Cu(II).
At 9 M HCl concentration, the competition between Co(II) and Cu(II) led to much reduced extraction percentage of Cu(II) by the two extractants (see Eqs. (2-5)). Our results are in good agreement with previous data27).
3.2. Effect of extractant concentration
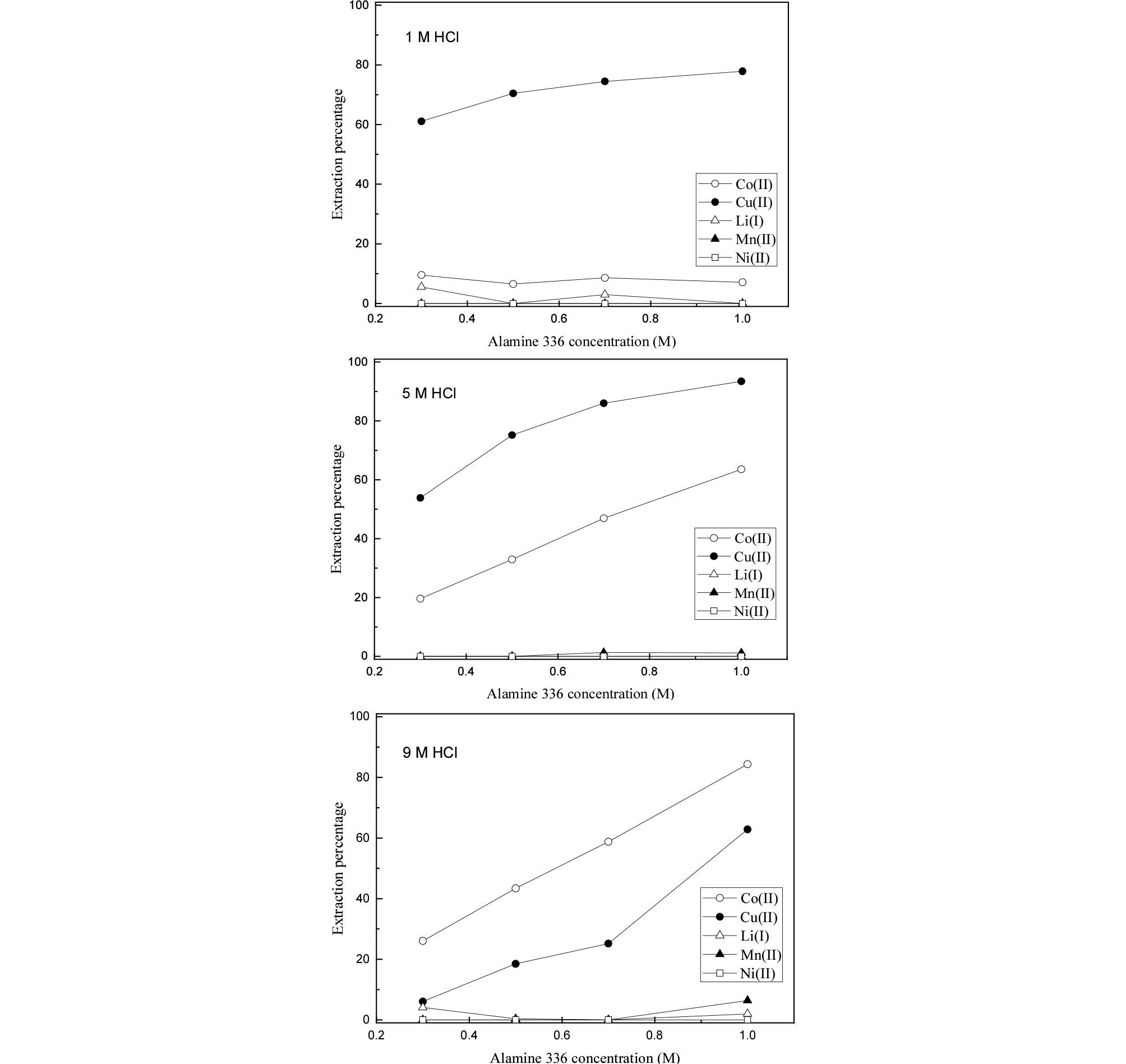
The effect of extractant concentration on the extraction of metals was investigated by varying Alamine 336 and Aliquat 336 concentration from 0.3 to 1.0 M at 1, 5, and 9 M HCl solution. The obtained results on the influence of Alamine 336 and Aliquat 336 concentration are shown in Figs. 3 and 4, respectively. It can be observed in Fig. 3 that the extraction percentage of Cu(II) increased with increasing Alamine 336 concentration from 0.3 to 1.0 M at all HCl solution. Cu(II) was selectively extracted over Co(II), Li(I), Mn(II), and Ni(II) by Alamine 336 at 1 M HCl concentration. At 1 M HCl solution, Co(II) cannot form anionic species with chloride ion and thus it would not be extracted into amines, which are in good agreement with the results. At 5 and 9 M HCl solution, the co-extraction of Cu(II) and Co(II) was observed, while the extraction percentage of Li(I), Mn(II) and Ni(II) was very low in the extractant concentration range. Owing to the existence of Li(I) and Ni(II) as cationic form in strong hydrochloric acid solution, these metal ions are not extracted by amine extractants in our experiments.
