1. Introduction
2. Experimental Section
2.1. Materials
2.2. Methods
3. Result and Discussion
3.1. Effect of pH
3.2. Effect of contact time
3.3. Effect of adsorbent dose
3.4. Effect of aqueous feed concentration
3.5. Loading capacity of adsorbents
3.6. Column studies
3.7. Kinetics of adsorption
3.8. Adsorption isotherm
4. Conclusions
1. Introduction
Metals are essentially utilized in modern society and have significant role in industrial processing due to its unique properties. Recent technological innovations and its swift commercialization increased the demand of metals in industrial application. Especially copper is one of the essential metals used in electronic industry due to higher conductivity and less resistance property1). Natural resources of these metals are getting depleted due to its high demand and limited availability. Despite of strict environmental regulations, effluent generated from these industries contains large amount of heavy metals including copper, which gets discharged in to water bodies directly or indirectly2). This creates water pollution, which depletes quality of water and shows harmful effect on human health and organism by bioaccumulation3). Consumption of copper above 5 ppm (According to WHO norms) can lead to several health issues like hepatotoxic and nephrotoxic effect, gastrointestinal diseases, hemolysis and ultimately Wilsons disease1). To overcome such issues, industrial effluent/ waste water treatment was performed for metal recovery.
Many authors have reported different techniques of effluent treatment viz. membrane filtration, coagulation- flocculation, chemical precipitation, electrochemical recovery, solvent extraction, ion–exchange, adsorption etc. But all these techniques attained some problems in cost of operation, sludge generation, time consuming, critical handling, etc. Amongst these techniques, adsorption method based on ion exchange technique found to be appropriate, cost effective, efficient method showing faster kinetics. This process can recover metals from dilute effluent in the range of parts per million4).
Many biological and synthetic adsorbents were investigated for copper reclamation from industrial effluents5,6,7,8). Bioadsorbent like Sunflower stalk, banana peel, neem leaf powder, cow bone charcoal was utilized for copper recovery from effluents. Cow bone charcoal and neem leaf powder had shown maximum adsorption potential i.e. 9615 and 185.58 mg/g, respectively9,10).
Datura (Datura inoxia) plant leaves were utilized for recovery of cadmium, chromium and lead from aqueous solution showing high adsorption potential11), Datura (Datura stramonium) fruit was utilized for chromium adsorption from waste water showing 44.34 mg/g adsorption capacity12). Datura (Datura stramonium) root powder selected for this study has still not been reported as an adsorbent for copper recovery from effluents. Synthetic adsorbent like Amberlite IR 120 Na, Purolite S 984, Lewatit MonoPlus TP 220, etc were studied. Amongst all Lewatit MonoPlus TP 220 had shown maximum adsorption i.e. 230 mg/g1,13), while Tulsion T-42 selected for this study, has also not been reported as an adsorbent for copper recovery from aqueous solutions.
Plant root adsorbents are environment friendly, cost effective, and easily available as compared to synthetic adsorbent which has different environment issues and challenges like disposal of adsorbent after it get poisoned14). Keeping in view of the above, the present investigation has been made to compare two adsorbent s i.e. Datura (Datura stramonium) root powder and synthetic Tulsion T-42 for the adsorption of copper in cost effective way. Various process parameters like adsorbent dose, pH, feed concentration, contact time, etc. have been optimized for maximum adsorption of copper. Based on these studies, process can be developed for copper recovery using Datura root powder for commercial exploitation after the scale up studies.
2. Experimental Section
2.1. Materials
Effluent having copper concentration of 50–250 ppm was taken for the studies. Synthetic solution of the effluent was prepared by dissolving estimated quantity of its sulfate salt in distilled water. Biological adsorbent Datura root powder was prepared in the laboratory by some initial treatment, while synthetic adsorbent Tulsion T-42 was supplied by Thermax, India. Laboratory grade chemicals like copper sulfate, hydrochloric acid, sulfuric acid, ammonium hydroxide were purchased from E. Merck, India for the use of experimental purpose.
2.2. Methods
2.2.1. Preparation of adsorbent
Datura plants were collected from the road side land/bushy areas; and the roots were separated from the plant. Collected root sample was first washed with tap water thoroughly to remove soil particles then with distilled water for complete cleaning. Further, roots were cut into small pieces and kept in an oven at 100°C until dry weight of roots become constant11). Then sample was crushed in to motor and pestle to get fine Datura root powder. This root powder was taken directly for adsorption studies without any further activation process, while Tulsion T-42 (polystyrene copolymer with active group –SO3H) was used for experimental purpose without any purification.
