1. Introduction
2. Iridium
2.1. Iridium Resources
3. Commercial Ir Recovery Processes from PGMs byproducts
3.1. Lonmin Western Platinum Refinery
3.2. Johnson Matthey/Anglo American Process
3.3. Impala Platinum Refining Process
4. Hydrometallurgical Processes of Iridium
4.1. Leaching of Iridium
4.2. Separation of Ir
5. Conclusion
1. Introduction
Platinum Group Metals (PGMs) consists of six elements: Platinum (Pt), Palladium (Pd), Rhodium (Rh), Iridium (Ir), Ruthenium (Ru) and Osmium (Os). When gold (Au) and silver (Ag) are included, they are classified as precious metals (PMs). Among them, Ir is a transition metal with atomic number 77. It has the second-highest density (22.56 g/cm3), after Os (22.59 g/cm3)1). Also, Ir offers remarkable corrosion resistance, mechanical strength and chemical and electrical stability2). Because of its properties, Ir is widely used in various industrial sectors as shown in Table 1. Ir is also employed as an alloying element to enhance the hardness of Pt and Pd3).
Table 1.
In spite of broad industrial applications, Ir is extremely rare in the Earth’s crust, containing 0.3–5 ng/g8). Ir can be recovered as a byproduct during the refining of nickel and platinum ores9). The global supply of Ir is concentrated in specific regions such as South Africa and Zimbabwe. Also, price of Ir is highly unstable due to its much lower production compared to other PGMs. Since South Korea heavily depends on imports for PGMs, Ir recovery from secondary resources is essential to ensure a stable Ir supply for high-tech industries.
Ir can be recovered primarily through two methods: Pyrometallurgical and Hydrometallurgical methods. Pyrometallurgical process involves melting metal-containing materials at high temperature to separate impurities and refine the target metal. PGMs are recovered and concentrated through alloying with collectors such as Cu, Fe and Pb5). The process is suitable for large-scale processing as it consumes a large amount of energy10). In contrast, hydrometallurgical process uses chemical solvents to dissolve metals into aqueous phase, followed by refining and separation steps to extract the target metal. Hydrometallurgical process operates at lower temperatures, consuming less energy. However, the high corrosion resistance of Ir makes it difficult to leach.
To facilitate Ir leaching, Cl2 can be used as a strong oxidizing agent11). This method has significant drawbacks, such as equipment corrosion and safety risk. In response, researchers have been investigating thermal pretreatment techniques such as alkali fusion12,13,14,15). Alkali fusion involves heating insoluble metals with alkaline fluxes at high temperatures to convert them into a more soluble form16,17).
Hydrometallurgical refining process after leaching is essential to purify and concentrate Ir to high purity. Common refining methods include precipitation, solvent extraction and ion exchange, depending on the types and concentration of metals in leachate18). Among these, the solvent extraction method is widely used due to its high separation efficiency and ability to produce high-purity products19). However, the similar chemical properties of Ir and other PGMs makes their separation a significant challenge20). Therefore, previous research has focused on improving solvent extraction techniques for the efficient purification of Ir.
The purpose of this paper is to provide an overview of Ir resources and production. This paper also examines properties of Ir and explores its industrial application. Then, it investigates commercial processes to extract Ir from other PGMs. Finally, it reviews hydrometallurgical studies of leaching and solvent extraction through case studies.
2. Iridium
Ir, with an atomic number of 77, is a transition metal that belongs to group 9 (8B) of the periodic table and is classified as one of the PGMs. Its name originates from the Greek goddess of the rainbow, Iris, reflecting the vivid and varied colors of its compounds. Ir was first discovered in 1803 by British chemist Smithson Tennant, who identified it in the insoluble black residue remaining after Pt was dissolved in aqua regia during a purification process21).
Table 2 summarizes the physical and chemical properties of Ir1,22). Due to its extremely high melting and boiling points, Ir maintains its mechanical strength even at high temperatures. Furthermore, Ir has remarkable corrosion resistance, remaining stable in both strongly acidic and alkaline environments.
Table 2.
Ir typically states octahedral coordination complexes in the +3 and +4 oxidation states (Table 3)3). It forms a number of stable compounds with halogen and oxygen donors3,23). Therefore, it can exist a wide range of oxidation states, from –3 to +6, and has an electron configuration of [Xe]4f145d76s224).
Table 3.
Oxidation state of Ir3)
2.1. Iridium Resources
Ir commonly exists as an alloy with Pt or Os rather than its native form25). Typical Ir-containing alloys include Osmiridium (Ir80Os20, syserkskite), Iridosmium (Os80Ir20, nevyanskite), Platiniridium (Ir50Pt50) and Ruthenosmiridium (Ir, Os, Ru)26,27). Platiniridium typically appears as small cubes with rounded edges. Iridosmium and Osmiridium generally occur as flat, irregular grains and occasionally as hexagonal prisms26). Ir is also present in arsenide minerals such as Irsarite (IrAsS)27,28).
The major sources of PGMs are located in South Africa and Russia. The United States and Canada follow with smaller reserves. South Africa is the largest PGMs producer in the world, with approximately 63,000 metric tons of reserves according to the U.S. Geological Survey29). Russia follows with an estimated 16,000 metric tons of reserves29).