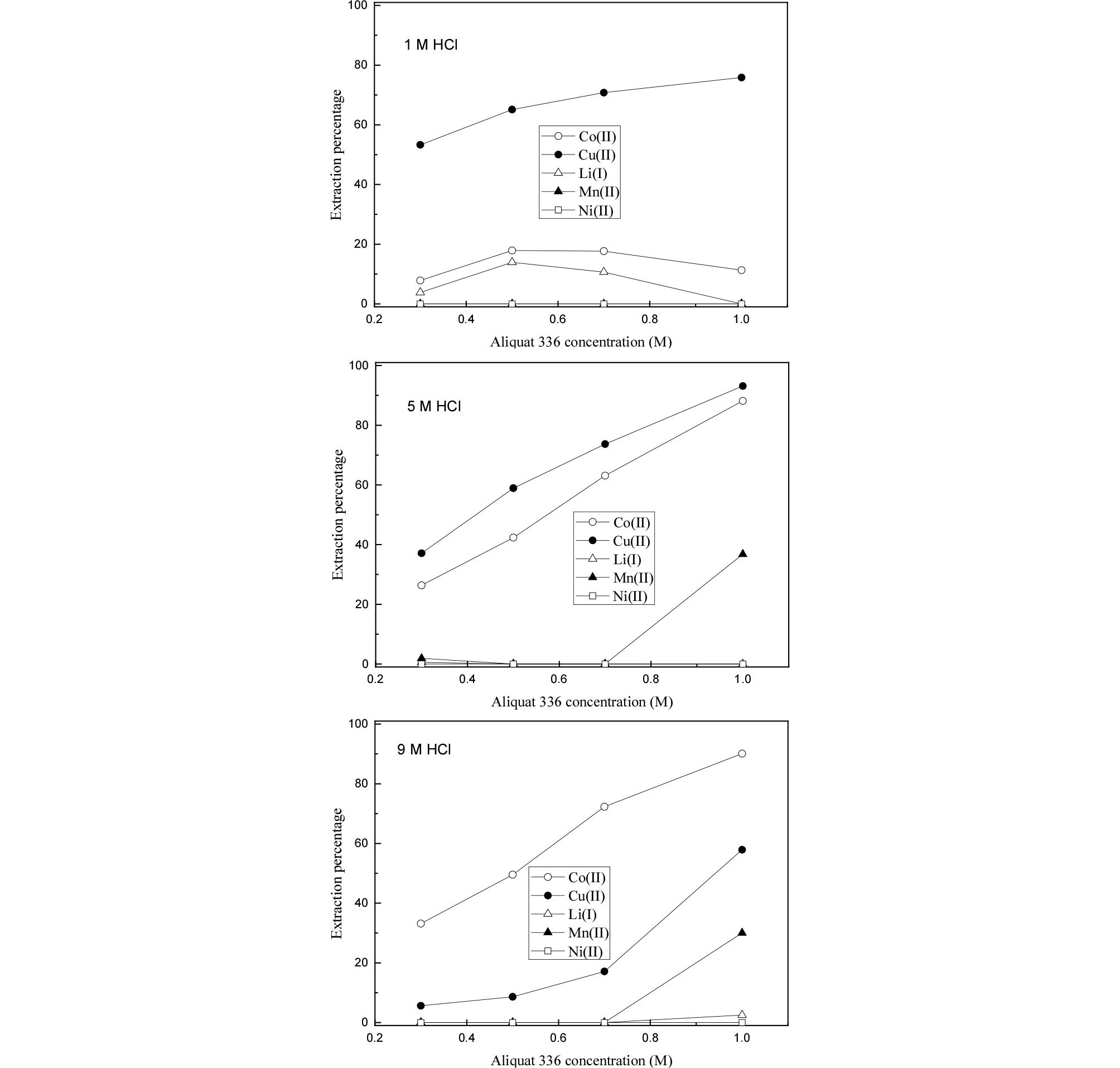
In the case of Aliquat 336, the extraction behavior of metals is almost similar to that of Alamine 336 (see Fig. 4). The difference occurred when 1 M Aliquat 336 was employed to the solution with 5 and 9 M HCl concentration. Namely, about 30-36% of Mn(II) was extracted into the Aliquat 336. Since anionic complexes of Mn(II) like MnCl3- can form in strong HCl solution, the anionic complex of Mn(II) can be extracted by amine extractants. However, our results showed that Mn(II) was not extracted by Alamine 336, while some of Mn(II) was extracted by Alamine 336 at 5 and 9 M HCl solution. The difference between Alamine 336 and Aliquat 336 as an anion exchanger lies in the fact that Alamine 336 needs to be protonated before extraction. At least 1 mole of HCl is needed to protonate 1 mole of Alamine 336. Therefore, the real concentration of HCl in the aqueous phase might be reduced after being contacted to the Alamine 336. However, this decrease in HCl concentration cannot explain the negligible extraction of Mn(II) by Alamine 336. Considering the low concentration of Mn(II) in the solution, there might be some difference in the chemical reactivity for Mn(II) between Alamine 336 and Aliquat 336.
3.3. Comparison of extraction performance between Alamine 336 and Aliquat 336
Comparison of Figs. 3 and 4 indicates that the separation of the metals by extraction with Alamine 336 and Aliquat 336 was almost similar to each other due to the amine group in their structure. However, Alamine 336 was better than Aliquat 336 in terms of metal separation efficiency. Cu(II) as well as Co(II) can be selectively extracted by Alamine 336 from 5 and 9 M HCl solution. Moreover, only Cu(II) can be extracted by Alamine 336 from 1 M HCl solution, while Co(II) is co-extracted with Cu(II) by Aliquat 336. Besides, Mn(II) was extracted by Aliquat 336 when the concentration of HCl in the solution was 5 and 9 M. Compared to the results by Alamine 336, the coexistence of Co(II), Cu(II), and