1. Introduction
2. Experimental
2.1. Materials and Method
2.2. Analysis
3. Results
3.1. Time experiment
3.2. pH and extractant concentration optimization
3.3. Extraction isotherms of tungsten and vanadium
4. Conclusion
1. Introduction
In the last decades, various secondary wastes has been used as source of different metals due to the rapid depletion of ores and the increased interest in green alternatives to supply the increasing demand of the same1,2). Among those sources, sludges, spent catalysts, scraps and e-waste have played a major role in the supply chain of strategic as well as base metals such as vanadium and tungsten3-6). Coupled with the green technology approach as well as the stricter regulations for emissions in stationary and mobile sources, selective catalytic reduction (SCR) has been proven as the primary method used for the reduction of nitrogen oxides (NOx) emitted to the environment causing air pollution7). Currently, the most used SCR catalyst for stationary and vehicle application is the V2O5-WO3 in TiO2 matrix which can contain around 0.5-1% wt V2O5 and 7-10% wt of WO38-11). Even tough, this catalyst offers great durability and regeneration capacity it has a limited chemical lifespan (roughly 3-6 years), after which it will end up most probably in landfills, becoming a major source of contamination, due to the toxicity of vanadium compounds8,9). The increasing demand of SCR catalyst has created a big niche for secondary resources of vanadium and tungsten, which could be recovered and reutilized.
During a previous study conducted in our research laboratory, the selective leaching of titanium was conducted, while leaving a soda roasted liquor containing approximately 7000mg/L of tungsten and 600mg/L of vanadium, which cannot be easily separated due to their similar chemical and physical properties9,12-14).
In the present work, leach liquor obtained from SCR spent catalyst is used as the source of vanadium and tungsten recovery using solvent extraction. Various authors have reported different hydrometallurgical routes to extract vanadium and tungsten from different secondary sources, however there is no available study that successfully extracts and enriches these metals from a high concentration liquor. In addition, the extractant used is a quaternary amine, the commercial extractant Aliquat 336 which is well known as a green ionic liquid (IL) reported for the extraction of several base and strategic metals15,16). Based on this study it would be possible to determine the optimum parameters for both metal enrichment and recovery.
2. Experimental
2.1. Materials and Method
The leach liquor used for this study was provided from a previous work of our research group9), which was made using a honeycomb spent catalyst from the Samcheonpo thermoelectric power plant in South Korea and contains around 600mg/L of vanadium and 7000mg/L of tungsten. For the extraction experiments, Aldrich Aliquat 336 was used as received and Exxol D80 (non aromatic) was used as a diluent for every experiment and taken without further purification. The solvent extraction investigation was carried out in a 125 ml volumetric flask mounted over incubated Shaker (SI-300/300R/600/600R). In each of the experimental study, equi-volume (unless otherwise specified) of aqueous and organic solution was contacted for 30 minutes to attain the equilibrium. After the phase disengagement, either of the phases were separated out and read for determination of the W/V concentration. The aqueous phase was analyzed by ICP-OES after making suitable dilution. All the experiments are performed under ambient temperature (±25°C). The extraction efficiency was determined based upon the equations as follows.
| $$D=\frac{\lbrack M_{itorganic\;phase}\rbrack}{\lbrack M_{aqueous\;phase}\rbrack}$$ | (1) |
Where, M represents vanadium or tungsten.
| $$\%E=\frac{D\ast100}{D+1}$$ | (2) |
2.2. Analysis
Chemical analysis of the aqueous phase to determine the content of vanadium and tungsten (before and after extraction) was conducted using an inductively coupled plasma optical emission spectrometer (ICP-OES, iCAP Duo6300, Thermo Scientific, UK).
