1. Introduction
2. Materials and Methods
2.1. Material
2.2. Magnetizing Roasting of the raw material
3. Results and Discussion
3.1. Thermodynamic Analysis of Reductive Roasting
3.2. Magnetizing Roasting of the raw material
3.3. Effect of the carbon dose in the magnetizing roasting
3.4. Effect of the temperature in the magnetizing roasting
4. Conclusions
1. Introduction
Titanium (Ti) is the ninth most abundant element, making up 0.63% of the Earth's crust by weight. Its main ores are rutile (TiO2) and ilmenite (FeTiO3), which are widespread throughout the Earth's lithosphere and crust. Primary ilmenite deposits are found mainly in rock formations in Norway, Finland, Canada, China, and Russia. Nonetheless, a significant portion of ilmenite is also mined from beach sands and placer deposits. The top producers of titanium minerals include South Africa, Australia, Canada, India, Madagascar, Malaysia, Vietnam, and China. Worldwide, approximately 4 million tons of ilmenite are produced annually1). Production is constrained by various environmental factors, including reserve availability, reserve density, mineral properties, and political issues2,3,4,5). Titanium is considered a future advanced material and a strategic global resource because of its exceptional material properties. It is utilized across multiple sectors, including the aerospace, marine, and weapons manufacturing industries, chemical processing, and the medical fields, as well as in titanium-based reactions such as photocatalysis1,2).
Korea’s titanium imports are projected to grow by approximately 65%, rising from around KRW 865.2 billion (USD 588.2 million) in 2020 to KRW 1.33 trillion (USD 904.2 million) in 2030. The urgent need for titanium in Korea’s industry is driven by high demand. In 2020, the country imported 12,850 tons of rutile and 44,475 tons of ilmenite concentrate3). Domestic titanium ore, mainly ilmenite, is abundantly buried in the Hadong-Sancheong region. Although mineral processing research has been conducted previously, progress has been limited by low ore grades and challenges in achieving economic viability. Additionally, laboratory-scale studies on mineral processing and refining technologies for titanomagnetite ore have been conducted at the Gwanin Mine in Pocheon. However, pilot testing is still in its early stages. Therefore, Korea needs a robust titanium industry to lessen its reliance on imports.
Due to the long life cycle of titanium materials, both the recycling rate and the level of related smelting technology are low. Small and medium-sized companies primarily engage in low-level process technologies, such as importing and processing semi-finished products. The Taebaek area Myeon-San Layer deposit is newly discovered; research and Development (R&D) has just begun, and it will take some time to extract and produce titanium6,7). However, because the deposit is primarily rutile, it may be easier to exploit. On the other hand, although iron is not a rare metal, it is significant, particularly for steel manufacturing. A combination of gravity, magnetic, and flotation was used to beneficiate the Myeon-San Layer deposit7). Although the grades of the iron and titanium concentrates obtained were significant, at 48.80% and 30.41%, respectively, the recovery rate was low (46.2%)7). The main reasons for this are that hematite and rutile, the primary minerals of the deposit, contain high levels of gangue, and the fine hematite and rutile minerals are intricately embedded within the gangue. As a result, fine grinding is necessary to separate them from the gangue. Consequently, conventional mineral processing methods, such as gravity separation, magnetic separation, flotation, or combinations thereof, are ineffective at recovering minerals from this mine.
Given the disadvantages of traditional beneficiation methods for these minerals, some studies have proposed using coal-based direct reduction techniques to convert hematite to FeO, which can then react with rutile to produce ilmenite8). Magnetizing roasting is a heat-treatment process that transforms weakly magnetic iron-bearing minerals, such as hematite (Fe2O3) and goethite (FeO(OH)), into strongly magnetic magnetite (Fe3O4). This transformation improves the material's magnetic properties, thereby facilitating low-intensity magnetic separation. The process is usually carried out in a reducing atmosphere with gases like CO or H2, or with solid reductants such as coal or biomass. For titanium-bearing ores, which often contain complex associations of iron oxides with minerals like rutile or ilmenite, magnetizing roasting can convert the normally diamagnetic titanium minerals into iron-titanium magnetic minerals. This allows for physical separation, enhancing the efficiency of subsequent beneficiation and recovery processes8,9,10,11).
This study aims to improve the grade and recovery of titanium from the Taebaek Myeon-San Layer deposit through magnetizing roasting with coal, followed by additional magnetic separation. It characterizes the raw material and investigates factors that influence coal reduction in the deposit and in the magnetic products, thereby enhancing subsequent magnetic separation.
2. Materials and Methods
2.1. Material
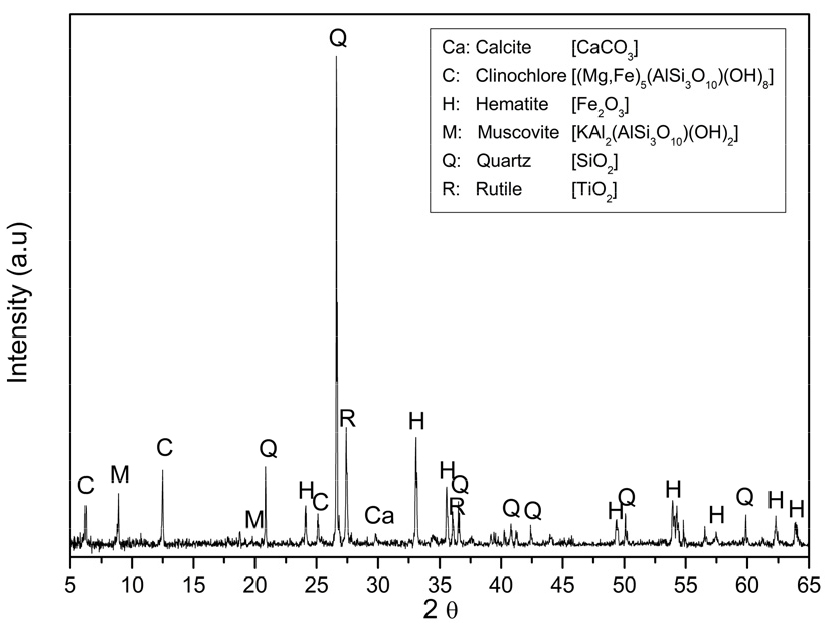
The Taebaek Myeon-San Layer deposit mineral was utilized in this study. The sample was crushed using a Jaw crusher, followed by a cone crusher, to obtain particles less than 10 mm. The mineral composition was analyzed by an X-ray diffractometer (XRD; Bruker D8 Advance A25, Bruker-AXS, Germany). The diffractogram was obtained for 2θ values between 5° and 65°. A divergence slit of 0.3°, a Soller slit of 2.5°, and a sample rotation speed of 20 RPM. The results are displayed in Fig. 1. Chemical composition was analyzed using an X-ray fluorescence spectrometer (XRF, XRF-1800, Shimadzu, Japan).