The Bushveld Complex in South Africa is the most significant source of PGMs, with ore bodies including the UG-2 Layer, Merensky Reef and Platreef. The UG-2 and Merensky Reef are the primary mining targets30). Table 4 provides data about the concentration of PGMs and base metals within these layers31).
Table 4.
Concentration of PMs and base metals of Merensky Reef and UG-231)
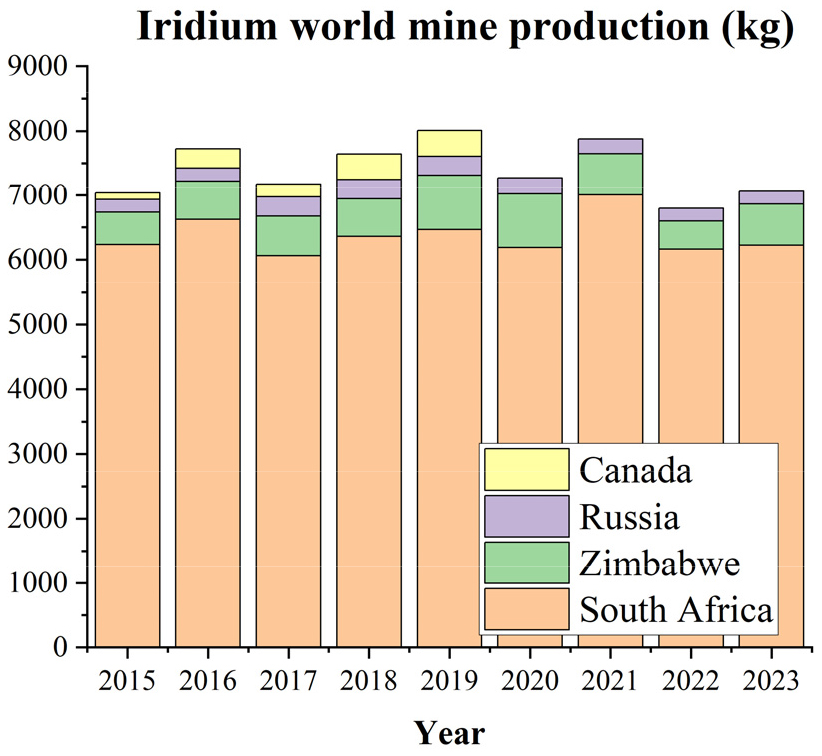
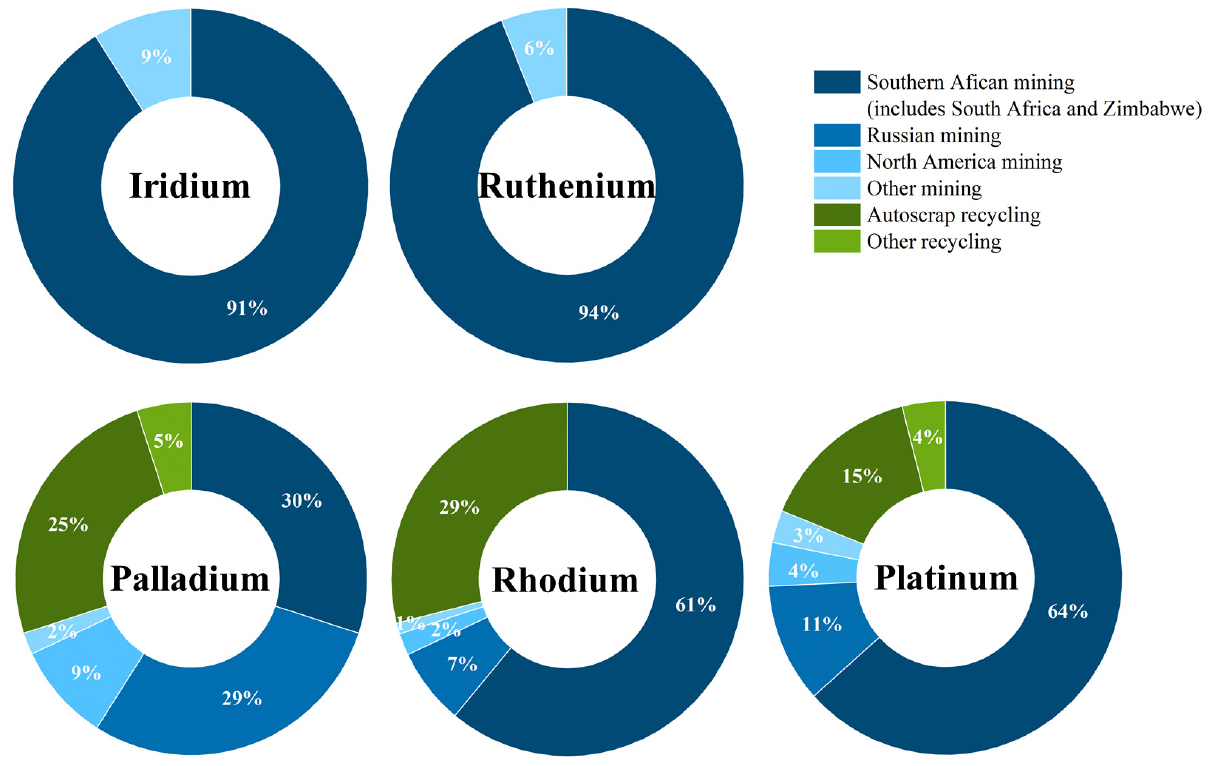
World mine production of Ir is shown in Fig. 1. Annual Ir production has fluctuated between 7,000 and 8,000 kg from 2015 to 2023. Especially, global Ir production amounted to approximately 7,060 kg per annum in 202332,33). South Africa holds the largest share of Ir reserves and production, followed by Zimbabwe and Russia. Pt, Pd and Rh are recovered not only from primary ores but also from secondary resources through recycling. However, the production of Ir and Ru remains heavily dependent on primary resources as shown in Fig. 28,28). Notably, approximately 91% of global Ir production comes from South Africa, indicating a strong dependence on geographically concentrated primary sources34). According to the U.S. Geological Survey, both elements are classified as critical minerals with high supply risk due to their limited availability and strong reliance on specific countries8).