3. Results
3.1. Time experiment
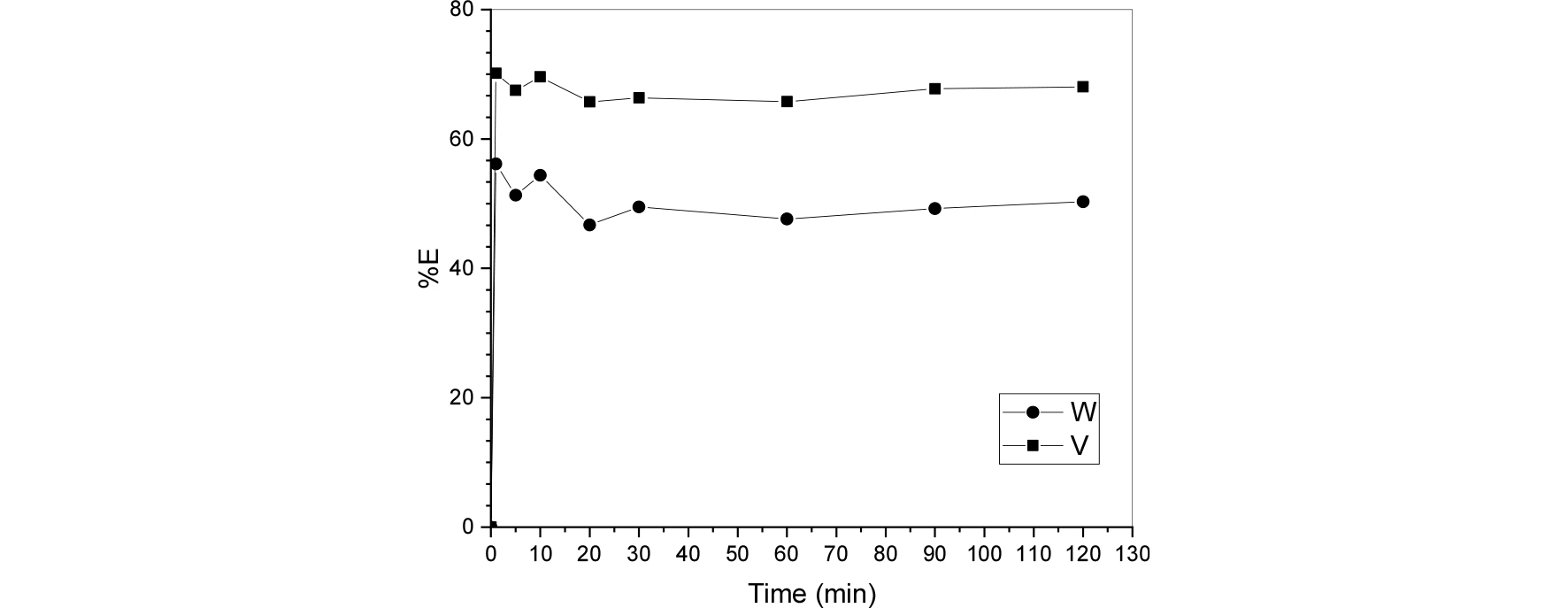
The pH of leach liquor used for the extraction was adjusted using hydrochloric acid until initial pH 7 and contacted in 125ml separating funnels with the same volume of the organic phase composed of 20ml of aliquat 336 0.5mol/L. The contact times were varied from 1 min to 120min to determine when the extraction equilibrium is attained.
Fig. 1 shows how the percentage of the extraction changes in time and it is observed that around 30min the percentage of extraction reaches a plateau zone were increasing the time would have no influence on the extraction capability of the process. For further studies in this paper, 30 minutes will be used as the extraction time unless specified. As can be seen from the results, after 30min of contact time the extraction of vanadium reaches an approximate of 65% while tungsten reaches 44% of extraction.