The main mineral components of the sample are rutile (TiO2, JCPDS: 21-1276), hematite (Fe2O3, JCPDS: 33-0664), quartz (SiO2, JCPDS: 33-1161), clinochlore ((Mg,Fe,Al)3(Si,Al)2O5(OH)4, JCPDS: 07-0078), albite (NaAlSi3O8, JCPDS: 09-0466), and muscovite (KAl2(AlSi3O10)(OH)2, JCPDS: 07-0025), with traces of calcite (CaCO3, JCPDS: 05-0586). Table 1 highlights a significant SiO2 content (28.93 wt%). Additionally, the percentages of Fe2O3 and TiO2 are notable: 32.96 wt% and 24.55 wt% for the high-grade mineral.
Table 1
Chemical composition (in wt.%) of the Myeon-San Layer titanium ore used as raw material
| SiO2 | Al2O3 | Fe2O3 | TiO2 | K2O | Na2O | CaO | MgO | P2O5 | MnO |
| 28.93 | 4.35 | 32.96 | 24.55 | 1.01 | 0.02 | 0.64 | 1.87 | 0.16 | 0.15 |
Table 2 summarizes the progressive mineralogical evolution of Fe–Ti ores during weathering (oxidation). In the early stages (1–3), homogeneous or ferrian ilmenite and rutile (TiO2) dominate, indicating limited Fe²⁺→Fe3+ oxidation. As oxygen fugacity increases, Ti-bearing hematite solid solutions (titanohematite) are expected to become thermodynamically stable and coexist with rutile (stages 4–5). In more advanced stages (6–7), extensive oxidation and Fe–Ti reorganization may result in the formation of pseudobrookite6,13).
Table 2
Phases of Fe-Ti ore deposit oxidation (weathering)
Based on this oxidation sequence, the Myeon-San Layer deposit is inferred to correspond to an intermediate-to-late weathering stage (approximately stages 5–6). Accordingly, the ore is interpreted to consist mainly of rutile and hematite-based Fe–Ti solid solutions, with possible trace amounts of pseudobrookite. Such minor or poorly crystalline phases are expected to occur below the detection limit of conventional XRD and, therefore, are not distinctly resolved in the diffraction pattern shown in Fig. 1.
The morphological characteristics of the sample were analyzed using a scanning electron microscope (SEM; JEOL JSM-6380LA, Japan) equipped with an energy-dispersive spectrometer (EDS; Bruker Nano GmbH, X Flash Detector 630M, Germany). Fig. 2 highlights these results. In the SEM image (Fig. 2a), angular particles ranging from 100 to 500 µm in size are observed within a matrix. While Fig. 2b reveals a heterogeneous distribution of the main elements, silicon (white) is predominant; iron (red) is attached to some white silica particles associated with quartz; and titanium (yellow) is found in regions attributed to rutile. The overlapping colors (orange) indicate potential coexistence of Fe-Ti phases, while the scattered distribution suggests a complex mineral association. These results confirm that the sample primarily consists of hematite, rutile, and quartz, consistent with Fig. 1 and previous mineralogical analyses of the deposit6,7).
2.2. Magnetizing Roasting of the raw material
Carbothermic reduction is a high-temperature process in which metal oxides are reduced using carbon as the reductant. The experiments were carried out in an elevator-type electric furnace equipped with eight tungsten carbide electrodes (50/60 Hz, 2400 W, 220 V; Hanyang Lab. Scientific Services, Daejeon, South Korea). Hematite exhibits a higher magnetic susceptibility than the diamagnetic behavior of quartz and rutile12,13,14). To enhance hematite's magnetic response and promote ilmenite formation, the raw material was mixed with carbon and roasted.
This work analyzes the effects of temperature and carbon content on the recovery of iron and titanium from the raw material. To determine the optimal roasting temperature, 1 kg of ore was mixed with 200 g of carbon (1:2 ratio) and placed in a lidded alumina crucible, which was then roasted at various temperatures for 1 h. After cooling, the roasted product was wet ground with 10 kg of rods and 1 L of water for 60 min.
A magnetic test was conducted on various iron and titanium minerals to determine their magnetic recovery range. Magnetite (95 %), hematite (93 %), and rutile (99 %) chemical reagents were purchased from Daejung Chemicals & Metals Co., Ltd. Ilmenite was obtained from a previous study in which valuable heavy minerals were concentrated and recovered from dredged fine aggregate waste21). For ulvöspinel and titanomagnetite, synthetic samples were prepared by mixing hematite and rutile in stoichiometric proportions, adding 20% graphite, and calcining with the lid sealed at 950 °C. The samples were then separated from the coal via flotation.
Magnetic separation was performed at different magnetic field intensities using a wet-vortex magnetic separator (N1-3K-23-11, Eriez Manufacturing Co., USA). The magnetic field strength was controlled by adjusting the voltage. The separator was fed with a slurry containing 10 wt% solids at a constant flow rate of 50 ml/min, maintained by a peristaltic pump. Magnetic and non-magnetic fractions were collected, dried, weighed, and analyzed. To evaluate the effect of carbon dosage, 1 kg of raw material was mixed with 0, 50, 100, 150, 200, and 200 g of carbon, roasted under the same conditions, cooled, ground, and separated magnetically at 0.8 T.
3. Results and Discussion
3.1. Thermodynamic Analysis of Reductive Roasting
The graphite acts as a reducing agent. The principal reactions and standard Gibbs free energy are outlined in Table 3. Due to limited contact between hematite and carbon, Reaction 2 proceeds slowly. However, the CO produced by Reaction 1 enhances Reaction 3. When CO is generated in excess, it favors Reactions 5, 6, and 7 since they involve solid-gas interactions15). CO is produced via the Boudouard reaction. When the equilibrium partial pressure of CO2 in the Boudouard reaction exceeds the partial pressure of CO2, CO2 is consumed, and reductant CO is generated. An excess of CO could reduce the yields of these products. The formation of CO is favored as the temperature increases.