2.2.2. Adsorption studies
Batch experiments for the adsorption of copper from dilute solution/effluentwere carried out in 100 ml conical flask using Datura root powder or Tulsion T-42 varying different ratio of adsorbent and feed solution at room temperature. Mixing of the solution was done by using wrist action shaking machine. Different process parameters such as adsorbent dose, feed concentration, pH, etc. were standardised for maximum adsorption. The pH was varied in the range of 1 to 6 by using diluted ammonium hydroxide or diluted sulfuric acid.
Column experiments were performed to study adsorption process in continuous mode using cylindrical column having dimensions 50 cm of height and 2 cm of diameter. Column adsorption experiments using Datura root powder and Tulsion T-42 were performed by adjusting flow rate of copper solution to pass in the rate of 10–20 mL/min in continuous mode. Samples were collected at fixed time interval. Elution of loaded copper from adsorbent surface was done by using 10 % sulfuric acid in 60 min.
2.2.3. Analytical studies
CL 46 pH meter (Toshniwal Pvt. Ltd., Ajmer) was used to check and maintain the equilibrium pH of feed and raffinate for gaining maximum copper adsorption. Copper concentration in raffinate was detected by using Atomic Absorption Spectrophotometer (AAS) (Perkin Elmer model, USA) using 324.8 nm wavelength.
Adsorption percentage was calculated by using Equation (1)
Co = Initial copper concentration in aqueous feed solution (ppm)
Ce = Copper concentration in the raffinate at equilibrium (ppm)
Adsorption capacity was determined by using Equation (2)
m = Mass of adsorbent (gm)
V = volume of adsorbate (L)
qe = Adsorption capacity (mg/g)15)
3. Result and Discussion
Replica of original effluent containing copper solution in the range of 50–250 ppm was contacted with plant root/ synthetic adsorbent by varying contact time, adsorbent dose, metal concentration in aqueous feed, etc.
3.1. Effect of pH
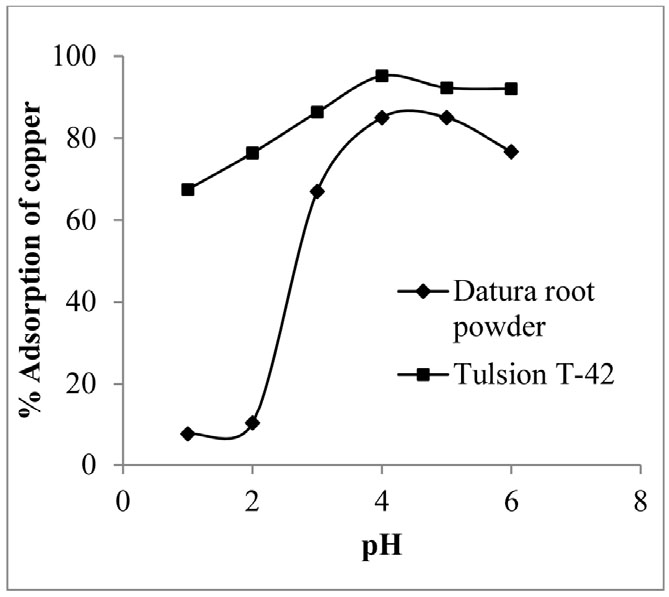
Effect of pH on the adsorption of copper was studied by varying pH of aqueous solution from 1 to 6 by keeping all other process parameters constant. Experiments were performed by taking 0.1 g of adsorbent in each case (Datura root powder/Tulsion T-42) per 50 ml of aqueous feed solution containing 100 ppm copper solution. Result indicates that adsorption percentage of copper with increase in pH 1 to 4 was found to be increased from 7.58 to 84.97 and 67.48 to 95.15 in case of Datura root powder and Tulsion T-42, respectively. Copper adsorption was decreased with further increase in pH. It indicates pH 4 is equilibrium pH showing maximum adsorption in each case (Datura root powder/ Tulsion T-42) (Fig. 1). pH is one of the important parameter for adsorption process as this technique involves the exchange of ion which can compete with H3O+ ions present in aqueous solution. As pH varies the concentration of these ions with respect to metal ions also varies16). Hence, pH 4 was selected for further adsorption studies.
