1. Introduction
2. Materials and Methods
2.1. Raw Materials
2.2. Synthesis of ferrous sulfate
2.3. Preparation of Cr(VI)-Spiked Cement Systems
2.4. Extraction and Analysis of Water-Soluble Cr(VI)
2.5. Characterization of Synthesized Ferrous Sulfate
3. Results
3.1. Phase Identification of Sludge-Derived Ferrous Sulfate
3.2. Dosage-Dependent Reduction of Water-Soluble Cr(VI)
3.3. Influence of Ferrous Sulfate Hydration State on Reduction Performance
4. Discussion
4.1. Mechanism of Fe(II)-Mediated Cr(VI) Reduction in Alkaline Cement Systems
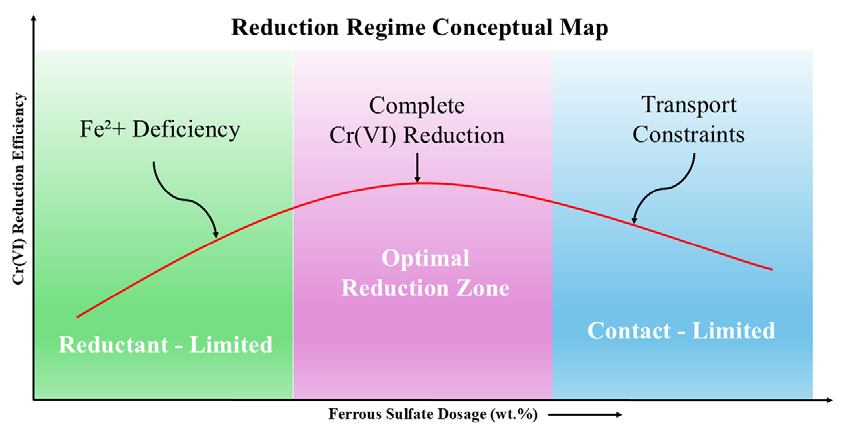
4.2. Dosage-Dependent Reduction Regimes
4.3. Role of Ferrous Sulfate Hydration State
4.4. Performance of Sludge-Derived Ferrous Sulfate and Practical Implications
5. Conclusions
1. Introduction
Hexavalent chromium (Cr(VI)) is widely recognized as one of the most hazardous trace constituents in cementitious materials due to its high solubility, mobility and toxicity under alkaline conditions1,2,3). In Ordinary Portland Cement (OPC), chromium is introduced in small amounts from raw materials, fuels, and refractory linings during clinker production4,5). Under the strongly oxidizing conditions of clinkerization, a fraction of chromium is converted to its hexavalent state. Upon hydration, Cr(VI) exists predominantly as chromate (CrO42-) species in the highly alkaline pore solution (pH > 12.5), where it remains readily water-soluble and bioavailable6,7,8). Exposure to water-soluble Cr(VI) is associated with severe occupational health risks, particularly allergic contact dermatitis among construction workers, leading to strict regulatory limits worldwide, including the European Union limit of 2 ppm for water-soluble Cr(VI) in cement9).
Chemical reduction of Cr(VI) to trivalent chromium (Cr(III)) is regarded as the most reliable mitigation strategy, as Cr(III) exhibits substantially lower toxicity and extremely low solubility in alkaline environments10). Once reduced, Cr(III) rapidly precipitates as chromium hydroxide and can be further immobilized through incorporation into cement hydration products such as calcium silicate hydrate (C–S–H) and aluminate phases11,12). Among various reducing agents, ferrous iron (Fe(II)) has been widely adopted due to its favorable redox potential, compatibility with cement chemistry, and practical applicability. Ferrous sulfate has been extensively used in industrial practice as an effective Cr(VI) reducing additive13).
However, the performance of ferrous sulfate in alkaline cement systems is governed not only by its dosage but also by its physicochemical properties14). The state of ferrous sulfate hydration affects its dissolution rate, stability of Fe(II) species, and interaction with chromate ions in the pore solution. In highly alkaline environments, rapid hydrolysis and oxidation of Fe(II) can limit its effective availability, even at relatively high dosages. Consequently, Cr(VI) reduction behavior may shift from a reductant-limited regime at low dosages to a contact or transport-limited regime when sufficient Fe(II) is present, but reaction efficiency is constrained by physical or kinetic factors15).
Beyond reduction efficiency, increasing attention has been directed toward sustainable materials processing and circular resource utilization in the cement and steel industries. Blast furnace (BF) and converter furnace (CF) sludge are generated in large quantities and are typically treated as low-value waste despite their high iron content. The conversion of these sludges into ferrous sulfate through reaction with waste sulfuric acid represents an attractive approach for waste valorization, reducing environmental burden while supplying functional reducing agents. Nevertheless, the influence of sludge-derived ferrous sulfate composition and hydration on Cr(VI) reduction performance in cementitious systems has not yet been systematically clarified16,17).