3. Commercial Ir Recovery Processes from PGMs byproducts
Pt and Pd are the most commercially significant PGMs and are the main products of industrial recovery processes. Other PGMs – such as Ir, Rh, Ru and Os – are typically recovered as byproducts during the refining of Pt and Pd35). Among the commercial refining techniques for PGMs, precipitation, solvent extraction and ion exchange are widely applied.
3.1. Lonmin Western Platinum Refinery
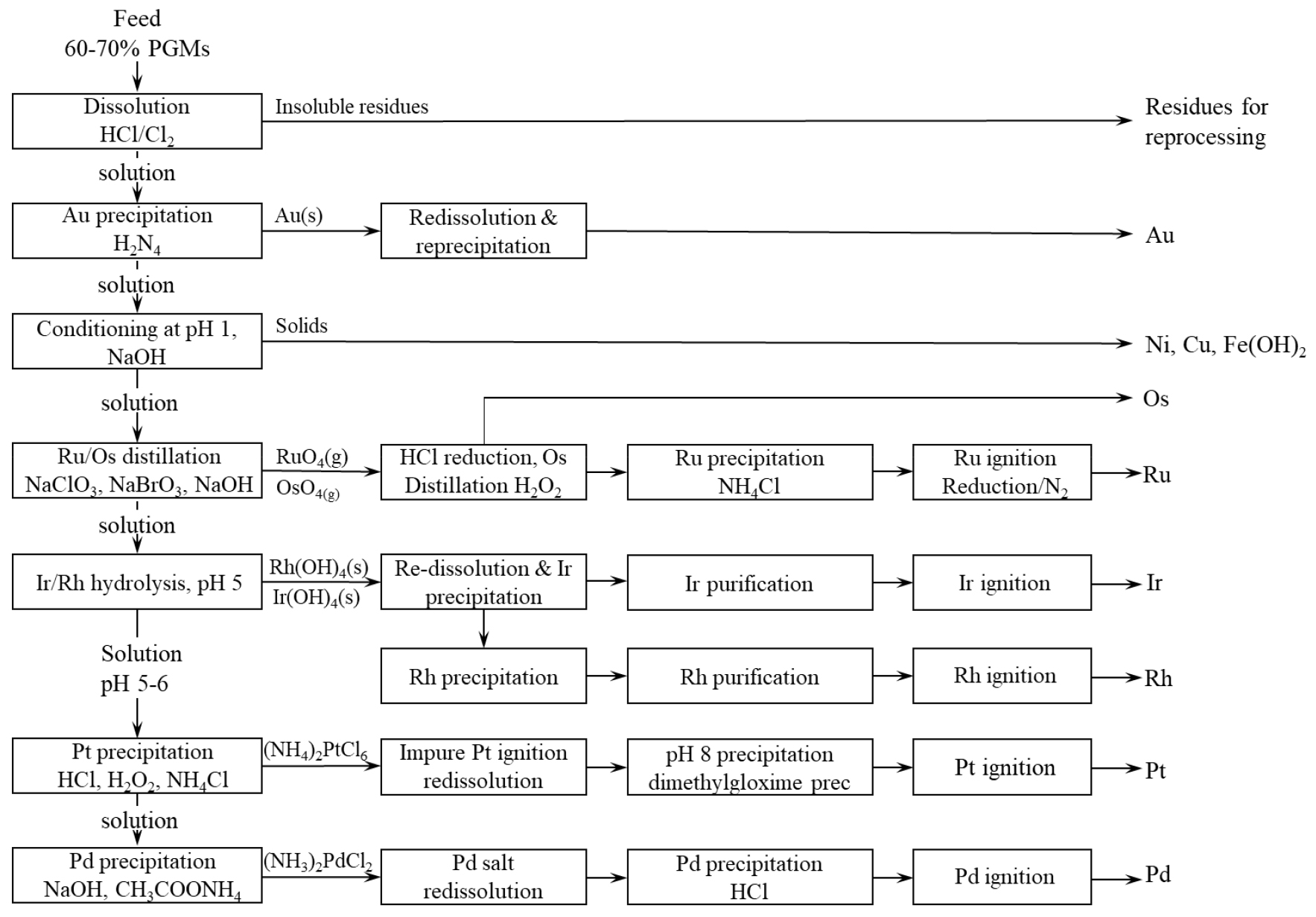
The Lonmin Western Platinum Refinery employs a precipitation method to separate and purify individual PGMs (Fig. 3)36,37). This process consists of leaching, filtration, distillation and precipitation steps, each of which is designed to selectively recover specific metals with high purity.