3.2. Effect of contact time
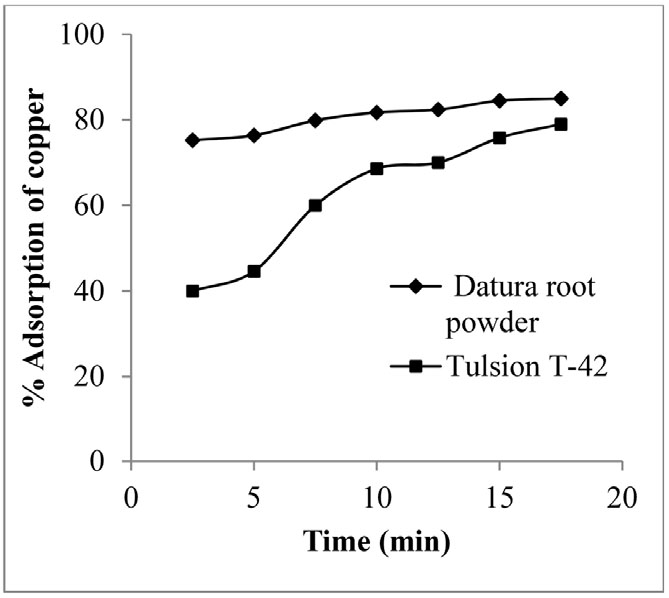
Further, to optimize the contact time, the adsorbent and copper metal solutions were contacted at different time period between 2.5 to 20 min, keeping all other parameters constant. Experiments were conducted by taking 0.1 g of adsorbent in each case (Datura root powder/Tulsion T-42) per 50 ml of aqueous feed solution containing 100 ppm copper solution at pH 4. Result indicated that there was increase in adsorption percentage of copper from 75.24 to 85 and 40 to 79 in case of Datura root powder and Tulsion T-42, respectively with increase in contact time from 2.5 to 15 min until equilibrium was attained. It states that 15 min time was found sufficient for Datura root powder for maximum adsorption, while more than 15 min time was required in case of Tulsion T-42 to attain equilibrium (Fig. 2). It may be attributed for change in interaction between copper ions and adsorbent surface with respect to time. Initially adsorption process was occurred at faster rate due to the availability of more surface area for copper adsorption and slows down later as equilibrium attained17).
3.3. Effect of adsorbent dose
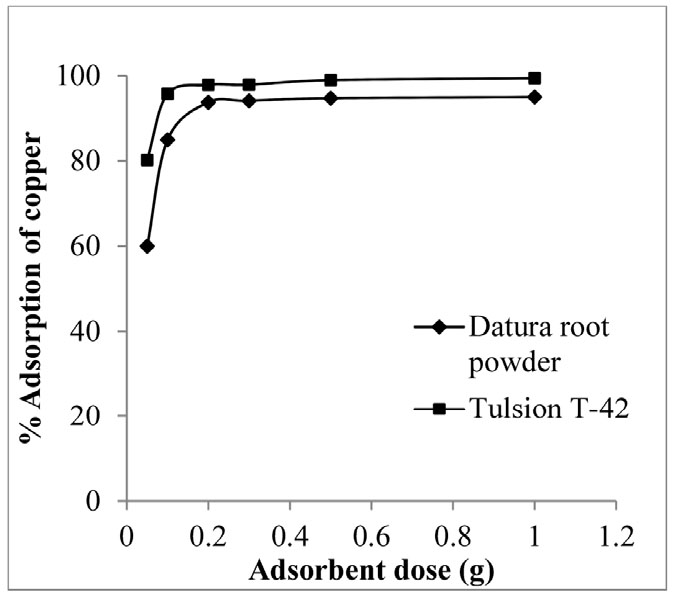
At the optimized pH and contact time, effect of adsorbent dose was studied. Minimum adsorbent dose for maximum adsorption process of metal is one of the essential factors to study adsorption behaviour of adsorbent. Experiments were performed by varying adsorbent dose from 0.05 to 1 g (Datura root powder/ Tulsion T-42) per 50 ml of aqueous feed solution containing 100 ppm copper concentration at pH 4. Result indicates that 0.2 g Datura root powder was necessary for complete adsorption, while 0.1 g of Tulsion T-42 was found sufficient for maximum adsorption (Fig. 3). It indicates that increase in adsorbent dose from 0.05 to 1 g leads to increase in copper adsorption percentage from 60 to 95 and 80.24 to 99.24 in case of Datura root powder and Tulsion T-42, respectively. It may be due to increase in adsorption site with respect to increase in adsorption dose10). To compare experimental results, 0.1 g dose was fixed for further studies.
