1. Introduction
2. Experimental
2.1. Reagents and Chemicals
2.2. Solvent extraction procedure and analytical methods
3. Results and Discussion
3.1. Removal of Fe(III) from the synthetic sulfate leaching solutions
3.2. Separation of Cu(II) from the Fe(III) free raffinate
3.3. Modified process
4. Conclusions
1. Introduction
Lithium-ion battery (LIB) is a preferred choice for portable electronics and electric vehicles owing to its merits such as high energy density, low maintenance, low self-discharge, and quick charging1). Lots of spent LIBs are accumulating, which could do harm to public health and the environment2). Spent LIBs contain valuable metals such as cobalt, nickel, lithium, and manganese and thus recycling of spent LIBs is a solution to the sustainable production of the LIBs as well as to minimize potential environmental impacts3).
The recovery of valuable metals from spent LIBs mainly involves mechanical, mechanochemical, and chemical processes4,5,6,7). Among these processes, pyrometallurgical and hydrometallurgical methods have been widely used. In hydrometallurgy, metal ions such as Co, Ni, Li, and Mn are leached by acidic solutions in the presence or absence of oxidizing agents. The leaching solutions are treated by precipitation, solvent extraction, and ion exchange for the separation of metal ions8,9,10,11,12,13,14,15,16). In general, the separation of metal ions from the leachate depends on the composition of the leaching solution and the nature of the metal ions. Especially, the presence of Fe(III) in the leaching solution poses some problems in separating the metal ions.
The application of pyrometallurgical methods has many advantages in the recovery efficiency of Co, Ni, and Cu like a decrease in the labor cost for classifying and dismantling organic matters like plastics from the spent LIBs17,18,19,20). Besides, the reduction of metal ions in the spent LIBs at high-temperature results in the formation of metallic alloys and slags containing Li and Al, which can facilitate the selective recovery of the valuable metals.
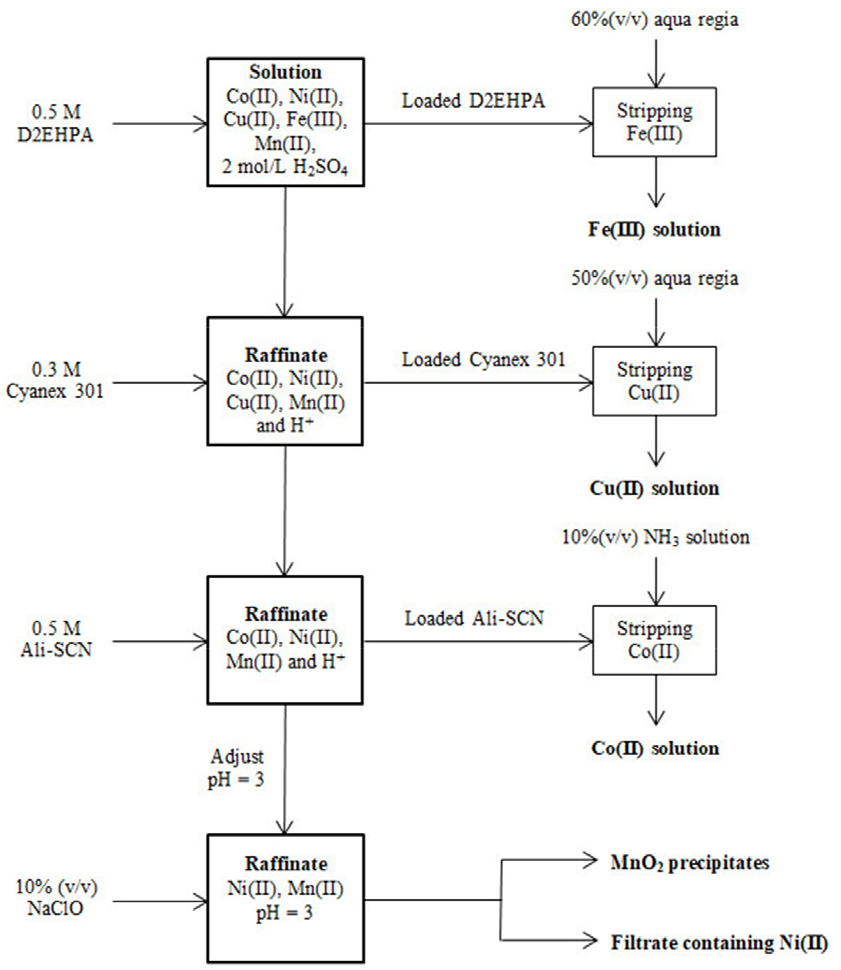
In our previous work, a combined process of pyrometallurgy and hydrometallurgy for the recovery of Co, Ni, Cu, Fe, and Mn from spent LIBs was reported21). First, the reduction smelting of spent LIBs at high-temperature results in metallic alloys containing Co, Ni, Cu, Mn, Fe, and Si. The metals from the alloys were completely leached except Si by the mixture of 2.0 M H2SO4 and 10% H2O2. The separation of metal ions such as Co(II), Ni(II), Cu(II), Mn(II), and Fe(III) from the sulfate leaching solution was carried out by solvent extraction with ionic liquids and oxidative precipitation (see Fig. 1). In this process, Co(II) and Fe(III) were co-extracted by ALi-SCN, a kind of IL and Fe(III) was separated from the loaded organic by precipitation stripping with NH3. Although the separation efficiency of this solvent extraction with ALi-SCN was high, precipitation stripping is difficult to run continuously in real operation. Moreover, the application of ILs to real industrial operations is limited due to their price and availability.