The initial concentrate, containing 65–70% PGMs, is leached at 65℃ for 6 hours using 6 M hydrochloric acid (HCl) and chlorine (Cl2). After leaching, dissolved chlorine is removed by blowing air through the solution, followed by filtration using a filter press. The remaining solids are redissolved under similar conditions. Au is first recovered by adding HCl and hydrazine (N2H4). N2H4 acts as a reducing agent, precipitating Au as crude gold. To enhance the purity, the precipitated Au is separated from co-precipitated PGMs through further dissolution and precipitation. After Au is precipitated, sodium hydroxide (NaOH) is added at ambient temperature to adjust the solution pH to 1. 20–40% of the base metals in the solution are precipitated and the slurry is filtered. The filter cake is reprocessed to recover any included PGMs.
The filtrate is then transferred to reactive distillation for Ru and Os recovery. Sodium chlorate (NaClO3) is added to the solution to oxidize Ru and Os to RuO4 and OsO4, respectively. Air is injected to strip RuO4 and OsO4 into the gas phase. The resulting gas stream moves through a reflux condenser, where water vapor is condensed and moves through two scrubbing columns. Later in the distillation, any unreacted Ru is further oxidized using NaBrO3 and reactive distillation continues until the concentration of Ru drops below 250 mg/L. In the first scrubbing column, HCl is added to reduce RuO4 and OsO4 to their non-volatile aqueous states. As a result, Ru and Os are removed from the gas phase to aqueous phase. In the second scrubbing column, the chlorine gas produced in the first scrubbing column is scrubbed from the gas phase by NaOH. The solution collected from the first scrubbing column is treated with hydrogen peroxide (H2O2) to re-oxidize Os into OsO4 and reduced again using HCl. As a result, Ru and Os are separated. Ru recovered from the scrub liquor is precipitated by the addition of ammonium chloride (NH4Cl), followed by ignition in a muffle furnace at 1000℃. The resulting product is cooled under a nitrogen atmosphere to produce pure Ru metal.
After Os and Ru removal from solution, Ir and Rh are recovered by raising the pH to 5 using sodium bicarbonate (NaHCO3), precipitating Ir(OH)4 and Rh(OH)4. These are filtered to obtain a filter cake containing Ir and Rh. The filter cake is dissolved and Ir is then selectively recovered as ammonium hexachloroiridate ([(NH4)2IrCl6]) by adding ammonium chloride (NH4Cl). Rh is precipitated as Claus’ salt (pentaamminechlororhodium(III) dichloride ([Rh(NH3)5Cl]Cl2)).
The remaining solution after Ir and Rh recovery proceeds for Pt recovery. HCl is added to lower the pH of the solution. H2O2 is added to adjust the redox potential to 900–1000 mV vs. SCE and ammonium chloride (NH4Cl) is used to precipitate Pt as (NH4)2PtCl6. The precipitate is filtered and then ignited to produce impure Pt sponge.
Finally, Pd is recovered from the filtrate. NaOH is added to adjust the pH to about 0.9. The solution is then boiled until the pH rises to about 3. Ammonium acetate (NH4OAc) is added to further increase pH. The solution is boiled to increase the pH to 4.2 and is subsequently cooled. The Pd precipitates as Pd(NH3)2Cl2. The slurry is filtered and the Pd undergoes final refining steps to yield high-purity metal36,37).