In this study, ferrous sulfate synthesized from BF and CF sludge using waste sulfuric acid is evaluated as a reducing agent for hexavalent chromium in alkaline cement systems. Emphasis is placed on the combined effects of hydration state and dosage on Fe(II) availability and Cr(VI) reduction efficiency. By employing a controlled Cr(VI)-spiked cement system and correlating phase composition, hydration state, and reduction behavior, this work provides mechanistic insight into Fe(II)-mediated chromate reduction and demonstrates a sustainable strategy for effective chromium control in cement-based materials.
2. Materials and Methods
2.1. Raw Materials
Ordinary Portland Cement (OPC) conforming to KSL 5201 was used as the base cementitious material in this study. The cement was stored in sealed containers under laboratory conditions to prevent moisture uptake and unintended hydration prior to sample preparation. Hexavalent chromium was introduced in a controlled manner using analytical-grade chromium trioxide (CrO3), which served as a well-defined and reproducible source of Cr(VI). Blast furnace (BF) sludge and converter furnace (CF) sludge obtained from a domestic steel production facility were used as iron-bearing raw materials for the synthesis of ferrous sulfate.
Preliminary characterization indicated that BF sludge contained a relatively high fraction of ferric iron (Fe3+), which limited the direct formation of ferrous sulfate (FeSO4) when BF sludge was used alone. To enhance the availability of ferrous iron (Fe2+) during the synthesis process, BF sludge was mixed with CF sludge, which provided a more favorable redox balance for FeSO4 formation. In this study, the sample designated as BF5 corresponds to a mixed-sludge composition consisting of 50 wt.% BF sludge and 50 wt.% CF sludge. This mixed composition enabled more stable formation of ferrous sulfate phases suitable for use as Cr(VI) reducing agents in alkaline cement systems.
Waste sulfuric acid (H2SO4) supplied from an industrial process stream was used as the acid source for reacting with the iron-containing sludge mixture. This approach allowed the synthesis of ferrous sulfate while simultaneously valorizing industrial by-products. The synthesized ferrous sulfate samples were stored in airtight containers and handled under low-humidity conditions to minimize premature oxidation of Fe(II). Deionized water was used exclusively for Cr(VI) extraction and analytical procedures and was not involved in the preparation or dry blending of cement samples.
2.2. Synthesis of ferrous sulfate
Ferrous sulfate was synthesized using blast furnace (BF) and converter furnace (CF) sludge as iron sources through reaction with waste sulfuric acid. Prior to synthesis, the sludge samples were dried, homogenized, and accurately weighed, and then placed in a chemically resistant reaction vessel. Waste sulfuric acid (H2SO4) was gradually added to the sludge under continuous stirring to promote a controlled acid–solid reaction and dissolution of iron-bearing phases.
The reaction between the sludge mixture and waste sulfuric acid was conducted at approximately 70 °C under continuous stirring to promote the dissolution of iron-bearing phases. After filtration, the ferrous-rich solution was subjected to controlled crystallization at 5 °C to induce the precipitation of ferrous sulfate. The resulting crystals were subsequently collected and dried at 40 °C for 12 h under controlled conditions to minimize oxidation of Fe(II).
The precipitated solids were collected by filtration and dried under controlled temperature and humidity conditions to minimize the oxidation of Fe(II). Samples prepared under different synthesis and drying conditions were designated as BF5, BF6, BF8 and BF9 for clarity and traceability. The characteristics of the synthesized ferrous sulfate samples, including sludge source, identified crystalline phase, hydration state, and expected Fe2+ availability, are summarized in Table 1.
Table 1.
Characteristics of ferrous sulfate samples synthesized from mixed BF/CF sludge used as Cr(VI) reducing agents in alkaline cement systems. The samples differ in hydration state identified by XRD analysis, which influences the expected availability of Fe2+ during chromate reduction
The phase composition and hydration state of the synthesized ferrous sulfate samples were further identified by X-ray diffraction (XRD) analysis. This synthesis route enabled the effective conversion of iron-rich industrial sludge and waste sulfuric acid into functional ferrous sulfate suitable for use as a Cr(VI) reducing agent in alkaline cement systems.
2.3. Preparation of Cr(VI)-Spiked Cement Systems
Cr(VI)-spiked cement systems were prepared using a dry-blending approach to ensure accurate control of composition and to prevent premature cement hydration or oxidation reactions. Ordinary Portland cement was weighed for each batch and mixed with a pre-determined amount of chromium trioxide (CrO₃), corresponding to a nominal Cr(VI) concentration of 20 ppm relative to the cement mass. Under alkaline conditions, Cr(VI) exists predominantly as water-soluble chromate species in Fig. 1a.