Table 3
Reactions during the reductive roasting of hematite (Fe2O3) with graphite (C) as a reducing agent
| Chemical ReactionReaction | Standard Gibbs free energy (ΔG0) (KJ/mol) | |
| 1 | ||
| 2 | ||
| 3 | ||
| 4 | ||
| 5 | ||
| 6 | ||
| 7 |
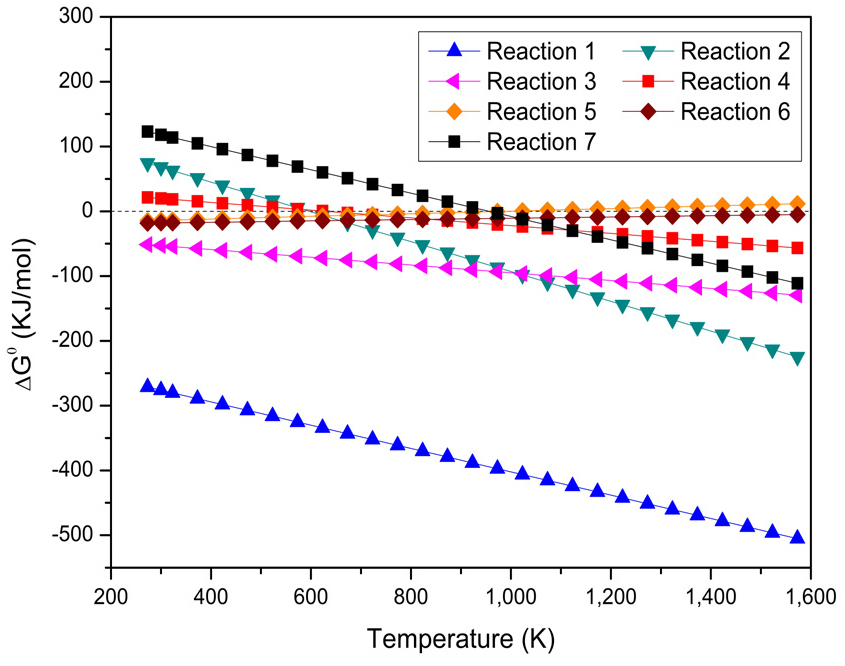
Using the data from Table 3, the standard variation of Gibbs free energy (ΔG0) with temperature (K) for the seven reactions was plotted in Fig. 3. Reactions 1, 2, and 3 show strongly negative ΔG0 values throughout the temperature range, indicating that these reactions are thermodynamically favorable and spontaneous under the conditions considered. Reaction 2 exhibits a less steep decline compared to Reactions 1 and 3, suggesting moderate sensitivity of ΔG0 to temperature. In contrast, Reactions 4, 5, 6, and 7 have ΔG0 values closer to zero or positive at lower temperatures. Reaction 4 has a positive ΔG0 and decreases steadily, crossing the zero line at higher temperatures, indicating it becomes spontaneous only at elevated temperatures. Reactions 5, 6, and 7 remain near zero or slightly negative, indicating limited spontaneity. Overall, Fig. 3 shows that Reactions 1, 2, and 3 are highly favored, whereas the others are only marginally spontaneous or require higher temperatures to become thermodynamically feasible.
The reduction of hematite (Fe2O3) to magnetite (Fe3O4) is straightforward because the equilibrium concentration of CO remains very low at any temperature in Reaction 3. Reaction 3 is more thermodynamically favorable because it has a lower (more negative) ΔG0 across the temperature range considered, as shown in Fig. 3. This suggests it would be more spontaneous under similar conditions. Moreover, increasing the temperature promotes this reaction9). However, a high concentration of CO should be avoided, as it can lead to the formation of Fe, which has a high magnetic susceptibility. Furthermore, at high temperatures, magnetite may be over-reduced to wüstite (FeO). Wüstite is less magnetically susceptible than magnetite12). Therefore, temperature and coal quantity are two key factors influencing the magnetizing roasting process.
Using the standard Gibbs free energy, the effects of CO and temperature on the products of the reduction reactions can be calculated using the Van’t Hoff equation (Eq. (1)).
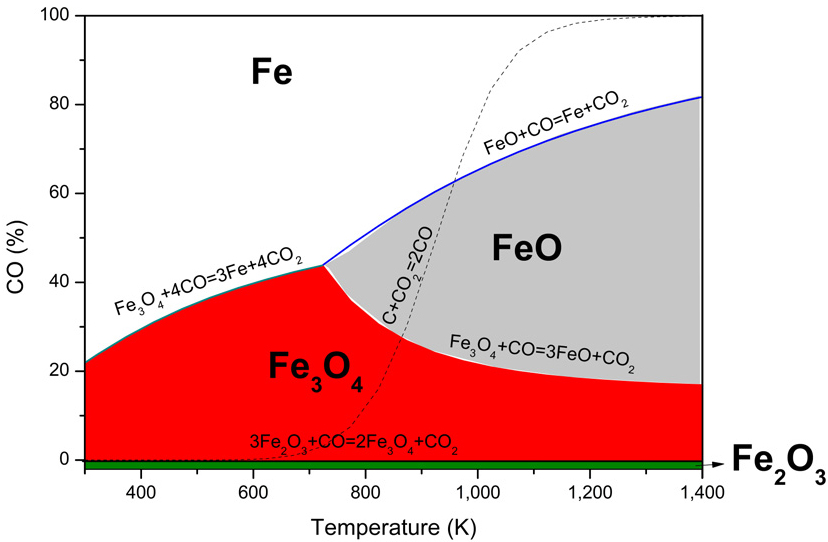
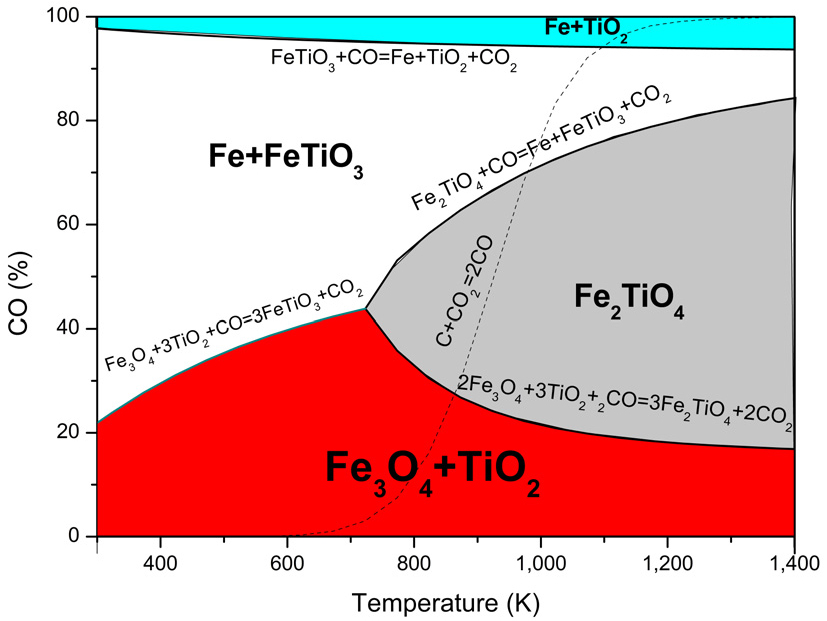
Where ΔG0 is the standard variation of Gibbs free energy (kJ/mol), ΔG is the variation of the Gibbs free energy during the reaction (kJ/mol), R is the ideal gas constant (0.008314 kJ/mol·K), T is the reaction temperature (K), and Q is the reaction quotient (a measure of the relative amounts of products and reactants present at any given time, regardless of whether the reaction is at equilibrium). Q can be expressed as a relationship of pressures, assuming the activities of all solids are 1. At equilibrium, ΔG equals 0. Therefore, we can relate temperature to the percentage of CO. This relationship, called the Baur–Glässner diagram10,11,15), is illustrated in Fig. 4.