3.2. Johnson Matthey/Anglo American Process
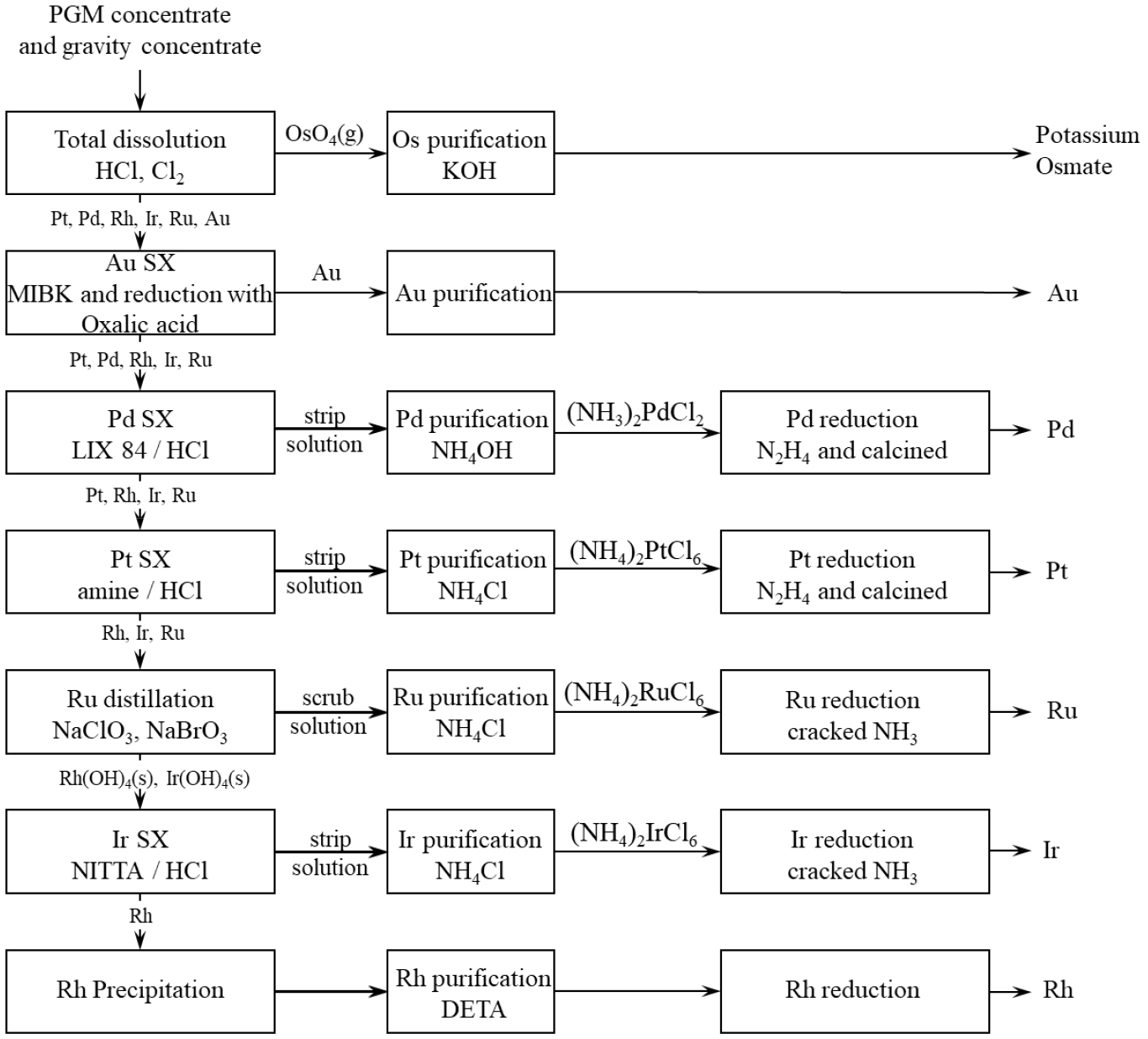
Jointly developed by Johnson Matthey and Anglo American, this process is characterized by the use of solvent extraction for the separation of PGMs as shown in Fig. 436,37). It treats the concentrate feed containing 50–60% PGMs. The feed is dissolved under conditions of 120℃ and 4 bar pressure using HCl and Cl2, resulting in the complete dissolution of PGMs and base metals. During this process, Os is oxidized to OsO4 and recovered from the off-gas by reaction with potassium hydroxide (KOH).
Au is selectively extracted from the filtrate using methyl isobutyl ketone (MIBK) at high acidity. Impurities such as Fe, Se, Te and Sb co-extracted with Au are removed by scrubbing with HCl. Au is then recovered from the organic phase by oxalic acid through direct reduction, producing metallic Au powder. The product is filtered, dried and further refined to improve purity.
After Au removal, Pd is extracted from Au raffinate using LIX84A (ketoxime, β-hydroxyoxime). An organic amine compound is added as an accelerator, but it decreases selectivity. Pd is stripped using 6 M HCl and precipitated as diamino-palladous dichloride by the addition of NH4OH. N2H4 is then used to reduce the salt to metal, followed by calcination for metal with purity in excess of 99.95%.
Pt is recovered using amine-based solvent extraction. To avoid co-extraction of Ir, Ir has to be in its lower (+3) oxidation state. Pt is stripped using 11 M HCl and precipitated as (NH4)2PtCl6 by adding NH4Cl. The precipitate is then redissolved and reduced with N2H4 to obtain metallic Pt.
After Pt removal, all the entrained organic is removed from the raffinate, as they form a potential risk. The raffinate is neutralized and oxidizing agents such as NaClO3 and NaBrO3 are added to oxidize Ru and residual Os to their tetroxides (RuO4, OsO4). These tetroxides are stripped to the gas phase that is pumped through the solution. Ru is absorbed in a packed column using HCl. Possible Os traces may be treated with H2O2 to selectively volatilize Os from the HCl solution. Os is then recovered in a similar method as Ru and precipitated using KOH. Ru is precipitated from the HCl solution using NH4Cl. The precipitate is calcined to from RuO2, which is then reduced to Ru sponge using cracked ammonia.
Following Ru and Os removal, the pH of the solution is increased to 5.8, leading to the precipitation of Ir and Rh as Ir(OH)4 and Rh(OH)4, respectively. The slurry is filtered and subsequently redissolved. Ir is oxidized to higher oxidative state using H2O2 and is extracted with a novel amide solvent, n-iso-tridecyltri-decanamide. Ir is then stripped using HCl and precipitated as (NH4)2IrCl6 with NH4Cl. After redissolution and reduction, the precipitate is calcined to form IrO2, which is further reduced with cracked ammonia to produce Ir sponge.
Finally, Rh remaining in the raffinate is precipitated using diethylene triamine (DETA). The Rh precipitate is redissolved and reprecipitated as chloropentamine rhodium(III) dichloride by adding NH4Cl. After calcination and reduction, Rh is recovered as Rh sponge36,37).