The cement and Cr(VI) source were homogenized by gentle tumbling in sealed polyethylene containers for 5 min to achieve uniform chromium distribution while avoiding moisture exposure. Subsequently, synthesized ferrous sulfate was added to the dry cement mixture at specified dosages, expressed as weight percent relative to cement mass. Upon addition, ferrous sulfate dissolves and supplies Fe2+ ions to the system in Fig. 1b. The mixtures were further blended for an additional 5–7 min under dry conditions to ensure homogeneous dispersion of the reducing agent.
The reduction of Cr(VI) proceeds via electron transfer from Fe2+ to chromate species, resulting in the oxidation of Fe2+ to Fe3+ and the concomitant reduction of Cr(VI) to Cr(III) in Fig. 1c. The reduced Cr(III) species are subsequently immobilized through precipitation as chromium hydroxide and co-precipitation with ferric hydroxides, forming stable and low-toxicity solid phases within the cement matrix in Fig. 1d. No water was introduced during sample preparation, thereby preventing early cement hydration and minimizing oxidation of Fe(II) prior to Cr(VI) reduction.
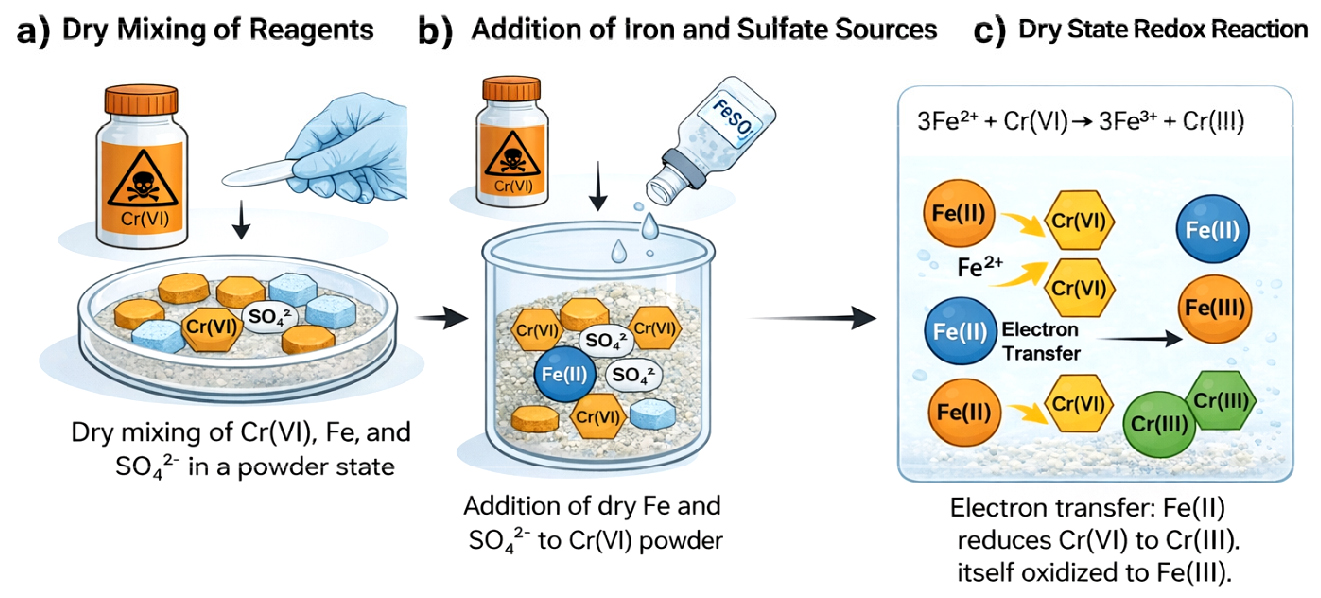
Fig. 1.
Schematic illustration of the Fe(II)-mediated reduction pathway of hexavalent chromium (Cr(VI)) in a dry-blended cement system: (a) dry mixing of cement with a Cr(VI) source, Fe(II) source, and sulfate-bearing additive; (b) homogeneous distribution of the solid reactants in the dry state prior to hydration; and (c) redox interaction in the dry matrix, where Fe(II) reduces Cr(VI) to Cr(III) while being oxidized to Fe(III).
All prepared cement blends were stored in airtight containers under controlled laboratory conditions and analyzed within a short time frame to minimize changes in chemical speciation before Cr(VI) extraction and quantification. This dry-preparation method enabled reproducible evaluation of dosage-dependent Cr(VI) reduction behavior and facilitated systematic investigation of the influence of ferrous sulfate hydration state on reduction performance in alkaline cement systems.
2.4. Extraction and Analysis of Water-Soluble Cr(VI)
The concentration of water-soluble hexavalent chromium in the prepared cement samples was determined using a standardized extraction and colorimetric analysis procedure. For each test, a predetermined amount of the dry cement blend was mixed with deionized water at a fixed solid-to-liquid ratio and agitated for a specified extraction period to allow dissolution of water-soluble Cr(VI) species under alkaline conditions generated by cement hydration.