Baur–Glässner diagram in Fig. 4 illustrates the equilibrium stability regions of iron oxides during reduction with carbon monoxide as a function of temperature and gas composition. At low temperatures and low CO concentrations, magnetite (Fe3O₄) is the stable phase; increasing both temperature and CO content promotes the formation of wüstite (FeO) and, ultimately, of metallic iron (Fe) at higher CO levels. The curves represent equilibrium boundaries: above each line, the oxide on the right becomes stable, whereas below it, the oxide on the left dominates.
When rutile (TiO2) is introduced into the system, ilmenite (FeTiO3), ulvöspinel (Fe2TiO4), or rutile combined with metal iron can be formed under reducing conditions. Reactions 8 through 23 outline the chemical reactions involving carbon within the system. Rutile can be decreased to titanium, as shown in Reactions 8 and 9. Hematite may react with rutile to produce ilmenite, as demonstrated in Reactions 10 and 11. During the reducing roasting of hematite, magnetite can react with rutile to form ilmenite, as illustrated in Reactions 12 and 13. When rutile is the limiting reactant, ilmenite cannot be created; instead, ulvöspinel is produced, as indicated in Reactions 16, 17, 22, and 23. Additionally, ilmenite can react with carbon to yield iron and rutile, as detailed in Reactions 14 and 15. Finally, ulvöspinel can react with carbon and CO to produce metal iron and rutile, as demonstrated in Reactions 18 and 19. Using data from Table 4, the variation of Gibbs free energy (ΔG0) with temperature (K) for the 14 reactions was divided and plotted in Fig. 5.
Table 4
Reactions for the reductive roasting of rutile (TiO2) and hematite (Fe2O3) using graphite (C) as a reducing agent
| Chemical ReactionReaction | Standard Gibbs free energy (ΔG0) (KJ/mol) | |
| 8 | ||
| 9 | ||
| 10 | ||
| 11 | ||
| 12 | ||
| 13 | ||
| 14 | ||
| 15 | ||
| 16 | ||
| 17 | ||
| 18 | ||
| 19 | ||
| 20 | ||
| 21 | ||
| 22 | ||
| 23 |
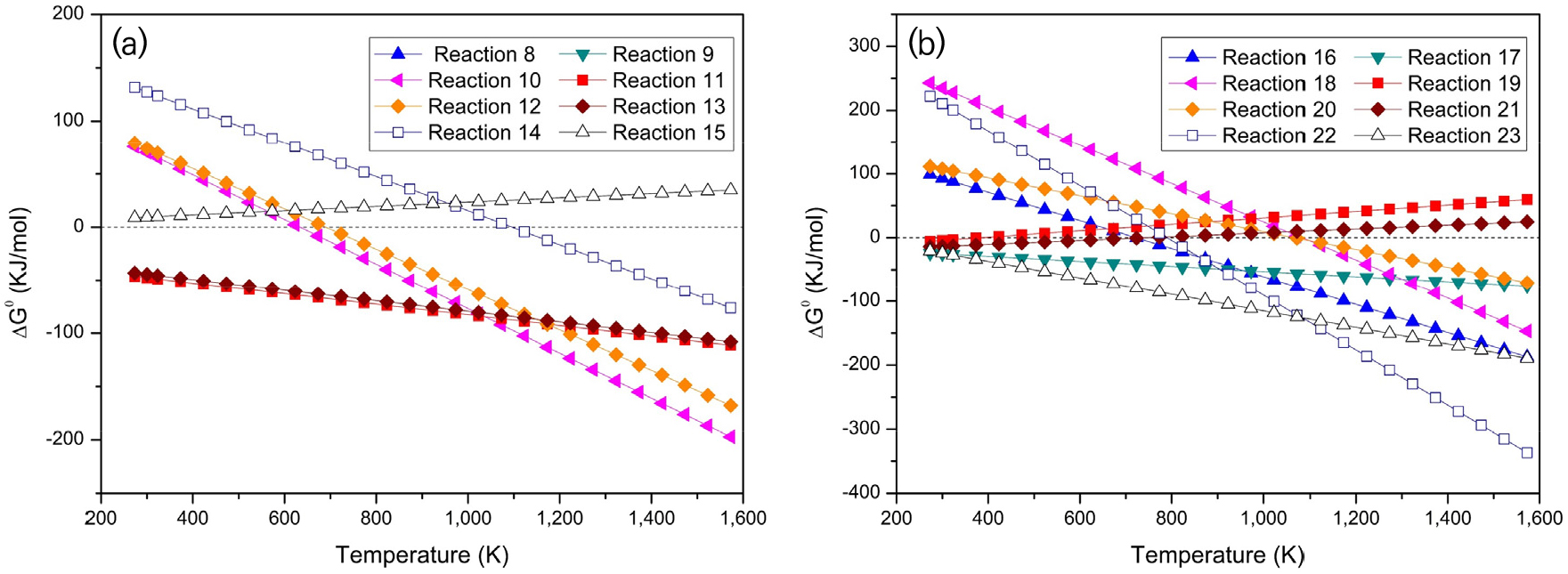
Reactions corresponding to Reactions 10-13, 18, and 19 show an apparent decrease in ΔG0 with increasing temperature, crossing the zero line at different critical points (around 700–1,100 K). This indicates that these reactions shift from being non-spontaneous at lower temperatures to thermodynamically favorable at higher temperatures. In contrast, Reaction 16 (carbothermic reduction of ilmenite) has the highest positive ΔG0 value (>100 kJ/mol at 300 K). It only approaches spontaneity above approximately 1,100 K, requiring significantly more thermal energy to proceed. Meanwhile, Reactions 14, 15, and 17 show nearly constant ΔG0 values across the studied range, remaining close to zero or slightly negative, suggesting these reactions are only weakly dependent on temperature and can occur under relatively mild thermal conditions.