3.2. pH and extractant concentration optimization
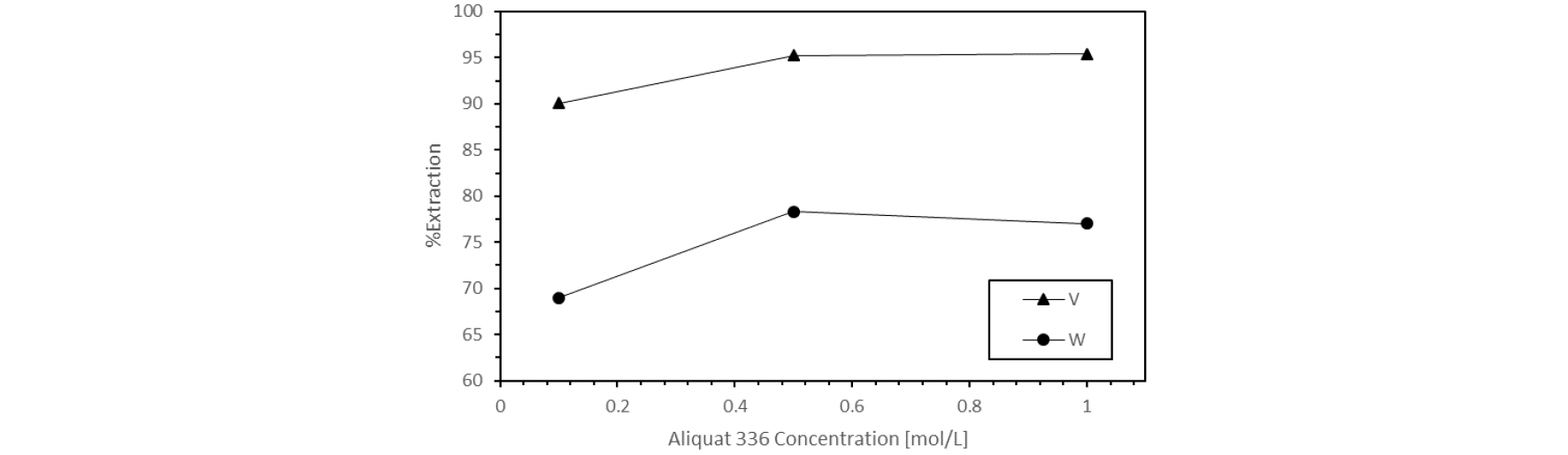
The effect of extractant concentration for in the vanadium and tungsten extract ion were investigated. For 20 ml aqueous samples (initial pH 7 adjusted with HCl), the concentration of extractant was varied from 0.1 mol/L to 1mol/L. The extraction was carried out using an organic/aqueous ratio of one for 30 min in a wrist shaker at 250rpm.
The extraction efficiency of vanadium and tungsten when aliquat 336 concentration is modified can be seen in Fig. 2. There is a steep increase on the efficiency when concentration of the extractant changes from 0.1mol/L to 0.5 mol/L, however there is no significant increase in the percentage of extraction above 0.5mol/L of extractant. Since the extraction trend reaches a plateau zone, it was determined that 0.5mol/L will be the concentration of aliquat 336 used for further experiments.
In addition, the distribution coefficient for vanadium and tungsten were studied and plotted against the aliquat 336 concentration (not shown) giving linear relationships with a slope close to the unity for both metals, which signifies the association of one mole of the extractant during the extraction process. Since, vanadium and tungsten offer a variety of anions in solution, it favors an anion exchange mechanism with aliquat 33617).
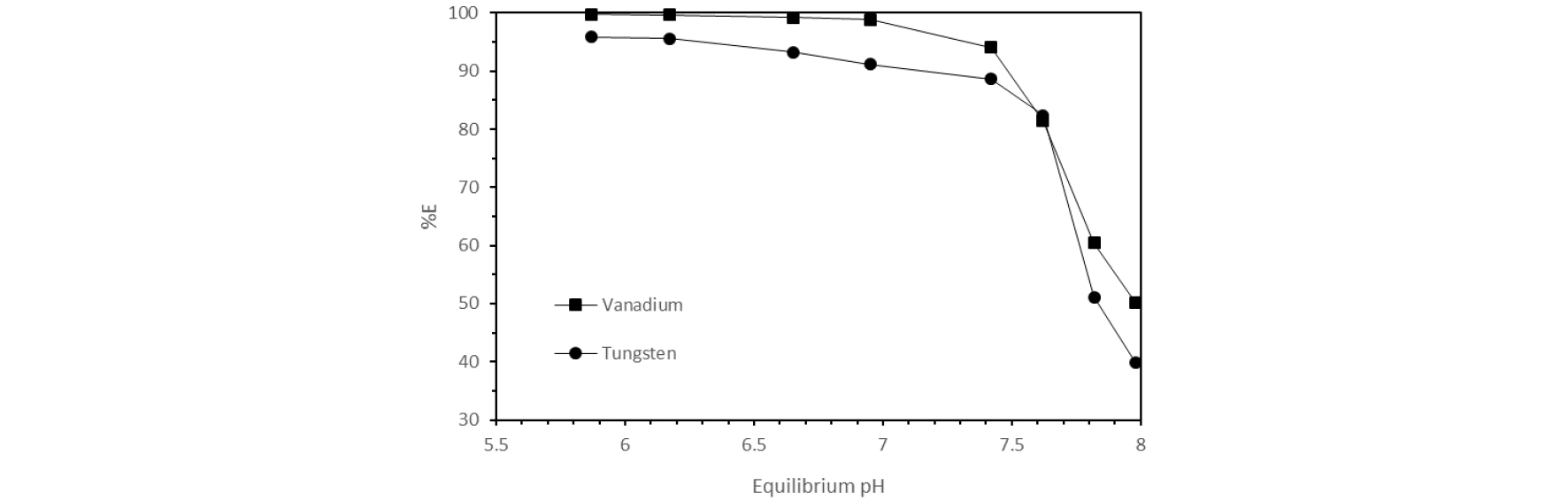
Equilibrium pH is one of the most important parameters for solvent extraction because it determines the species present in the aqueous phase and the nature of the species will determine if Aliquat 336 can extract them18). The pH of the leach liquor aqueous solution was varied using HCl from an initial pH of 5.0 to 7.35, while keeping the organic phase constant using Aliquat 336 (A/O=1) in D80 at a concentration of 0.5mol/L during 30 min in a wrist shaker at 250rpm.