After extraction, the suspension was filtered to remove solid residues, and the clear filtrate was collected for Cr(VI) analysis. The concentration of hexavalent chromium in the extracted solution was quantified using the diphenylcarbazide (DPC) colorimetric method. In this method, Cr(VI) reacts with diphenylcarbazide under acidic conditions to form a purple-colored complex, the absorbance of which was measured using a UV–visible spectrophotometer at a wavelength of 540 nm.
Calibration curves were established using standard Cr(VI) solutions prepared from potassium dichromate, and all measurements were performed within the linear range of the calibration. Each sample was analyzed in triplicate to ensure reproducibility, and the reported Cr(VI) concentrations represent mean values with corresponding standard deviations.
This analytical approach enabled reliable quantification of residual water-soluble Cr(VI) and provided the basis for evaluating dosage-dependent reduction efficiency and the influence of ferrous sulfate hydration state on Cr(VI) reduction behavior in alkaline cement systems.
2.5. Characterization of Synthesized Ferrous Sulfate
The phase composition and hydration state of the ferrous sulfate synthesized from BF and CF sludge were characterized by X-ray diffraction (XRD). Powder XRD measurements were performed using an X-ray diffractometer equipped with a Cu Kα radiation source (λ = 1.5406 Å), operated at appropriate voltage and current conditions. Diffraction patterns were collected over a 2θ range of 5–70° with a fixed step size and scanning rate.
Prior to analysis, the synthesized ferrous sulfate samples were gently ground to obtain homogeneous powders and minimize preferred orientation effects. To limit oxidation and dehydration during measurement, samples were prepared immediately before analysis and handled under controlled laboratory conditions. Phase identification was conducted by comparing the obtained diffraction patterns with standard reference data for ferrous sulfate hydrates, including FeSO4·H2O and FeSO4·4H2O.
The XRD analysis enabled clear distinction between different hydration states of ferrous sulfate produced under varying synthesis and drying conditions. These phase identification results were subsequently correlated with Cr(VI) reduction performance to elucidate the role of ferrous sulfate hydration state and Fe(II) availability in alkaline cement systems.
3. Results
3.1. Phase Identification of Sludge-Derived Ferrous Sulfate
The crystalline phases and hydration states of the synthesized ferrous sulfate samples were identified by X-ray diffraction (XRD) analysis to confirm the effectiveness of the synthesis route and to evaluate phase-dependent characteristics relevant to Cr(VI) reduction. The XRD patterns of samples BF5, BF6, BF8 and BF9 are presented in Fig. 2. All samples exhibited well-defined diffraction peaks, indicating the formation of crystal-line ferrous sulfate phases from blast furnace sludge through reaction with waste sulfuric acid.
As shown in Fig. 2, samples BF5 and BF6 display diffraction peaks that are consistent with FeSO4·4H2O (tetrahydrate). The presence of these peaks suggests that crystallization and drying under milder conditions favors the stabilization of higher hydration states. In contrast, samples BF8 and BF9 exhibit diffraction patterns characteristic of FeSO4·H2O (monohydrate), indicating partial dehydration of ferrous sulfate under more severe drying conditions. No additional peaks associated with ferric sulfate or iron oxide impurities were detected, confirming that oxidation of Fe(II) during synthesis and post-treatment was effectively minimized.
The variation in hydration state observed in Fig. 2 is expected to have a direct impact on the physicochemical behavior of the synthesized ferrous sulfate in alkaline cement systems. Hydrated phases such as FeSO4·4H2O generally exhibit higher solubility and faster dissolution kinetics, which can enhance the immediate availability of Fe2+ ions upon contact with alkaline pore solutions. Conversely, the monohydrated phases identified in BF8 and BF9 may dissolve more slowly, potentially leading to delayed but sustained release of Fe2+ ions.
These phase-dependent dissolution characteristics are particularly relevant for Cr(VI) reduction in cementitious environments, where reaction kinetics and Fe2+ availability play critical roles in determining reduction efficiency. The XRD results presented in Fig. 2 therefore provide a fundamental basis for interpreting the differences in Cr(VI) reduction performance among the synthesized ferrous sulfate samples, as discussed in the following sections.