3.3. Impala Platinum Refining Process
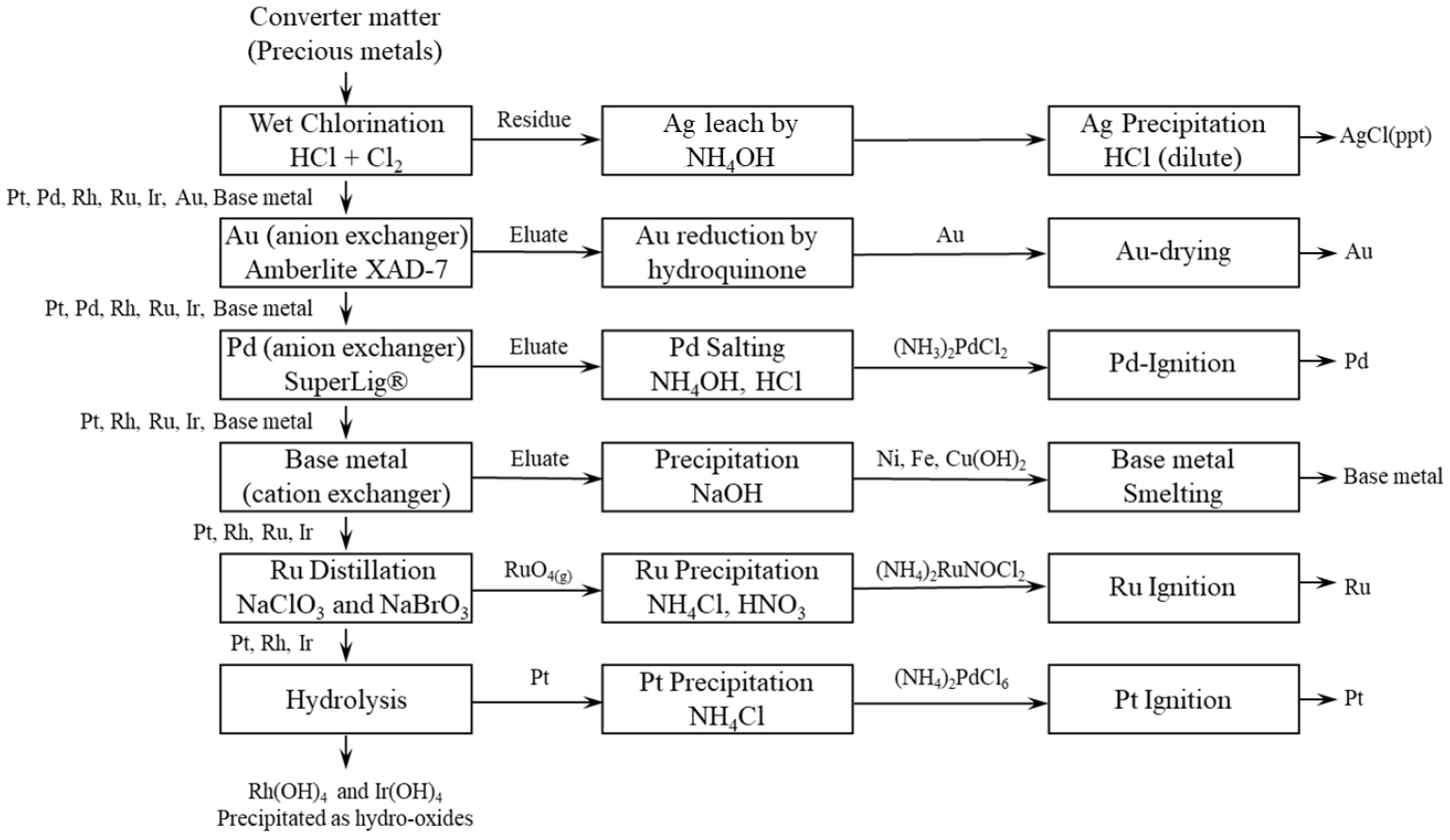
Impala Platinum process includes HCl leaching of converter matte and refining based on ion-exchange and precipitation as shown in Fig. 537,38). The converter matte containing approximately 65% PMs is dissolved by HCl and Cl2 and the metals are recovered sequentially.
After wet chlorination, the residue is leached with NH4OH and then HCl is added to precipitate silver ions (Ag+) as silver chloride (AgCl). The raffinate is passed through an anion exchange, Amberlite XAD-7, which selectively recover gold ions (Au3+). Au3+ is then eluted from XAD-7 and recovered as metallic Au (99.95% purity) through reduction precipitation.
After the Au is removed, Pd is selectively recovered using SuperLig®, a functionalized polystyrene-based anion exchanger, at a specific limit of redox potential and precipitated in salt form. The raffinate is then subjected to cation exchange to remove base metals, which are collectively precipitated.
Once the base metals are removed, the raffinate is boiled with the addition of NaClO3 and NaBrO3 to oxidize Ru to its tetroxide. RuO4 is than stripped to the gas phase and scrubbed with HCl in the distillation process, followed by precipitation as (NH4)2RuNOCl5. The precipitate is then ignited to obtain pure Ru metal.
After Ru distillation, the remaining metal-containing raffinate consists of Pt, Ir and Rh. Raffinate is adjusted to pH 6 to precipitate Ir and Rh as hydroxides. The resulting slurry is filtered, boiled to eliminate excess acid, re-dissolved in demineralized H2O and subjected to oxidation and neutralization with alkali for further purification. Pt is finally recovered by precipitation with NH4Cl as (NH4)2PtCl6, followed by ignited to form pure Pt metal37,38).