Using the data in Table 4, the Baur–Glässner diagram was generated and shown in Fig. 6. The curves distinguish the stability zones of different phases, such as ulvöspinel (Fe2TiO4), ilmenite (FeTiO3), iron metal (Fe), and rutile (TiO2), illustrating the reduction reaction sequence under a CO atmosphere. At lower temperatures and CO levels, ulvöspinel remains stable. However, increasing temperature and reducing conditions gradually shift the system toward ilmenite and iron, eventually forming metal Fe with rutile. Including the Boudouard equilibrium highlights the gas composition produced by carbon reduction, emphasizing the interaction between the gas phase and solid-state transformations.
3.2. Magnetizing Roasting of the raw material
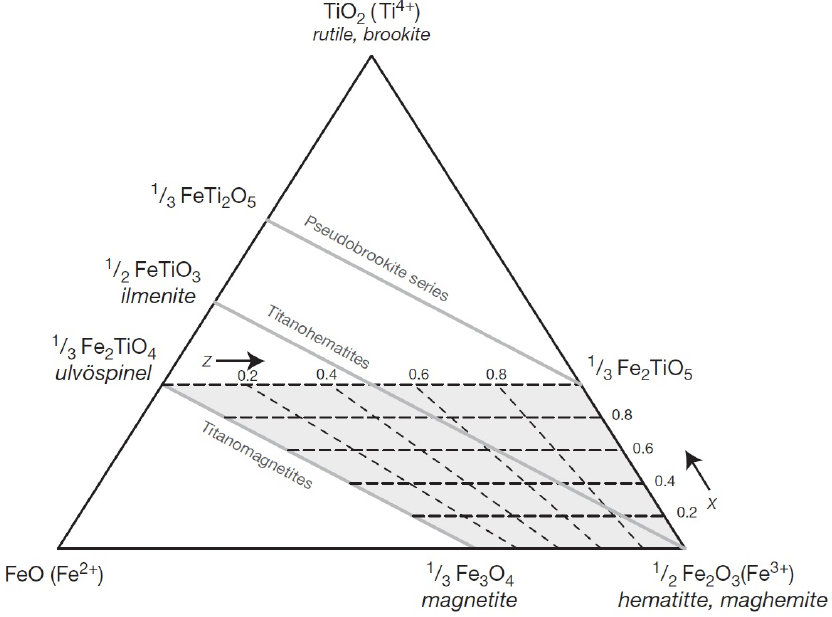
Almost all natural magnetic minerals contain Fe and belong to the Fe–Ti oxides, Fe sulfides, Fe oxyhydroxides, and Fe–Ni metal groups. Among these, the most common rock-forming magnetic minerals are Fe–Ti oxides, whose compositions can be summarized in an FeO–Fe2O3–TiO2 ternary diagram, shown in Fig. 7. This diagram includes the titanomagnetite (Fe3-xTixO4, 0>x>1), titanohematite (Fe2-yTiyO4, 0>y>1), and titanomaghemite (oxidized titanomagnetites) series. Most natural magnetic minerals mainly fall within the quadrilateral defined by magnetite, ulvöspinel, pseudobrookite, and hematite20). The occurrence and abundance of magnetic minerals in rocks and unconsolidated materials are controlled by a variety of complex processes, including magma composition, subsolidus and exsolution reactions during cooling of plutonic rocks, low-temperature oxidation of volcanic stones, serpentinization of ultramafic rocks, and reduction diagenesis in marine and lake sediments20).
Table 5 summarizes the magnetic properties of several iron-bearing oxide minerals and related compounds. Magnetite (Fe3O4) and maghemite (γ-Fe2O3) exhibit ferrimagnetic (FI) behavior with high Curie temperatures (Tc ≈ 850–950 K), indicating strong magnetic ordering at elevated temperatures. Substituting Ti in titanomagnetite (Ti0.6Fe2.4O4) significantly reduces both Tc and the magnetic moment (Ms), reflecting the dilution of Fe3+–Fe2+ exchange interactions. Like magnetite, titanomagnetite is a cubic spinel. In these minerals, Ti replaces Fe by substituting two Fe3+ ions with one Ti4+ ion and one Fe2+ ion, thus maintaining charge balance. Many intrinsic magnetic and physical properties of titanomagnetites, including saturation magnetization, Curie temperature, magneto-crystalline anisotropy, magnetostriction, and electrical conductivity, depend on various interconnected crystal chemical properties19). These include chemical composition, cation ordering, and defect content, each of which may be altered during initial petrogenesis or subsequent thermal history20).