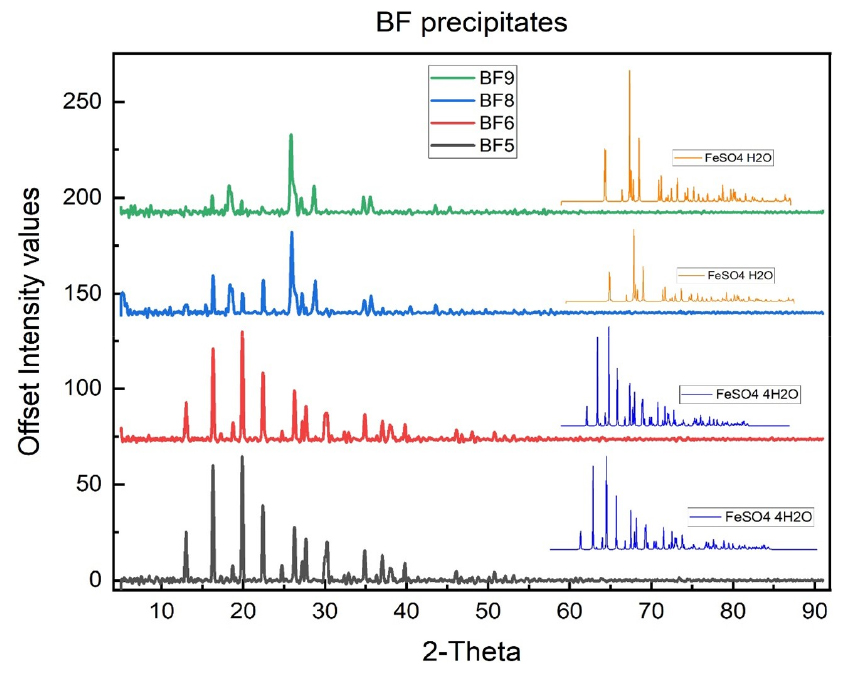
Fig. 2.
X-ray diffraction (XRD) patterns of ferrous sulfate synthesized from BF and CF sludge under different synthesis and drying conditions. Samples BF5 and BF6 are dominated by tetrahydrate ferrous sulfate (FeSO4·4H2O), whereas samples BF8 and BF9 primarily consist of monohydrated ferrous sulfate (FeSO4·H2O).
3.2. Dosage-Dependent Reduction of Water-Soluble Cr(VI)
The effect of ferrous sulfate dosage on the reduction of water-soluble Cr(VI) in ordinary Portland cement was evaluated by measuring residual Cr(VI) concentrations after extraction. The dosage-dependent reduction tests were conducted using the BF5 sample, which was identified as FeSO4·4H2O based on XRD analysis. This sample was selected because it exhibited higher solubility and more stable Fe2+ availability under the experimental conditions. The variation in residual Cr(VI) concentration and the corresponding reduction efficiencies as a function of ferrous sulfate dosage are shown in Fig. 3, and the quantitative results are summarized in Table 2.
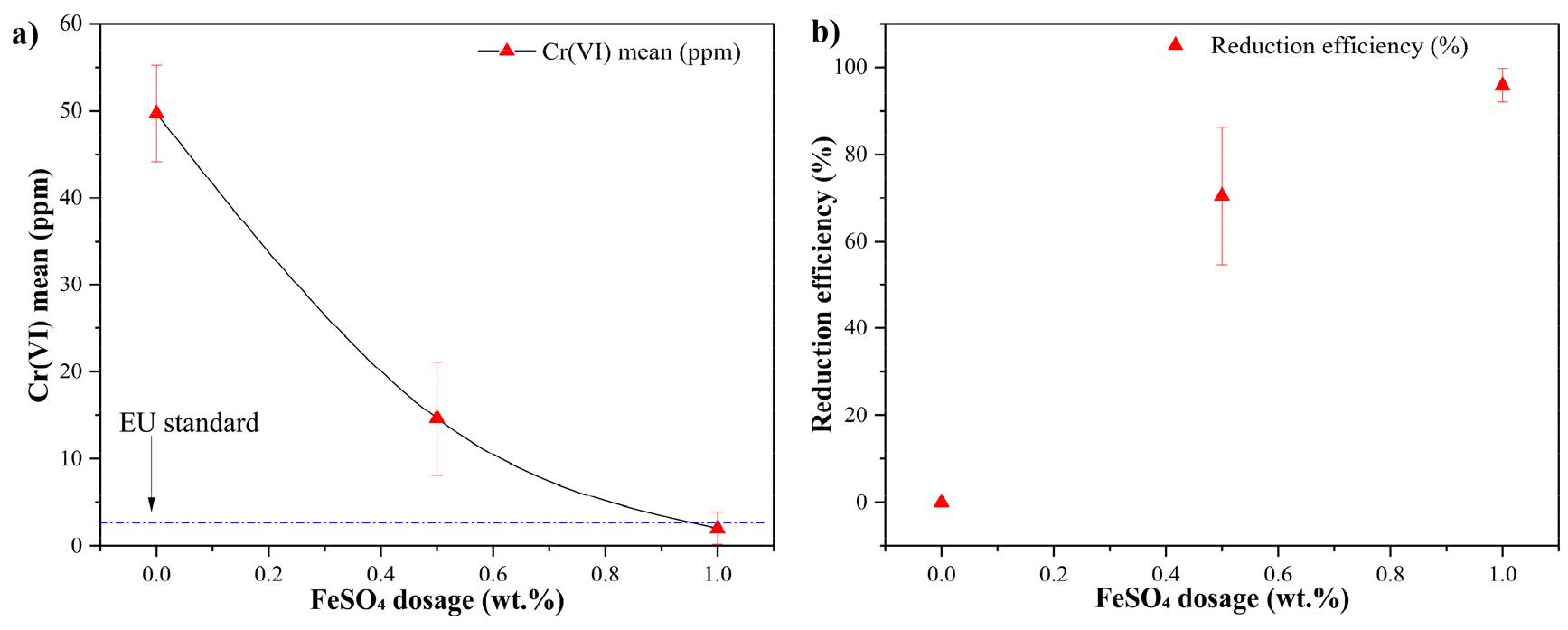
Fig. 3.