Mn(II) in Aliquat 336 results in some difficulties in separating the 3 metals by stripping. Therefore, Alamine 336 is considered to be an appropriate extractant for the extraction of metals from the hydrochloric acid leaching solutions of spent Li-ion batteries. In addition, control of the acidity of the solution can lead to selective separation of the desired metal. The purification of Cu(II) at low acid media as well as the separation of Co(II) and Cu(II) in high acid concentration by Alamine 336 should be investigated in further works.
Our results confirmed that employment of Alamine 336 at strong HCl solution could result in selective extraction of Co(II) and Cu(II) from the HCl leaching solution of LiBs. Then, Li(I), Mn(II) and Ni(II) would remain in the raffinate. Therefore, the separation possibility of these three metal ions should be investigated from dilute HCl solution.
4. Conclusions
The spent LiBs contain some valuable metals which deserve to be recovered. The present study focused on the separation of cobalt(II) and copper(II) from the hydrochloric acid leaching solution of spent LiBs containing Li(I), Mn(II) and Ni(II). In this work, synthetic leaching solutions of spent LiBs were employed for aqueous phase, while commercial amines like Alamine 336 and Aliquat 336 were used as organic phase. The effect of hydrochloric acid and amine concentrations on the extraction and separation was investigated. The results showed that Cu(II) was selectively extracted by the two amines at 1 M HCl solution, while both Cu(II) and Co(II) were extracted as the HCl concentration increased from 5 to 9 M. The highest extraction percentage of metals was attained as follows: 90% for Co(II) at 9 M HCl by 1 M Aliquat 336; 93% for Cu(II) at 5 M HCl by 1 M Aliquat 336. The competition for the extraction between Co(II) and Cu(II) from concentrated HCl solution led to the difference in the extraction efficiency of both metals. Alamine 336 was better than Aliquat 336 in separating Co(II) and Cu(II) from the solution.