3.4. Effect of aqueous feed concentration
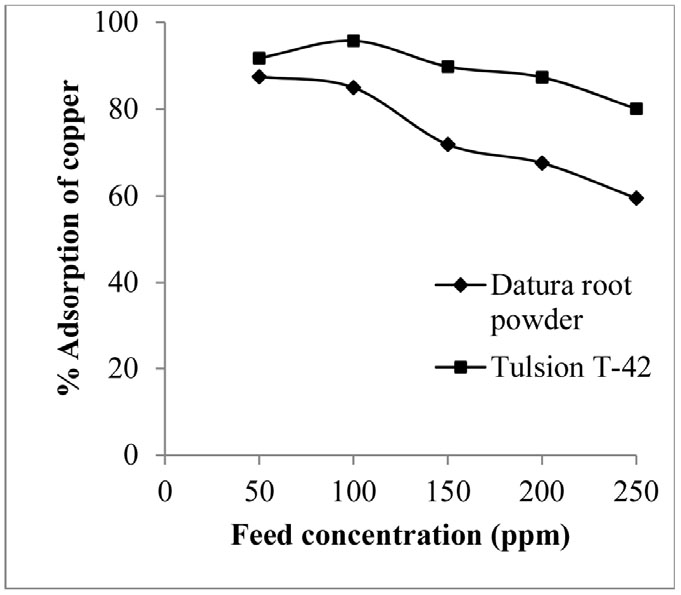
Effect of copper concentration in aq. feed on the copper adsorption process was investigated by varying copper concentration from 50 to 250 ppm in feed. The studies were performed by taking 0.1 g of adsorbent dose (Datura root powder/ Tulsion T-42) per 50 ml aqueous feed solution and kept in contact for 15 min for Datura root powder and 30 min for Tulsion T-42 at pH 4. Result indicates that copper adsorption percentage decreased from 84.97 to 59.19 and 95.78 to 85.11 in case of Datura root powder and Tulsion T-42, respectively with increase in copper concentration from 100 ppm to 250 ppm (Fig. 4). It can be attributed to the exchange sites present on adsorbent getting saturated at equilibrium metal concentration and thus further increase in metal concentration decreases adsorption percentage18).
3.5. Loading capacity of adsorbents
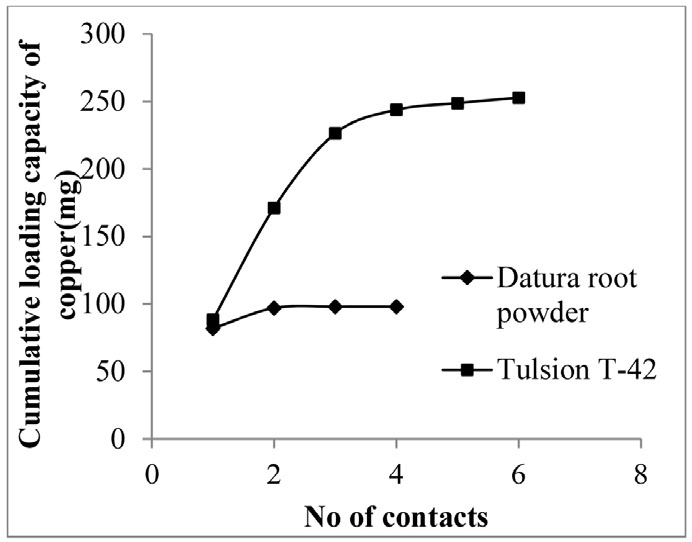
After the optimization of all experimental parameters, the determination of loading capacity of both the adsorbents were studied. Maximum loading capacity of adsorbent was determined by repetitive contact of the 50 ml fresh aqueous feed solution containing 100 ppm copper and same adsorbent (Datura root powder/ Tulsion T-42). Multiple repeated contacts with previously loaded adsorbent gave idea about maximum loading capacity of adsorbent. For efficient comparative study of copper adsorption using both the adsorbents, the similar experimental conditions (30 min contact time, 0.1 g adsorbent dose, pH 4) were maintained. Result indicates that 2 contacts of 100 ppm copper solution were required for saturation of 0.1 g of Datura root powder, while 5 contacts of 100 ppm copper solution were found to be needed for complete saturation of 0.1 g of Tulsion T-42 (Fig. 5). It showed copper adsorption capacity of Datura root powder was 997 mg/g, while adsorption capacity of Tulsion T-42 was found 2570 mg/g. Loaded adsorbent with copper was eluted successfully to get copper enriched solution using of 10 % sulfuric acid in 60 min.