Table 5
Magnetic properties of selected iron-bearing oxide minerals20)
| CompositionMineral | Tc (K), Tn (K) | Ms (A·m2/Kg) | Type of Magnetism | |
| Iron | α-Fe | 1043 | 218 | FM |
| Magnetite | Fe3O4 | 853 | 92 | FI |
| Maghemite | γ-Fe2O3 | 863–948 | 73 | FI |
| Titanohematite (y = 0.7) | TiyFe2-yO3 | ~430 | 30 | FI |
| Titanomagnetite (x = 0.6) | TixFe3-xO4 | 423 | 25 | FI |
| Hematite | α-Fe2O3 | 948 | 0.4 | Canted AF |
| Ilmenite | FeTiO3 | 40 | PM | AF |
| Ulvöspinel | Fe2TiO4 | 120 | PM | AF |
| Fe–chromite | FeCr2O4 | 88 | PM | FI |
| Pseudobrookite | Fe2O3·TiO2 | ~55-60 | < 0.5 | AF |
| Goethite | α-FeOOH | 393 | < 0.5 | Defect-AF |
Hematite (α-Fe2O3) shows canted antiferromagnetism with very low magnetization and a high Néel temperature (Tn = 948 K). Ti substitution in titanohematite (Ti0.7Fe1.3O3) lowers Tn to around 430 K and induces weak ferrimagnetism. Ilmenite (FeTiO3) and ulvöspinel (Fe2TiO4) are mainly antiferromagnetic or paramagnetic at room temperature, with very low ordering temperatures. Fe–chromite (FeCr2O4) displays ferrimagnetic behavior despite a low Tc (~88 K), while metal iron (α-Fe) shows strong ferromagnetism (FM) with the highest saturation magnetization (Ms = 218 A·m2/kg) and Tc (1043 K). Overall, Ti and Cr substitutions tend to weaken magnetic ordering and lower Curie temperatures compared with pure magnetite or iron20). Additionally, Table 5 highlights minerals labeled as PM, which have unpaired electrons but do not exhibit spontaneous magnetization at room temperature. Their magnetic moments are randomly oriented, producing negligible net magnetization under normal conditions. They respond weakly to an external magnetic field and exhibit antiferromagnetic ordering only below their Néel temperature (Tn). Examples include ilmenite and ulvöspinel.
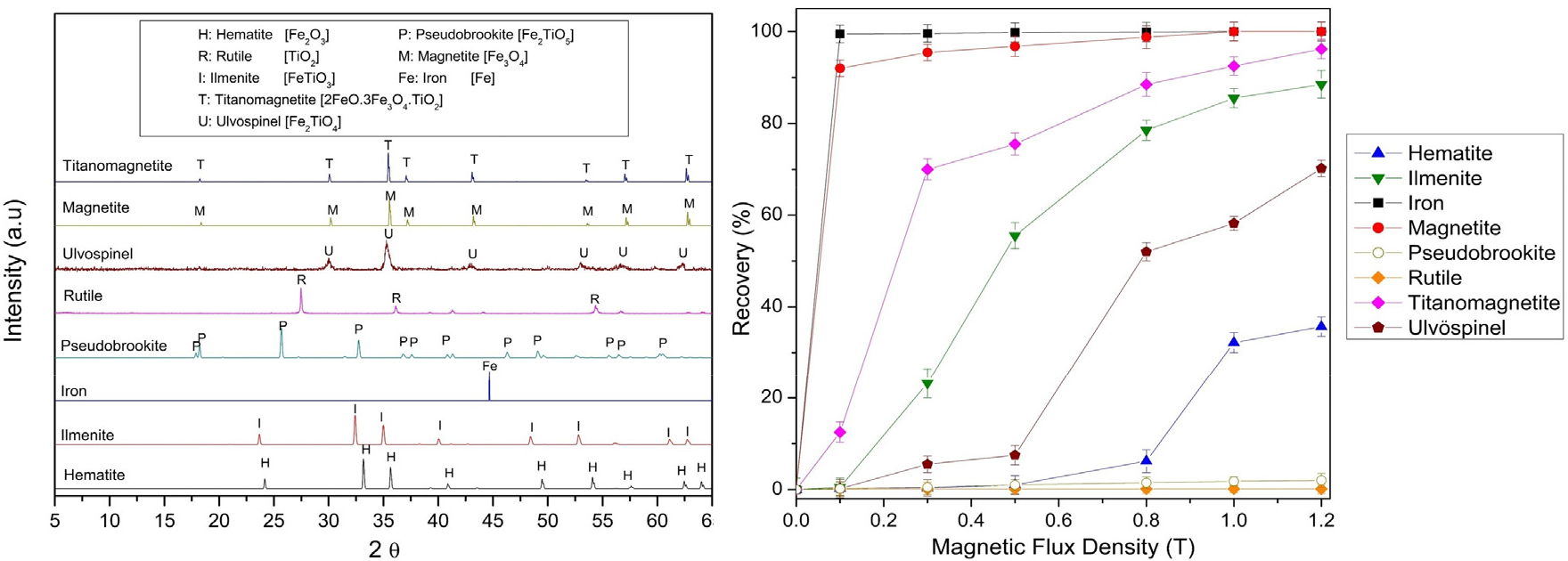
Fig. 8 shows the magnetic response of typical Fe–Ti oxide minerals based on magnetic flux density. The results clearly indicate that rutile and hematite, which make up most of the raw Myeon-San Layer ore, have very weak magnetic properties. In contrast, magnetite and titanomagnetite show strong magnetic responses. This comparison elucidates how carbothermic roasting operates, transforming weakly magnetic Fe–Ti oxides into more magnetic phases, thereby greatly enhancing the efficiency of subsequent magnetic separation.
Magnetite and metal iron exhibit nearly complete recovery even at low field strengths (< 0.2 T) because of their strong ferromagnetic properties. Titanomagnetite shows a steady increase in recovery with increasing field intensity, reaching almost complete separation at 1.2 T, consistent with its ferrimagnetic nature. In contrast, minerals such as hematite, ilmenite, ulvöspinel, and pseudobrookite exhibit weak or no recovery below 0.6 T, reflecting their predominantly paramagnetic or antiferromagnetic behavior at room temperature (Table 5). Rutile remains non-magnetic across the tested range. These results indicate that adequate separation of weakly magnetic Fe–Ti oxides requires high-intensity or high-gradient magnetic fields.