Residual water-soluble Cr(VI) concentration in Ordinary Portland Cement as a function of ferrous sulfate dosage. (b) Corresponding Cr(VI) reduction efficiency. Data represents mean values with standard deviations obtained from triplicate measurements. The dashed horizontal line indicates the European Union regulatory limit for water-soluble Cr(VI) in cement (2 ppm).
Table 2.
Effect of ferrous sulfate dosage on the residual water-soluble Cr(VI) concentration and corresponding reduction efficiency in the BF5-treated cement system
| Ferrous sulfate dosage (wt.%) | Residual Cr(VI) (ppm) | Standard deviation (ppm) | Reduction efficiency (%) |
| 0 | 49.7 | 5.5 | 0 |
| 0.5 | 14.7 | 12.6 | 71 |
| 1.0 | 2.1 | 1.9 | 95-96 |
The Cr(VI)-spiked reference cement prepared without ferrous sulfate exhibited a high mean water-soluble Cr(VI) concentration of 49.7 ± 5.5 ppm, confirming the presence of readily soluble chromate species under alkaline cement conditions. Upon addition of 0.5 wt.% ferrous sulfate, the residual Cr(VI) concentration decreased substantially to 14.7 ± 12.6 ppm, corresponding to a reduction efficiency of approximately 71%. This pronounced decrease suggests that, at low dosages, the reduction process is primarily governed by Fe2+ availability, consistent with a reductant-limited regime.
Further increasing the ferrous sulfate dosage to 1.0 wt.% resulted in an additional decrease in residual Cr(VI), lowering the concentration to 2.1 ± 1.9 ppm. At this dosage level, the Cr(VI) concentration approached or fell below the European Union regulatory limit of 2 ppm for water-soluble Cr(VI) in cement, while the corresponding reduction efficiency reached approximately 95–96%. These results demonstrate highly effective chromium detoxification under optimized conditions.
In contrast, samples BF8 and BF9, which are dominated by FeSO4·H2O (monohydrate), exhibited lower Cr(VI) reduction performance during preliminary tests. This behavior is attributed to the lower dissolution rate of the monohydrate phase in water under the experimental conditions, which limits the availability of Fe2+ ions required for chromate reduction. Therefore, BF5 was selected as the representative sample for the dosage-dependent reduction evaluation.
3.3. Influence of Ferrous Sulfate Hydration State on Reduction Performance
The reduction of water-soluble Cr(VI) in alkaline cement systems by ferrous sulfate is governed by the stoichiometric availability of Fe(II) relative to Cr(VI). According to the redox reaction mechanism, Fe2+ acts as the primary electron donor, reducing Cr(VI) to Cr(III) while being oxidized to Fe(III). Therefore, sufficient Fe(II) dosage is required to ensure complete reduction of Cr(VI) species present in the cement matrix.
At lower ferrous sulfate dosages, the reduction behavior is predominantly controlled by Fe(II) availability. As observed at 0.5 wt.% dosage, a substantial decrease in residual Cr(VI) concentration was achieved; however, incomplete reduction and relatively large variability were still evident. This behavior suggests that the system operates within a reductant-limited regime, where localized depletion of Fe(II) restricts full conversion of Cr(VI) to Cr(III).
With increasing ferrous sulfate dosage, the system transitions toward a stoichiometrically sufficient regime. At a dosage of 1.0 wt.%, residual Cr(VI) concentrations approached or fell below the regulatory limit for water-soluble Cr(VI) in cement, indicating that adequate Fe(II) was available to drive near-complete reduction. Under these conditions, excess Fe(II) ensures sustained reduction capacity and minimizes the influence of local heterogeneities within the cement matrix.