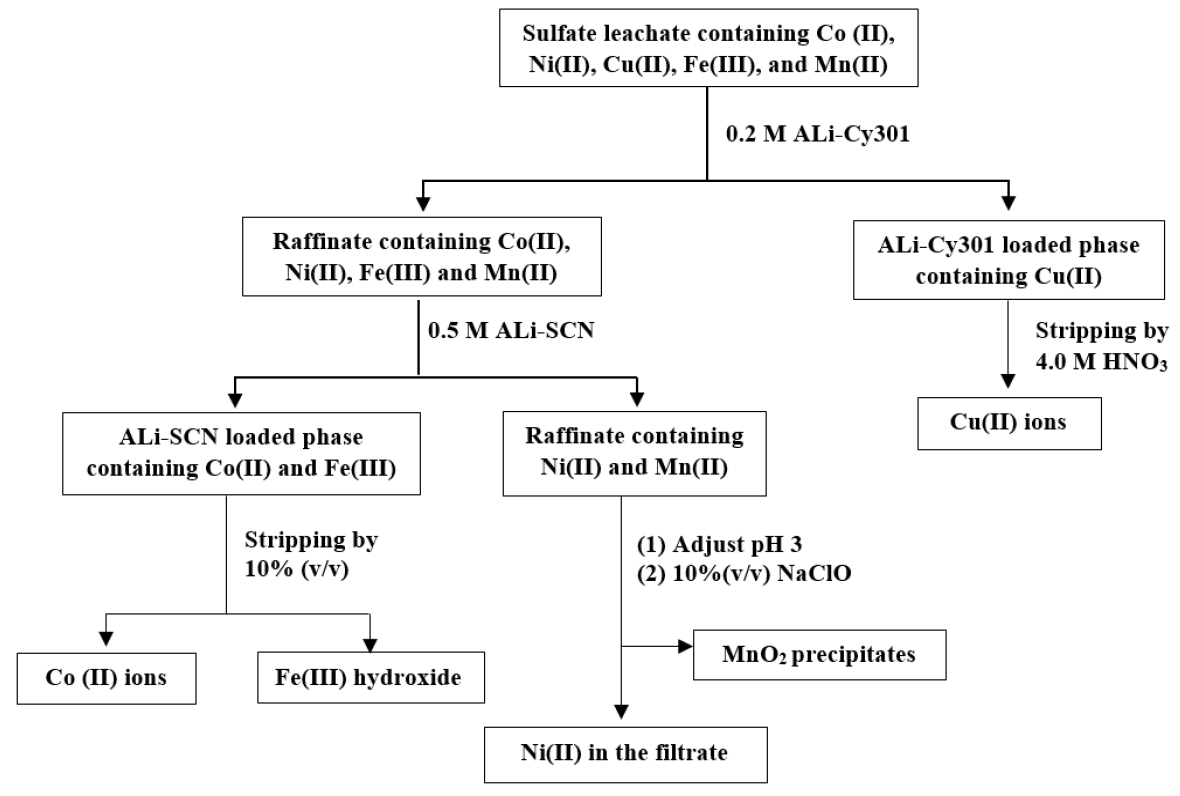
Fig. 1.