Fig. 3 shows that increasing the pH towards an alkaline solution decreases the extraction for both metals and in slightly acid solutions (equilibrium pH ~6) 95% of extraction of vanadium and tungsten can be achieved. In addition, it can be observed that the equilibrium pH (plotted in Fig. 3) is higher than the initial pH for all the experiments done, which indicates the complexation of the whole vanadium or tungsten molecule without the release of any H+ leading to the increase of the pH of the aqueous phase18,19).
Finally, the distribution coefficient for vanadium and tungsten were plotted against the equilibrium pH (not shown) giving linear relationships with a slope close to the unity for both metals, which signifies the association of one proton towards the organic phase. From literature is well know that in slightly acidic media vanadium and tungsten can be present as a series of anionic species18) including HV2O5- and HWO4- which helped for the elucidation of the extraction mechanism as follows.
| $$HWO_{4\lbrack aq\rbrack}^-+CH_3R_3N^+C{l^-}_{\lbrack org\rbrack}\leftrightarrow CH_3R_3N\cdot HWO_{4\lbrack org\rbrack}+Cl_{\;\;\;\lbrack aq\rbrack}^-$$ | (4) |
3.3. Extraction isotherms of tungsten and vanadium
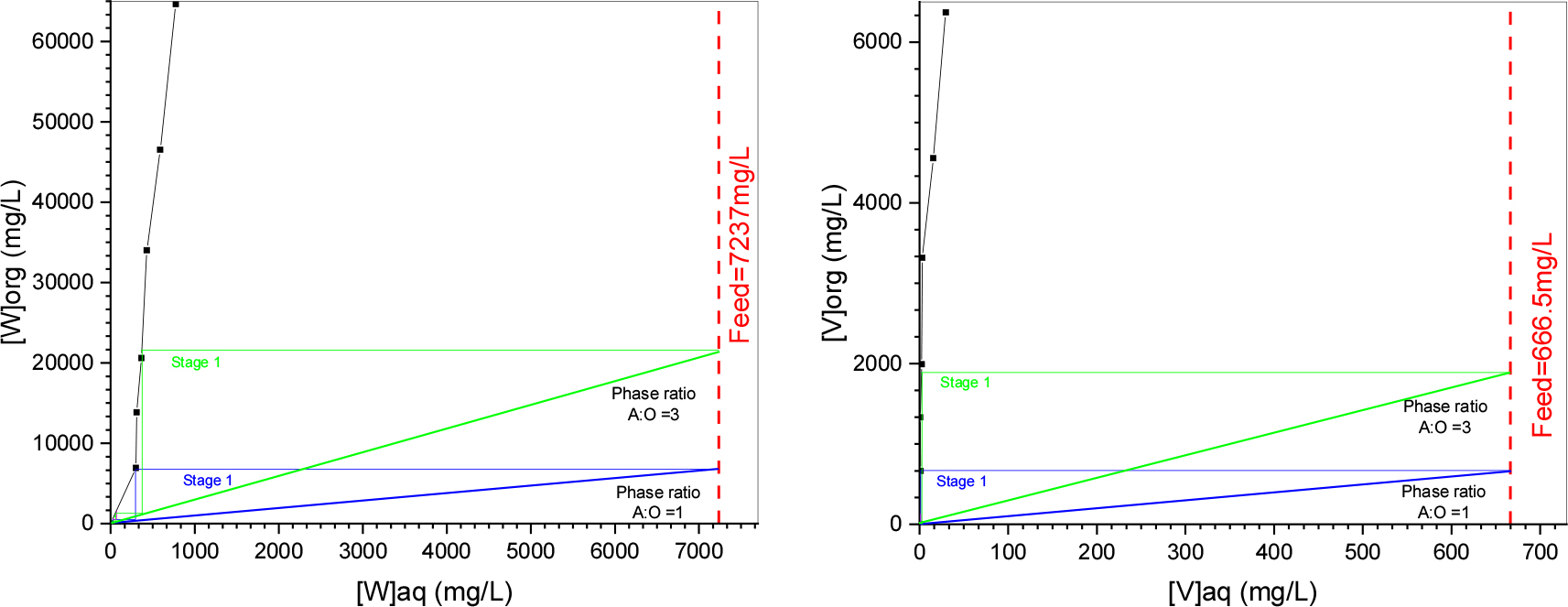
To find out the number of stages and possibility of enriching vanadium and tungsten in the loaded organic phase during extraction, the extraction isotherm for both metals was obtained by contact of the aqueous feed at pH 5 with Aliquat 336 0.5 M at different A/O ratios from 1 to 10 (Fig. 4). From the extraction isotherms it can be observed that for tungsten at a phase ratio A/O=3 almost complete extraction can be done in two-stages, while for vanadium at the same phase ratio a complete loading of the organic phase can be achieved at one stage.