3.6. Column studies
Based on the batch adsorption studies optimization, further study was validated in column. Column experiments were performed to study adsorption behaviour of these adsorbent in continuous mode of operation. Experiments were performed by taking 1 g of adsorbent (Datura root powder/ Tulsion T-42) and 250 ppm copper solution having pH 4 in column. 10–20 mL/ min flow rate was adjusted to pass aqueous feed solution in continuous mode. The obtained experimental result from batch scale adsorption studies was validated satisfactorily with the result of column studies. Cumulative loading capacity of both the adsorbent (Datura root powder, Tulsion T-42) were compared. It indicates that adsorption of copper in Datura root powder equally stands with the adsorption of copper using Tulsion T-42. Loaded adsorbent with copper was eluted successfully to get copper enriched solution using of 10 % sulfuric acid in 60 min.
3.7. Kinetics of adsorption
Mechanism of copper adsorption on Datura root powder and Tulsion T-42 adsorbent were determined using kinetic models. Pseudo first order and pseudo second order kinetic equations were calculated by using aqueous feed solution containing 100 ppm copper.
Equation (3) represents pseudo first order rate reaction as follows:
Where, t is contact time, k1 is rate constant (min-1), q and qe are the amount of copper (mg/g) adsorbed on adsorbent at start and at equilibrium, respectively. Integrating and applying boundary conditions (t= 0 and q= 0 to t= t and q= qe) on Equation (3), it takes form as follows
To calculate rate constant k1, graph was plotted by taking log (qe–q) against time (min)1). Rate constant k1 and correlation coefficient R2 were determined from the slopes obtained from the graph mentioned in Table 1.
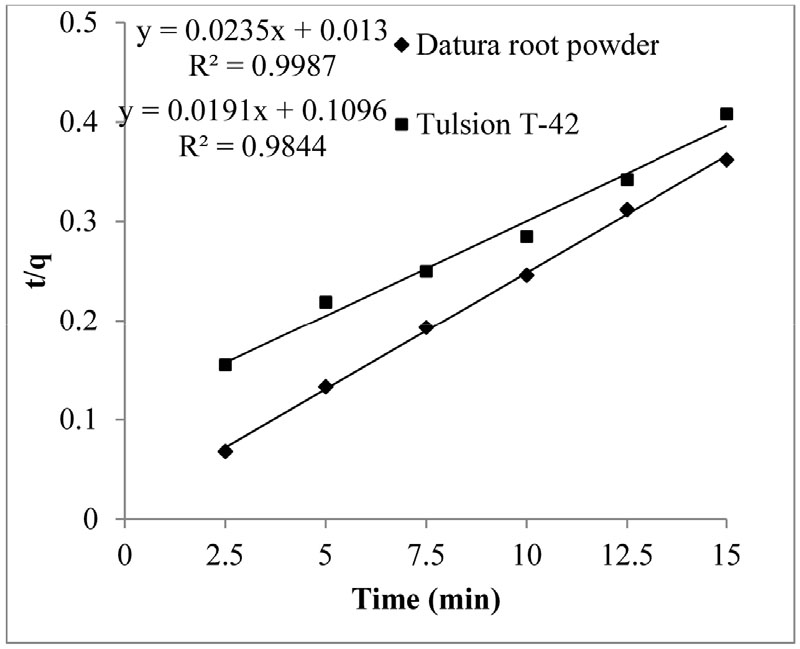
Second order kinetics for copper adsorption was obtained by putting same experimental data in to following Equation (5)
Where, t is contact time, k1 is rate constant (min-1), q and qe are the amount of copper (mg/g) adsorbed on adsorbent at start and at equilibrium, respectively. Integrating and applying boundary conditions (t= 0 and q= 0 to t= t and q= qe) on Equation (5), it takes form as follows
Where, h= k2 qe2
To calculate rate constant k2, graph was plotted by taking (t/q) against time (min). Rate constant k2 and correlation coefficient R2 was determined from the slope obtained from the graph (Fig. 6) and also mentioned in Table 1.
Table 1.
Rate constants for first and second order for the adsorption of copper on Datura root powder and Tulsion T-42 adsorbent
Second order rate reaction was more satisfactorily followed by Datura root powder and Tulsion T-42 as compared to first order rate reaction.