A flow sheet for the recovery of Co(II), Ni(II), Cu(II), Fe(III), and Mn(II) from the spent LIBs21).
In order to overcome the above-mentioned disadvantages of our proposed process, commercial extractants instead of ILs were employed in this work for the selective removal of Fe(III) and Cu(II) over Co(II), Ni(II), and Mn(II) from the sulfate leachate. For this purpose, D2EHPA and Cyanex 301 were employed for the selective extraction of Fe(III) and Cu(II) from the leaching solution. Stripping of Fe(III) and Cu(II) from the loaded phases was done by using aqua regia. The optimum conditions for the extraction and stripping were obtained. From the obtained results, a modified process was proposed.
2. Experimental
2.1. Reagents and Chemicals
The synthetic leaching solution containing Co(II), Ni(II), Cu(II), Fe(III), Mn(II), and Si(IV) was prepared by the dissolution of a certain amount of sulfate salts of the respective metals, such as CoSO4⋅7H2O (Daejung Co., >99%), NiSO4⋅6H2O (Daejung Co., > 99%), CuSO4⋅5H2O (Duksan Co. Japan, > 99%), Fe2(SO4)3⋅nH2O (Kanto Co., 60% – 80%), MnSO4⋅4H2O (Duksan Co. Japan, > 99%), Na2SiO3 solution (Daejung Co., > 99%) in 2.0 M H2SO4 solution. The concentration of metal ions in the synthesis leaching solution is represented in Table 1.
Table 1.
The concentration of metal ions in synthetic sulfuric acid leaching solutions of reduction smelted metallic alloys (2.0 M H2SO4 solution)
| Metal ions | Co(II) | Ni(II) | Cu(II) | Fe(III) | Mn(II) | Si(IV) |
| mg/L | 3229.0 | 15498.0 | 5584.2 | 125.9 | 395.3 | 82.4 |
Sodium hydroxide (Duksan Co., >99%) was used to adjust the pH value of the solution. Solutions of HCl (Daejung Co., 35%), HNO3 (Daejung Co., 60%), H2SO4 (Daejung Co., 95%), NH4OH (Junsei Chemical Co. Ltd, Japan, 28%), H2O2 (Daejung Co., > 30%), and NaClO (Junsei Chemical Co. Ltd, Japan, >99%) were diluted with doubly distilled water to desired concentrations. Organic extractants such as D2EHPA (di-2-ethylhexyl-phosphoric acid, Cytec Inc., 95%), Cyanex 301 (bis(2,4,4-trimethylpentyl)dithiophosphinic acid, Cytec Inc, 70%), and Aliquat 336 (N-methyl-N,N,N-trioctylammonium chloride, BASF Co., 93%) were employed without further purification. Commercial grade kerosene (Daejung Co., > 90%) was used as a diluent organic phase, while 1-decanol (Daejung Co., > 98%) was added as a modifier to prevent a third phase formation.
Preparation of 1.0 M ALi-SCN: 1.0 M Aliquat 336 in kerosene was contacted to 1.6 M NH4SCN (Daejung Co., 99.0%) aqueous solution several times to form an IL like ALi-SCN. AgNO3 solution was used to test the complete transfer of chloride ions from the organic phase to the aqueous phase.
2.2. Solvent extraction procedure and analytical methods
Extraction and stripping experiments were performed by mixing equal volumes of organic and aqueous phases (each 20 mL) in a screwed cap bottle. The mixture was shaken by a Burrell wrist action shaker (model 75, USA) for 30 minutes at room temperature (25 ± 1°C). The separation of phases from the shaken solutions was obtained by a glass separatory funnel. The concentration of metals in the aqueous phase was determined by ICP-OES (Inductively coupled plasma-optical emission spectrometry, Spectro Arcos) analysis. The extraction percentage (%E) was calculated as: , where mi and ma are the mass of a metal in the aqueous phases before and after the extraction. The stripping percentage of metal ions was calculated as: , where mo and ma* are the mass of a metal in the organic and aqueous phases before and after the stripping, respectively. The mass of metals in the aqueous and organic phases was obtained by ICP-OES measurement.
The concentration of hydrogen ions and pH of aqueous solutions was determined by volumetric titration methods22) and an Orion Star thermal scientific pH meter (model A221, USA). Fourier transform infrared (FT-IR, Vertex 80 V, Bruker, Germany) spectra were used to verify the effect of stripping agent like aqua regia on D2EHPA.