In addition to stoichiometric effects, the hydration state of ferrous sulfate may further influence effective Fe(II) availability through differences in dissolution behavior. More highly hydrated phases are expected to dissolve more rapidly, providing Fe2+ ions at earlier stages, whereas less hydrated phases may contribute to slower but prolonged Fe(II) release. These combined stoichiometric and phase-dependent effects play a critical role in determining overall Cr(VI) reduction efficiency.
Overall, the results indicate that effective Cr(VI) detoxification in alkaline cement systems requires not only the presence of ferrous sulfate but also an optimized dosage that satisfies stoichiometric Fe(II) demand. This understanding provides a framework for designing efficient Cr(VI) reduction strategies using industrially derived ferrous sulfate in cementitious applications.
4. Discussion
4.1. Mechanism of Fe(II)-Mediated Cr(VI) Reduction in Alkaline Cement Systems
The reduction of hexavalent chromium in alkaline cement systems proceeds primarily through electron transfer from ferrous iron (Fe(II)) to chromate species, followed by precipitation and immobilization of the reduced chromium. Upon cement hydration, a highly alkaline pore solution (pH > 12.5) is established, in which Cr(VI) predominantly exists as water-soluble chromate ions. When ferrous sulfate dissolves, Fe2+ ions become available for redox reactions with chromate, resulting in the oxidation of Fe(II) to Fe(III) and the reduction of Cr(VI) to trivalent chromium (Cr(III)).
Under alkaline conditions, the reduced Cr(III) rapidly precipitates as chromium hydroxide due to its extremely low solubility, as described in Equation (1). Simultaneously, Fe(III) generated during the redox process undergoes hydrolysis and precipitates as ferric hydroxide Equation (2). These hydroxide phases play a critical role in immobilizing chromium within the cement matrix.
Beyond direct precipitation, ferric hydroxide phases further enhance chromium stabilization through co-precipitation and surface sorption of Cr(III), thereby contributing to long-term immobilization. The coupled redox–precipitation mechanism effectively converts mobile and toxic Cr(VI) into stable, low-toxicity solid phases, explaining the substantial decrease in water-soluble Cr(VI) observed upon ferrous sulfate addition.
The overall stoichiometry of the Fe(II)-mediated reduction of chromate in alkaline conditions can be summarized by the global reaction shown in Equation (3), which incorporates both electron transfer and hydroxide precipitation processes. This mechanistic framework provides a clear basis for understanding the dosage-dependent Cr(VI) reduction behavior observed in the experimental results.
4.2. Dosage-Dependent Reduction Regimes
The results demonstrate a clear transition in Cr(VI) reduction behavior with increasing ferrous sulfate dosage. At low dosages (e.g. 0.5 wt.%), the reduction efficiency is primarily governed by Fe(II) availability indicating a reductant-limited regime. Under these conditions, insufficient Fe(II) is present to fully reduce all soluble chromate species, resulting in partial reduction and relatively high variability in residual Cr(VI) concentrations. Reduction efficiency in this regime can be quantitatively described using the definition given in Equation (4).
In the reductant-limited regime, Fe(II) supplied by ferrous sulfate is rapidly consumed by redox reactions, and the stoichiometric requirement for complete Cr(VI) reduction is not satisfied. In addition to direct electron transfer, stabilization of reduced chromium is promoted through co-precipitation with ferric hydroxides, as represented in Equation (5). However, when the molar availability of Fe(II) does not meet the stoichiometric condition expressed in Equation (6), complete Cr(VI) reduction cannot be achieved.
As the ferrous sulfate dosage increases to 1.0 wt.%, the system approaches a stoichiometrically sufficient regime in which Fe(II) availability is no longer the limiting factor. Under these conditions, residual Cr(VI) concentrations are reduced to near or below regulatory limits, and the reduction efficiency reaches approximately 95%. This dosage range corresponds to an optimal reduction zone, where both sufficient reductant supply and favorable reaction kinetics enable near-complete Cr(VI) detoxification.
Beyond the optimal dosage range, further increases in ferrous sulfate addition are not expected to significantly enhance efficiency. Instead, the reduction process becomes increasingly influenced by physical contact, mass transport, and kinetic limitations rather than reductant supply. This transition from a reductant-limited to a contact-limited regime is conceptually illustrated in Fig. 4, and the corresponding dosage-dependent reduction regimes are summarized in Table 3.
Table 3.
Summary of dosage-dependent Cr(VI) reduction regimes in alkaline cement systems treated with ferrous sulfate
Overall, these findings highlight that effective Cr(VI) reduction in alkaline cement systems requires not only the presence of ferrous sulfate but also an optimized dosage that satisfies stoichiometric Fe(II) demand while minimizing inefficiencies associated with excess reductant. The conceptual framework presented here provides practical guidance for designing dosage strategies for Cr(VI) detoxification in cementitious applications.