4. Hydrometallurgical Processes of Iridium
4.1. Leaching of Iridium
In hydrometallurgical processing, leaching of PGMs generally employs concentrated HCl with strong oxidizing agents such as nitric acid, hydrogen peroxide, or sodium chlorate. This is because PGM chloride complexes are stable under strongly acidic conditions39). While Pt and Pd readily dissolve in aqua regia to form soluble chloro-complexes, Rh, Ru and Ir are mostly insoluble even in aqua regia3). However, under sufficiently strong oxidative conditions, even Ir can be dissolved as shown in Table 3. Dissolution can be promoted by increasing the redox potential using oxidizing agent40). In addition, Ir can be converted into soluble salts by heating with NaCl and Cl2 gas at high temperature11).
Table 5 summarizes case studies on leaching of Ir12,41,42,49,50,51). Hodgson, A.P.J. et al. reported the dissolution of Ir without the use of chlorine gas by employing a pressure digestion vessel system. 10 mg Ir powder was placed in a 50 ml Teflon TFM PTFE liner containing 5 ml of HCl and 100 μL of H2O2 and heated at 210℃. Under these conditions, the leaching efficiency of Ir was 97.0 ± 14.5%41).
Table 5.
| Material | Pretreatment | Leaching agent | Leaching conditions |
Leaching efficiency | Ref |
| Ir powder | – | HCl, H2O2 |
Temp = 210℃, Time = 12 h | 97.0 ± 14.5% | Hodgson, A. P. J. et al. (2016) |
|
Medical technological product | Pyrolysis step at 550℃ | Agua regia |
Temp = 90℃ Time = 72 h | 95% | Kokkinos, E et al. (2024) |
| Ir powder |
Alkali fusion with Na2O2 at 600℃ | HCl |
Temp = 70℃ Time = 1 h | > 99% | Lee, J. et al. (2011) |
| Chip of Ir metal |
Alkali fusion with KOH and KNO3 at 900℃ | HNO3, NaNO2 | Time = 0.5 h | – | Silver, G. (1975) |
| Ir wire |
Chlorination by Dry aqua regia at 397℃ | KCl | – | 36.9% | Yoshimura, A et al. (2024) |
| Ir powder |
Heating with CaCO3 at 1300℃ | HCl |
Temp = 80℃ Time = 3 h | > 99% | Kobayashi, Y et al. (2019) |
Kokkinos, E. et al. investigated the leaching of noble metals from spent medical-technological products such as diagnostic electrophysiology catheter, diagnostic guide wire and self-expanding stent. The study employed pyrolysis step followed by acid leaching to recover Au, Pt, Ir and Ta. After removing plastic components through pyrolysis, Au was leached using 50% aqua regia (v/v). The residue, containing Ir, Pt and Ta, was further leached with concentrated aqua regia at 90℃ for 72 hours, with an additional 5 ml of aqua regia added every 6 hours to achieve complete dissolution of Pt and Ir42).
To improve Ir leaching efficiency, thermal pre-treatment methods have also been studied, including alloying with base metals and alkali fusion14,43,44,45,46,47,48,49,50,51). Alkali fusion is a well-known pre-treatment method used to improve the chemical reactivity of insoluble ores and metal compounds14,15). This method involves heating the material at high temperatures with alkaline reagents to convert the material into more soluble forms16). Common fluxing agents include Na2CO3, NaCl, Na2O2, NaOH, KOH and Ca(OH)215). Traditionally employed by analytical chemists, this approach has been increasingly applied to recover noble metals from secondary resources. In the case of Ir, alkali fusion converts the metal into soluble Na-Ir-O compounds such as Na2IrO3, which can be readily dissolved in acidic media14).
The fused Ir compound, typically in the +4 oxidation state, reacts with HCl to form the hexachloroiridic acid complex (H2IrCl6).
Lee, J. et al. reported the complete dissolution of Ir using alkali fusion. Ir (99.9% purity) was mixed with Na2O2 at a molar ratio of 1:2 and fused at 600℃ for 4 hours. This treatment formed Na-Ir-O compounds, inducing structural changes that facilitated dissolution. The resulting material was treated with 3 M HCl at 70℃ for 1 hour, leading to complete leaching of Ir12).
Silver, G. investigated a method involving the fusion of Ir with potassium hydroxide (KOH) and potassium nitrate (KNO3) at 900℃, followed by dissolution in HNO3 and NaNO249).
Yoshimura, A. et al. employed dry aqua regia – equimolar mixture of FeCl3 and KCl – to dissolve. The chlorination by dry aqua regia was conducted at temperature ranging from 357℃–397℃ for 2 to 8 hours. Ir was leached 36.9% in the case of 6 hours treatment at 397℃50).
Kobayashi, Y. et al. formed a complex oxide, Ca4IrO6 by heating Ir with calcium carbonate (CaCO3) at 1300℃ for 1 hours. The heated sample was then leached with HCl at 80℃ for 3 hours, resulting in complete dissolution of Ir.
4.2. Separation of Ir
The separation and purification of Ir is primarily conducted through hydrometallurgical methods. Common methods include precipitation, solvent extraction and ion exchange52,53). The selection of an appropriate separation method depends on the concentration and composition of PGMs in the solution. Among these, solvent extraction is widely employed in PGM separation due to its high selectivity and potential for continuous processing54). The method relies on the differential distribution of solutions between two immiscible phases – an organic phase and an aqueous phase – and generally consists of extraction, scrubbing and stripping steps. Solvent extraction improves separation efficiency and reduces the complexity of processing.