3. Results and Discussion
3.1. Removal of Fe(III) from the synthetic sulfate leaching solutions
Fe(III) extraction by D2EHPA
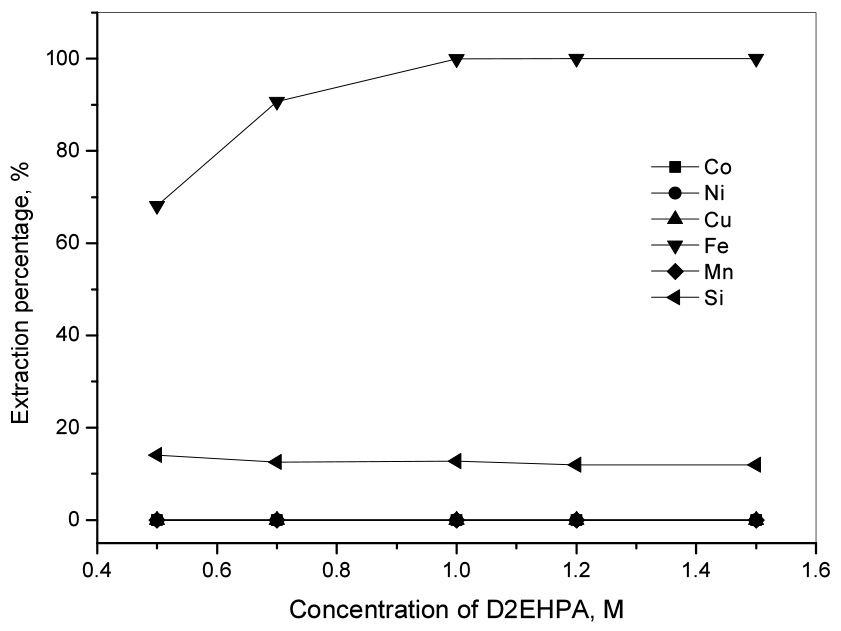
D2EHPA showed effective extraction ability for Fe(III) from acid solutions like HCl and H2SO4 compared to other commercial extractants such as PC88A and Cyanex 27223). Hence, in this work, D2EHPA was employed for the extraction of Fe(III) from the sulfate solution. To consider the effect of D2EHPA concentration on the selective extraction of Fe(III) over others, the concentration of D2EHPA was varied from 0.5 M to 1.5 M with an O/A ratio of unity. Fig. 2 shows that most of Fe(III) and 12.7% of Si(IV) were extracted by 1.0 M D2EHPA, while the extraction percentage of other metals was negligible in the range of studied D2EHPA concentration. Extraction reaction of Fe(III) by D2EHPA can be written as24)
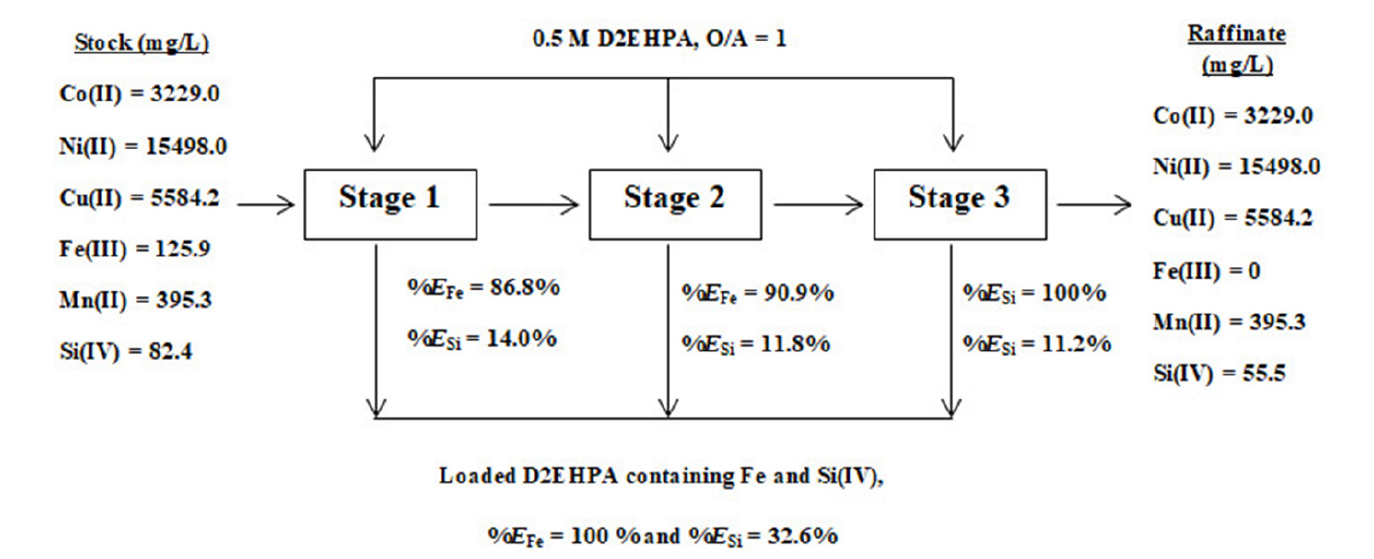
Since the stripping of concentrated Fe(III) from the loaded D2EHPA is difficult, 0.5 M D2EHPA was selected for the extraction of Fe(III) from the leaching solution25). To completely extract Fe(III) from the solution, three stages cross-current extraction by 0.5 M D2EHPA was done. The extraction percentage of Fe(III) and Si(IV) at each stage of extraction is displayed in Fig. 3. The obtained results indicated that the concentration of Fe(III) and Si(IV) after the three-stage cross-current was zero and 55.5 mg/L, corresponding to the extraction percentage of 100% and 32.6%, respectively. Meanwhile, the extraction percentage of other metal ions except for Fe(III) and Si(IV) was negligible.