4.3. Role of Ferrous Sulfate Hydration State
The state of the hydration of ferrous sulfate influences Cr(VI) reduction performance by affecting its dissolution behavior and Fe(II) release in alkaline cement systems. XRD analysis identified FeSO4·4H2O and FeSO4·H2O phases formed under different synthesis and drying conditions. More highly hydrated phases dissolve more readily, providing faster Fe2+ availability and promoting efficient early-stage reduction of soluble chromate species. In contrast, less hydrated phases dissolve more slowly, potentially delaying Fe(II) release and bilg(19)leading to kinetically limited reduction at intermediate dosages18).
Despite these kinetic differences, comparable overall Cr(VI) reduction efficiencies were achieved at sufficiently high ferrous sulfate dosages, indicating that the total Fe(II) supply ultimately governs the extent of Cr(VI) detoxification. Thus, hydration state primarily affects reduction kinetics rather than final reduction of capacity under optimized dosage conditions.
4.4. Performance of Sludge-Derived Ferrous Sulfate and Practical Implications
The high Cr(VI) reduction efficiency achieved using sludge-derived ferrous sulfate demonstrates that iron-rich industrial by-products can serve as effective and reliable alternatives to commercially produced agents in cementitious systems. Despite the inherent compositional heterogeneity typically associated with blast furnace (BF) and converter furnace (CF) sludge, the synthesized ferrous sulfate exhibited well-defined crystalline phases and reduction performance comparable to that of conventional ferrous sulfate materials. The absence of detectable iron oxide or ferric impurity phases in the XRD patterns further confirms that oxidation of Fe(II) was effectively controlled during synthesis and post-treatment, supporting the suitability of the synthesized products for cement applications.
From a practical standpoint, the ability to achieve approximately 95% reduction of water-soluble Cr(VI), with residual concentrations approaching or falling below regulatory limits, highlights the strong potential of waste-derived ferrous sulfate for industrial implementation. Effective Cr(VI) detoxification is critical for ensuring compliance with occupational health and safety regulations related to cement handling and use. The results of this study indicate that sludge-derived ferrous sulfate can meet these requirements while maintaining consistent reduction performance across different hydration states when applied at optimized dosages.
Beyond technical performance, the utilization of sludge-derived ferrous sulfate offers significant environmental and economic benefits. The conversion of iron-rich industrial sludge and waste sulfuric acid into a functional reducing agent represents a clear example of industrial waste valorization. This approach reduces the need for virgin raw materials, lowers waste disposal burdens, and aligns with circular economy and sustainability objectives in both the cement and steel industries.
Finally, the findings suggest that further optimization of synthesis and drying conditions to control the state of hydrate state of ferrous sulfate could enhance reduction reliability and robustness under field conditions. Tailoring hydration state may improve early-stage Fe(II) availability and reduction kinetics, particularly in large-scale cement processing where mixing efficiency and contact time can vary. Overall, the results support the feasibility of integrating sludge-derived ferrous sulfate into practical Cr(VI) mitigation strategies, offering a sustainable and effective solution for chromium detoxification in alkaline cement systems.
In addition to Cr(VI) reduction efficiency, the potential influence of sludge-derived additives on the physical properties and environmental safety of cementitious materials should also be considered. In this study, the dosage of ferrous sulfate was limited to ≤1 wt.% relative to the cement mass, which falls within the typical range used in industrial cement production for chromate reduction. At such low dosages, ferrous sulfate is not expected to significantly alter cement hydration kinetics or mechanical performance. Previous studies have reported that Fe(II)-based reducing agents generally have minimal effects on setting time and compressive strength when used at comparable levels.
Regarding environmental safety, trace metals potentially present in steelmaking sludge are likely to be effectively immobilized within the cement matrix under highly alkaline conditions. The precipitation of metal hydroxides and their incorporation into cement hydration products such as C–S–H and ferric hydroxide phases can further limit the mobility of these elements. Therefore, the use of sludge-derived ferrous sulfate as a reducing agent is not expected to pose significant environmental risks when applied within the dosage range investigated in this study.
5. Conclusions
1.Ferrous sulfate was successfully synthesized from iron-rich blast furnace and converter furnace sludge, producing well-crystallized phases with minimal Fe(II) oxidation.
2.The synthesized ferrous sulfate effectively reduced water-soluble Cr(VI) in alkaline cement systems, achieving approximately 95–96% reduction efficiency at a dosage of 1.0 wt.% and lowering residual Cr(VI) concentrations to near or below regulatory limits.
3.Cr(VI) reduction proceeded via Fe(II)-mediated electron transfer followed by precipitation of Cr(III), with the hydration state influencing early-stage kinetics while the total Fe(II) dosage governed the final extent of reduction.
4.These results demonstrate that sludge-derived ferrous sulfate is an effective and sustainable option for Cr(VI) mitigation in cementitious systems, offering a practical pathway for industrial waste valorization.