To confirm, the McCabe-Thiele diagram theoretical predictions, counter current extractions were performed at an A/O = 3. The results showed a raffinate containing 0.23mg/L of vanadium and 270.2mg/L of tungsten, which corresponds to a 99.95% and 96.07% extraction of vanadium and tungsten. To increase the percentage of extraction for the process, a three-stage counter current extraction was tested and it resulted in a 99.98% extraction of vanadium and 99.11% extraction of tungsten.
4. Conclusion
The extraction of vanadium and tungsten from SCR spent catalyst leach liquor using the quaternary ammonia salt Aliquat 336 in Exxol D80 was studied. The main parameters that affect extraction are time, concentration of extractant and equilibrium pH in the solution. It can be seen that 30 minutes is the optimum time for extraction and was used during the present study. In addition, the equilibrium pH was analyzed in the range of slightly acid (pH~6) to slightly basic (pH~8) and it was determined that slight acidity maximizes the extraction percentage for both metals. Furthermore, increasing the extractant concentration more than 0.5mol/L does not offer a great advantage for the extraction of the desired valuable metals, reason why it was chosen as the concentration used during the investigation. The extraction isotherms for vanadium and tungsten were determined and for an A/O phase ratio of 3 it can be seen that one stage is enough to reach 99% of extraction in the case of vanadium, however for tungsten at least 2 stages are necessary to reach the desired loading. Counter current extraction tests were performed and 99.9% vanadium and 99% of tungsten were extracted in a three stage counter current process.
In summary, the use of Aliquat 336 in D80 at slightly acidic conditions is an effective mean to extract and recover vanadium and tungsten from SCR spent catalyst. Further studies in stripping are necessary for a total understanding of the extraction and recovery system.