Stripping of Fe(III) from the loaded D2EHPA
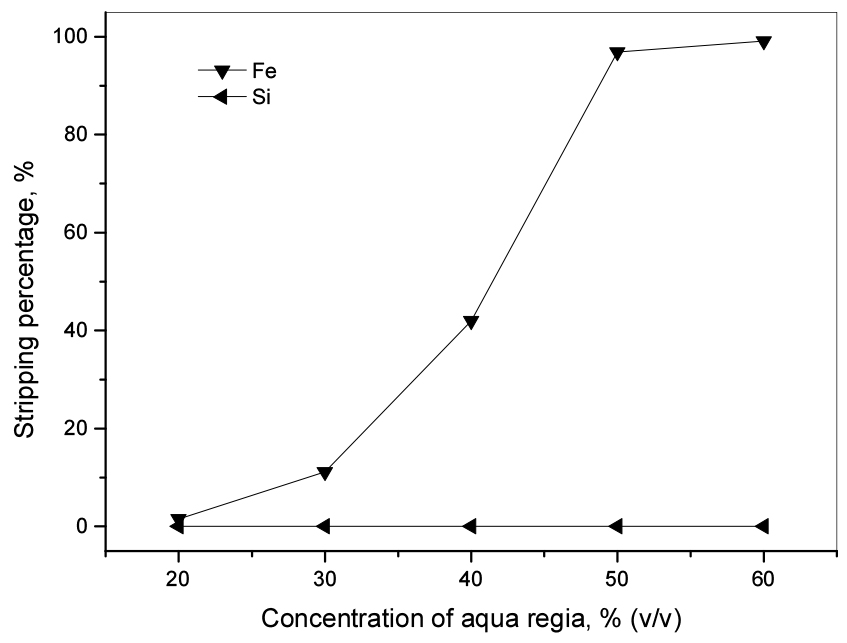
It has been reported that even concentrated inorganic acid solutions of H2SO4, HNO3, and HCl have some difficulty in stripping Fe(III) from the loaded D2EHPA. Although the use of reducing agents like Na2SO3 or H2SO3 can improve the stripping efficiency of Fe(III) from loaded D2EHPA, the complete stripping also requires multi-stages26). Therefore, in this work, aqua regia solution was tried for the stripping of Fe(III) from the loaded D2EHPA. The concentration of aqua regia in the stripping solution was varied from 20% (v/v). The loaded D2EHPA contained 125.9 mg/L Fe(III) and 26.9 mg/L Si(IV). In Fig. 4, the stripping percentage of Fe(III) from the loaded phase significantly increased with aqua regia concentration. Fe(III) was completely stripped from the loaded phase by 60% (v/v) aqua regia and the amount of Si(IV) in the stripping solution was negligible. The stripping reaction of Fe(III) from the loaded D2EHPA by aqua regia has not been manifest but might be proposed as
Thus, the selective removal of Fe(III) over others from the leaching solution was obtained by 0.5 M D2EHPA with three-stage cross-current extraction and stripping by 60% (v/v) aqua regia solution.