3.3. Effect of the carbon dose in the magnetizing roasting
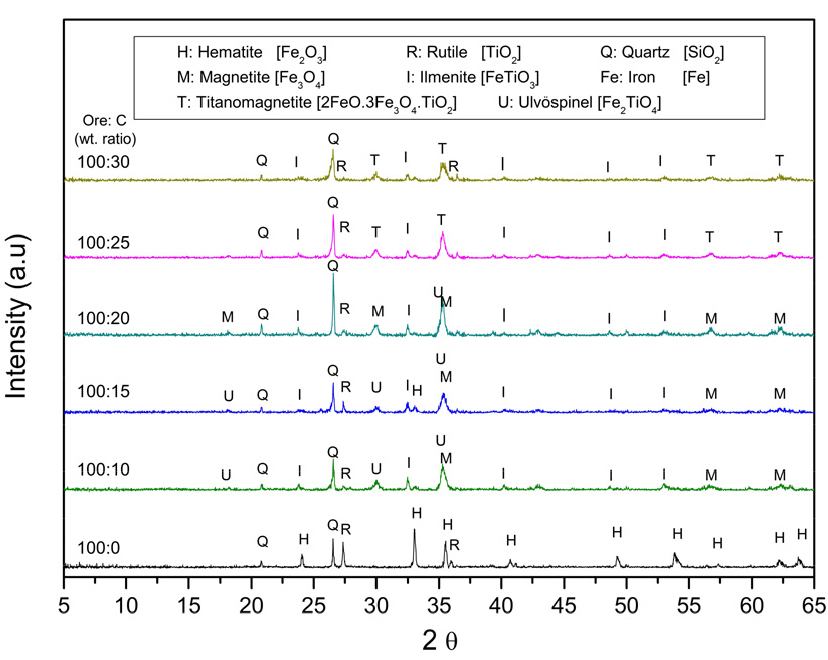
The effect of carbon dose on the magnetization roasting of the raw material is examined, and the results are shown in Fig. 9. The XRD patterns illustrate the phase evolution of the Fe–Ti–O system as the carbon dosage increases. At lower carbon levels, hematite and rutile are the dominant phases, but as the carbon input rises, ulvöspinel (Fe2TiO4, JCPDS: 34-0177) begins to form. Due to the excess CO produced through the Boudouard reaction (Reaction 7 in Table 3), ulvöspinel decomposes into magnetite and ilmenite at higher carbon additions (Reaction 18 and 19 in Table 4). In this process, some Ti4+ from rutile or ilmenite diffuses into the magnetite lattice, leading to the stabilization of titanomagnetite (2FeO·3Fe3O4·TiO2, JCPDS: 01-075-1374) rather than pure magnetite, as titanium readily substitutes into the spinel structure under reducing conditions19,20). This suggests that the reduction atmosphere significantly influences phase transformations and, consequently, the separation efficiency of Ti- and Fe-bearing phases, as shown in the Baur-Glässner diagram for the hematite–rutile system in Fig. 6. Therefore, the subsequent experiments were conducted with a carbon dose of 20%.
3.4. Effect of the temperature in the magnetizing roasting
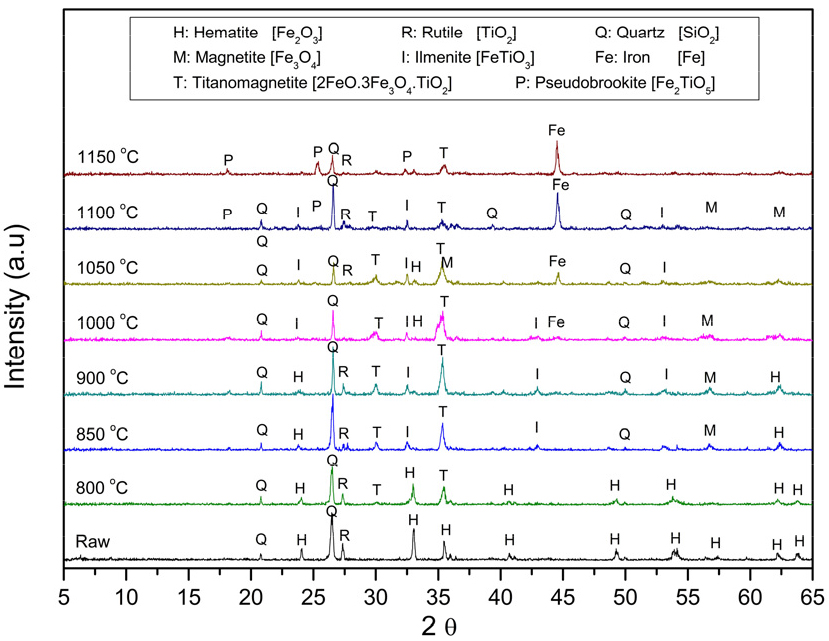
The effect of carbothermic reduction at various temperatures on the magnetic separation of the raw material, mixed with 20 wt.% graphite, is shown in Fig. 10. Fig. 10 presents the XRD patterns, which show that the magnetic fraction mainly consists of hematite, rutile, and quartz at room temperature. As the roasting temperature rises, carbon burns to produce CO, as highlighted in Fig. 6. Hematite gradually reduces to magnetite (Fe3O4, JCPDS: 19-0629), ilmenite (FeTiO3, JCPDS: 29-0733), and titanomagnetite, with new phases such as pseudobrookite (Fe2O3·TiO2, JCPDS: 01-070-2728), and metal iron (Fe, JCPDS: 06-0696) appearing above 1,000 °C. Titanomagnetites are common magnetic minerals found in igneous and metamorphic rocks. They form a complete solid solution above approximately 923 K (650 °C) between the end-members magnetite (Fe3O4) and ulvöspinel (Fe2TiO4), as illustrated in Fig. 7.
The roasting temperature significantly influences the upgrading of iron into the magnetic fraction during magnetic separation. These changes increase magnetic susceptibility, thereby facilitating the separation of iron-bearing phases from gangue minerals such as quartz. Titanium recovery increases notably between 850–1,000 °C due to the reaction of rutile and hematite to produce ilmenite and further effective separation from reduced gangue minerals. At the same time, iron recovery also improves within this range but fluctuates due to continued reductions in the metal iron content. At temperatures above 1,050 °C, excessive reduction leads to the formation of metallic iron and complex phases, thereby reducing the efficiency of selective recovery. Overall, the optimal separation occurs around 900–1,050 °C, where phase transformations maximize the recovery of both Fe and Ti, confirming findings from other studies8,9,10,11).