3.8. Adsorption isotherm
Langmuir and Freundlich adsorption isotherm are mostly used to justify adsorption phenomenon. Results obtained from batch adsorption studies were examined by using these isotherm equations. Langmuir isotherm model is based on concept as metal ion uptake occurs on homogeneous adsorbent surface by single layer adsorption without interaction between adsorbed ions. Langmuir equation is represented by Equation (7) as follows
Where, Ce is equilibrium concentration of copper metal in solution (mg/mL), q is amount of metal adsorbed at equilibrium, kl is Langmuir constant, and qm is adsorption capacity of adsorbent (mg/g).
To calculate Langmuir constant kl, graph was plotted by taking 1/Ce against 1/q. Langmuir constant kl, correlation coefficient R2 was determined from the slope obtained from the graph which is mentioned in Table 2.
Table 2.
Rate constants for Langmuir and Freundlich adsorption isotherm for copper adsorption on Datura root powder and Tulsion T-42 adsorbent
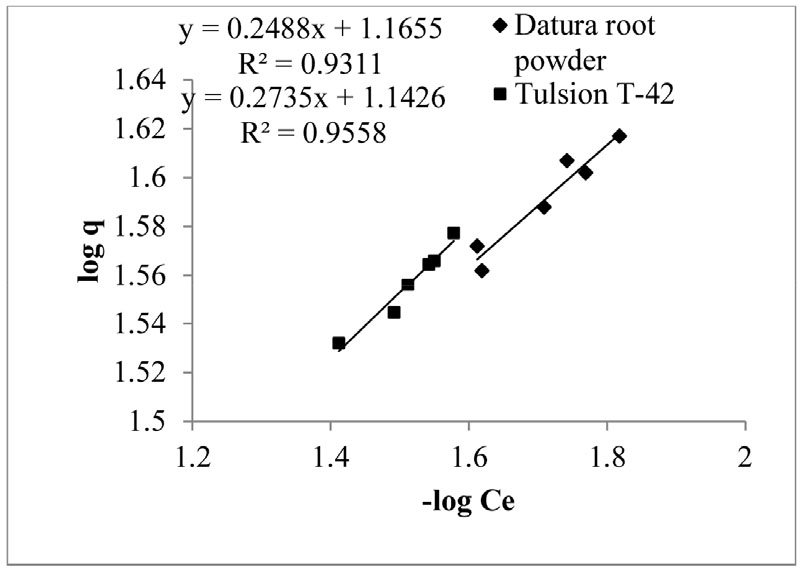
While, Freundlich isotherm model is based on concept that metal ion uptake occurs in heterogeneous surface on multiple layer with proper interaction between the ions. Freundlich equation is represented by Equation (8) as follows
Where, q is amount of copper metal adsorbed at equilibrium (mg/g), Ce is equilibrium concentration of copper metal in solution (mg/mL), kf is Freundlich constant and n is Freundlich exponent.
Logarithmic form of Equation (8) is
To calculate Freundlich constant kf, graph was plotted by taking log (Ce) against log (q). Freundlich constant kf, correlation coefficient R2 was determined1) from the slopes obtained from the graph (Fig. 7) of our experimental results, which is also mentioned in Table 2. Freundlich adsorption isotherm was found to be more suitable for Datura root powder and Tulsion T-42 with R2 value 0.931 and 0.955, respectively which indicates multilayer and heterogeneous copper adsorption process.
4. Conclusions
Batch and column experiments for copper adsorption were carried out to compare copper adsorption behavior of plant root adsorbent (Datura root powder) and synthetic (Tulsion T-42) adsorbent. Based on the obtained results following conclusions have been made, which are as follows:
• 95% copper adsorption was obtained for Datura root powder by using 0.2 g of adsorbent at 15 min contact time, while Tulsion T-42 required 0.1 g dose and 30 min contact time at pH 4.
• Kinetics data found to be fitted well in pseudo second order rate reaction and follows Freundlich adsorption isotherm for both adsorbents.
• Copper from the metal loaded adsorbents was eluted completely using 10% sulfuric acid in 60 min contact time to get copper enriched solution.
• Datura plant root adsorbent can replace the synthetic adsorbent to improve the cost efficiency of process related to copper adsorption from industrial effluents.
• The disposal issue related to poisoned synthetic resin (Tulsion T-42) can be solved by using plant root adsorbent (Datura root powder), which is environmental and easily disposable.
Datura plants are easily available in bushy areas, road side or forest. Therefore, it can be considered as efficient bioadsorbent for copper reclamation having potential to get commercialized after the scale up studies.