3.2. Separation of Cu(II) from the Fe(III) free raffinate
Cu(II) extraction by Cyanex 301
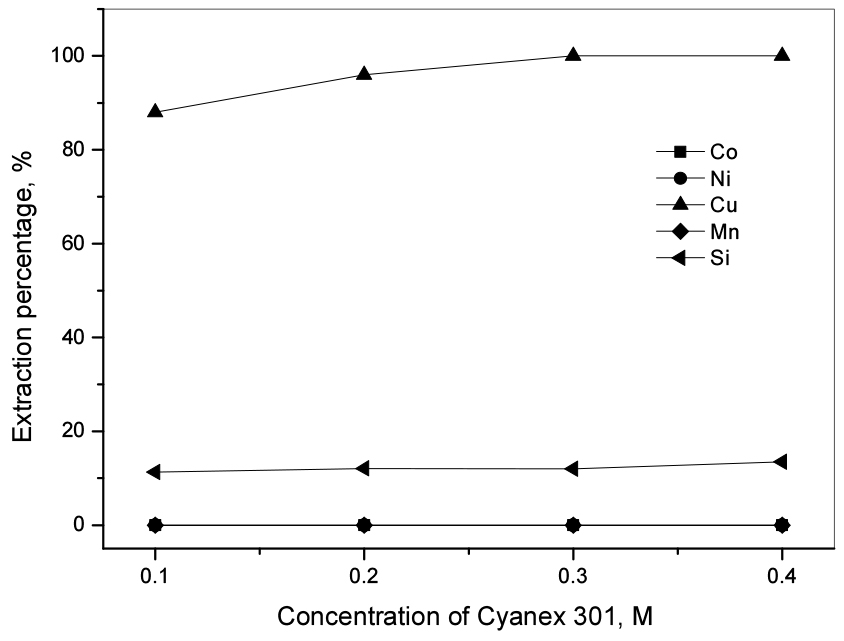
The concentration of metal ions in the free Fe(III) raffinate was 3229.0 mg/L Co(II), 15498.0 mg/L Ni(II), 5584.2 mg/L Cu(II), 395.3 mg/L Mn(II), and 55.5 mg/L Si(IV). According to the reported literature27), the use of Cyanex 301 showed the selective extraction ability for Cu(II) over Co(II), Ni(II), Mn(II), and Li(I) from the sulfate solution. Hence, in this work, Cyanex 301 was employed for the separation of Cu(II) over others from the leaching solution. The concentration of Cyanex 301 in kerosene was varied from 0.1 M to completely extract Cu(II). Fig. 5 shows that Cu(II) was completely extracted by 0.3 M Cyanex 301, while only 12.0% Si(IV) was extracted. The extraction percentage of Co(II), Ni(II), and Cu(II) were trivial in the range of studied Cyanex 301 concentration. These results can be ascribed to the strong interaction between Cu(II) and sulfur atoms of Cyanex 301 according to hard-soft acid-base theory (HSAB)28).
Stripping of Cu (II) from the loaded Cyanex 301
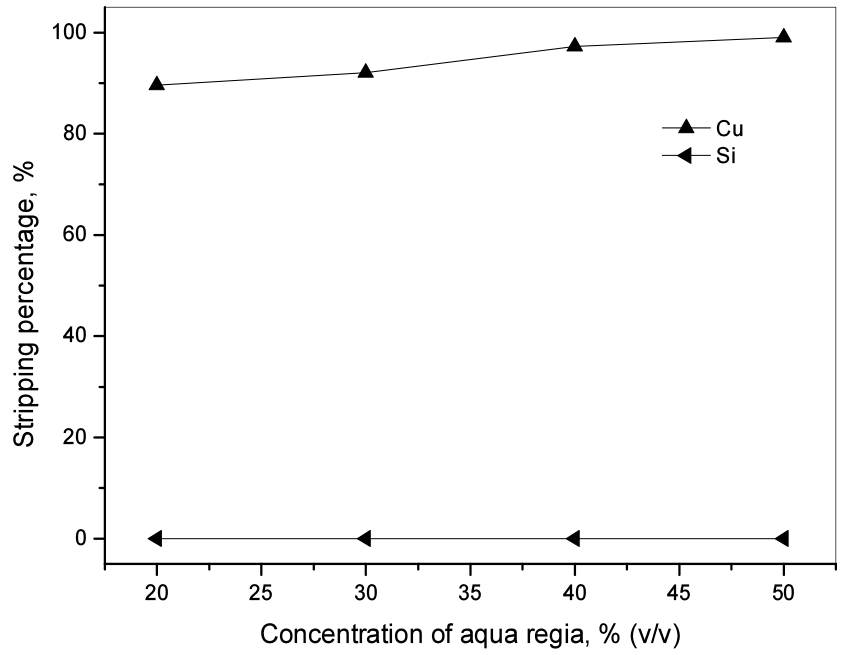
Due to the strong interaction between Cu(II) and Cyanex 301, the stripping of Cu(II) from the loaded Cyanex 301 is very difficult and thus use of aqua regia is needed29). Therefore, aqua regia solutions were employed for the stripping of Cu(II) and the concentration of aqua regia solutions were varied from 20 to 50% (v/v). The concentration of Cu(II) and Si(IV) in the loaded Cyanex 301 was 5584.2 mg/L and 6.7 mg/L. Our data showed that Cu(II) was completely stripped by 50% (v/v) aqua regia from the loaded Cyanex 301 (see Fig. 6). Meanwhile, Si(IV) was not stripped by aqua regia.