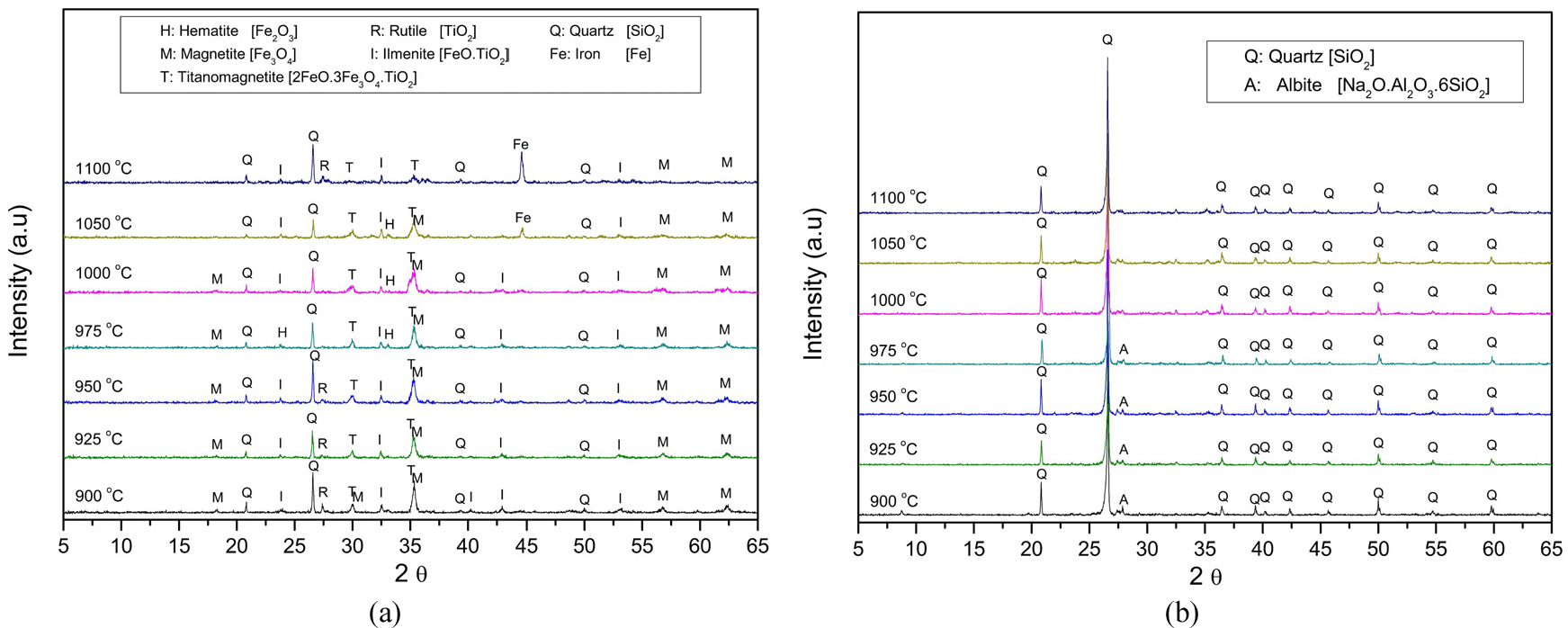
Since the optimal separation occurs between 900 and 1,050 °C, this range was examined in greater detail and separated by magnetic separation at 0.8 T. Fig. 11 shows the results. Fig. 11a displays the XRD patterns of the magnetic concentrate, indicating that hematite gradually transforms into magnetite, ilmenite, and titanomagnetite, with metallic iron appearing above 1000 °C. These reductions are mainly driven by the Boudouard reaction (Reaction 7 in Table 3), which produces CO as the reducing agent; CO reduces hematite to magnetite and, at higher temperatures, further to metal iron. Because gas-solid contact (CO(g) - Fe2O3) is naturally greater than solid-solid contact (C – Fe2O3), the reduction by carbon mainly occurs through the gas-solid contact, making it necessary to produce CO to enhance the reduction of hematite16). Fig. 11b shows the non-magnetic fraction, which is primarily composed of rutile and quartz, confirming that titanium minerals remain in the non-magnetic product. Meanwhile, iron-bearing phases shift to the magnetic fraction.
Excessive temperature causes over-reduction of the iron ore, forming Fe3O4, FeO, and Fe, which then attach to quartz. This attachment reduces magnetic separation efficiency and increases quartz content in the magnetic fraction16,17,18). Overall, this narrower range suggests that the optimal roasting window for selective Fe–Ti separation lies between 925 and 975 °C, at which iron is sufficiently reduced to magnetic phases by CO produced via the Boudouard reaction.
4. Conclusions
The magnetizing roasting characteristics of the ore and the magnetic separation properties of the roasted product were investigated to enhance the grade and yield of titanium oxide through magnetic separation of titanium ore from the Taebaek Myeon-San Layer titanium deposit. The following consistent conclusions are reached:
1. The study identified the raw material as a heterogeneous mixture of rutile (24.55 wt.%), hematite (32.96 wt.%), and significant gangue minerals, including quartz (28.93 wt.%), requiring a phase transformation for magnetic separation.
2. When titanium-iron minerals were magnetically separated, magnetite and metallic iron showed strong ferromagnetism, whereas increasing Ti content gradually weakened magnetic order, leading to ferrimagnetic or weakly magnetic phases. The order of magnetism in titanium-iron minerals is: metallic iron > magnetite > titanomagnetite > ilmenite > ulvospinel > hematite > pseudobrookite > rutile.
3. Carbothermic magnetizing roasting of the titanium ore promotes the reduction of hematite and rutile into magnetic minerals. Titanium recovery increases significantly between 850 and 1,000 °C due to the reaction between rutile and hematite, which forms ilmenite and enables more effective separation from reduced gangue minerals. At low carbon levels, hematite and rutile are the predominant phases, whereas as carbon input increases, ulvöspinel begins to form. At higher carbon levels, excess CO produced via the Boudouard reaction causes ulvöspinel to decompose into magnetite and ilmenite.
4. During this process, some Ti from rutile or ilmenite diffuses into the magnetite lattice, stabilizing titanomagnetite instead of pure magnetite. However, temperatures above 1,050 °C led to excessive reduction, yielding metallic iron and complex phases that reduced separation efficiency. The ideal roasting temperature was found to be between 925 and 975 °C, balancing phase transformations and magnetic separation effectiveness. In this range, hematite was reduced to magnetite, ilmenite, and titanomagnetite, while minimizing the formation of metallic iron and other complex phases that hinder selectivity.
Overall, these results emphasize that the magnetizing roasting process should aim to generate titanomagnetite, ilmenite, and ulvöspinel, in this order of preference, rather than metallic iron. These phases exhibit high magnetic susceptibility while maintaining phase selectivity, allowing efficient separation of Ti-bearing minerals such as rutile. Careful control of temperature, residence time, and carbon dosage is therefore essential to direct phase evolution toward these desirable magnetic oxides and maximize sustainable recovery of Fe–Ti minerals from the Myeon-San Layer deposit. Building on these fundamental findings, our future work will focus on enhancing the grade and yield of the titanium oxide through magnetic separation of the titanium ore from the Taebaek Myeon-San deposit.