3.3. Modified process
After the separation of Cu(II) by extraction with Cyanex 301, the concentration of metals in the Cu(II) free raffinate was 3229.0 mg/L Co(II), 15498.0 mg/L Ni(II), 395.3 mg/L Mn(II), and 48.8 mg/L Si(IV). The concentration of hydrogen ions in the raffinate was determined to be 4.25 M. To verify the recovery of Co(II), Ni(II), and Mn(II) from the Fe(III) and Cu(II) free raffinate, separation experiments of Co(II), Ni(II) and Mn(II) was performed according to the process shown in Fig. 1. Selective and complete extraction of Co(II) was obtained by extraction with 0.5 M ALi-SCN and stripping with 10% (v/v) NH3 solution. The oxidative precipitation of Mn(II) as MnO2 from the raffinate at pH 3 was achieved by using NaOCl solution and Ni(II) and Si(IV) stayed the filtrate. These results agreed well with the reported process.
From the obtained results, a modified process for the recovery of metals from the sulfate leaching solution of metallic alloys resulted from reduction smelting of spent LIBs is shown in Fig. 7. First, Fe(III) and Cu(II) were sequentially extracted by D2EHPA and Cyanex 301 and then these metals were stripped by aqua regia solution. Next, Co(II) was recovered over Ni(II) and Mn(II) by extraction with ALi-SCN and stripped with 10% (v/v) NH3 solution. Finally, Mn(II) from the raffinate containing Ni(II) at pH 3 was precipitated as MnO2 by adding NaClO solution. The mass balance from the continuous experiments for the separation of metal ions from the sulfate leaching solution is represented in Table 2. The obtained results indicated that the recovery percentage of metals was higher than 99.0% with above 99.6% purity. In the reported study, ALi-Cy301 and ALi-SCN were used to remove Cu and Fe. The application of these ILs to practical industrial operations is limited due to their price and availability. In addition, the precipitation stripping of Fe(III) from the loaded ALi-SCN is difficult to run continuously in actual operation. Instead, D2EHPA and Cyanex 301 were commercial extractants and showed high efficiency in removing Fe(III) and Cu(II). This process has some advantages compared to the reported process in terms of the use of commercial extractants for the complete extraction of less valuable metals like Fe(III) and Cu(II).
Table 2.
Mass balance from the continuous experiments for the separation of metal ions from the 2.0 M H2SO4 solution
4. Conclusions
The selective separation of Fe(III) and Cu(II) over Co(II), Ni(II), and Mn(II) from the synthetic sulfate leaching solution of metallic alloys of reduction smelted spent LIBs by commercial extractants such as D2EHPA and Cyanex 301 was studied. First, Fe(III) was completely extracted over others by 0.5 M D2EHPA with three stages cross-current at an O/A ratio of unity. Then, Cu(II) was selectively extracted from the Fe(III) free raffinate by 0.3 M Cyanex 301. The complete stripping of Fe(III) and Cu(II) from the loaded phases were obtained by aqua regia solutions. A modified process for the separation of Co(II), Ni(II), Cu(II), Fe(III), and Mn(II) from the sulfate leaching solution was proposed. From the mass balance results, this process indicated that the recovery and purity percentage of metals from the sulfate solution was higher than 99%.