PGMs extraction is typically conducted in chloride media, where all PGMs can be dissolved. In such environments, PGMs primarily form anionic chloride complexes rather than cationic species11). Depending on the PGMs’ oxidation state and chloride concentration, various chloro-complexes can form, including some neutral species40). These variations in speciation significantly influence the distribution coefficients and extraction efficiency of each PGMs. Also PGMs have similar chemical properties, making their separation difficult55). In addition, they can form multiple oxidation states simultaneously, so understanding their speciation in solution is essential.
In chloride media, Ir exists predominantly as stable chloro-complexes such as [IrCl6]2- (Ir(IV)) and [IrCl6]3- (Ir(III)). The distribution of these species depends on the chloride ion concentration and the oxidation state. Ir(IV) is generally more extractable than Ir(III), which can be explained by stoichiometry of the ion pair38,56). For example, assuming mono positive organic cation sites, it is harder to pack 3[MCl6]3- than 2[MCl6]2- or [MCl4]- around the complex anion. This reverses the normal charge effect and leads to the following observed order of extractability for PGMs chloro species : MCl4- > MCl42- > MCl62- > MCl63-. The Ir(IV) species [IrCl6]2- carries a lower negative charge than [IrCl6]3-, making it more favorable for ion-pair formation with organic cations. However, Ir(IV) is easily reduced to Ir(III) in aqueous media. Therefore it is important to stabilize the +4 oxidation state by adding an oxidizing agent40,57).
Neutral and amine-based extractants are commonly used on Ir extraction, in chloride media23,58). Table 6 summarizes case studies on solvent extraction of Ir58,59,60,61,62,63,64,65,66,67). Amine-based extractants - tertiary amines (e.g., Alamine 336, Alamine 300, Alamine 308) and quaternary amine (e.g., Aliquat 336) - are usually used. The extraction mechanism of Ir(IV) with tertiary and quaternary amines follows an anion-exchange mechanism, which can be presented as follows59):
Table 6.
| Extractant | Diluent | Aqueous |
Extraction percentage |
Stripping (agent, efficiency) | Ref |
| TBPa | Toluene |
Ir 0.00025 M, Rh 0.0005 M [HCl] 6 M |
Ir > 83% Rh < 25% | – | Sun, P. et al. (2011) |
| Cyanex 921a | Kerosene |
Ir 110 mg/L, Rh 100 mg/L [HCl] 1 M |
Ir > 90% Rh < 20% | – | Le, M. N. et al. (2018) |
| Cyanex 921a | Dodecane |
Ir 50 mg/L [Cl-] 4.0 M, [H+] 1.0 M | Ir 70% |
H2O Ir 84% | Kedari, S. et al. (2005) |
|
Cyanex 921a + TBPa | Kerosene |
Ir 110 mg/L, Rh 100 mg/L [HCl] 5 M |
Ir 90% Rh < 10% |
(NH4)2S2O3 Ir 99% Rh 32% | Le, M. N. et al. (2018) |
| Cyanex 923a | Kerosene |
Ir 50 mg/L, Ru 50 mg/L [HCl] 1 M | Ir 87% Ru 48% |
H2O Ir > 90% | Kedari, C. et al. (2006) |
| Cyanex 923a | Dodecane |
Ir 50 mg/L [Cl-] 4.1 M, [H+] 0.1 M | Ir 94% |
H2O Ir 83% | Kedari, S. et al. (2005) |
|
Petroleum sulfoxidea | Xylene |
Ir 100 mg/L [HCl] 4 M | Ir 95.4% |
NaOH Ir 99.5% | Liu, W. et al. (2013) |
| Aliquat 336b | Kerosene |
Ir 50 mg/L [Cl-] 0.2 M, [H+] 0.2 M | Ir 95% |
NH3 + NH4Cl Ir 100% | Kedari, S. et al. (2005) |
|
Aliquat 336b + LIX 54c | Dodecane |
Ir 50 mg/L [Cl-] 0.1 M, [H+] 0.1 M | Ir 97% |
NH3 + NH4Cl Ir 100% | Kedari, S. et al. (2005) |
| Aliquat 336b | Kerosene |
Ir(IV) 200 mg/L, Rh(III) 120 mg/L [HCl] 1 M |
Ir > 95% Rh < 30% | – | Nguyen, T. H. et al. (2016) |
|
Aliquat 336b + TBPa | Kerosene |
Ir(IV) 200 mg/L, Rh(III) 120 mg/L [HCl] 1 M |
Ir > 90% Rh < 25% |
HClO4 Ir 95.0%, Rh 86.4% | Nguyen, T. H. et al. (2016) |
| Alamine 336b | Toluene |
Ir 0.00025 M, Rh 0.0005 M [HCl] 1 M |
Ir > 95% Rh < 70% | – | Sun, P. et al. (2011) |
| Alamine 336b | Kerosene |
Ir(IV) = Ru(III) = Rh(III) 100 mg/L [HCl] 1 M |
Ir > 95% Ru > 80% Rh < 40% |
Na2CO3 Ir 100% Ru 99% | Goralska, E. et al. (2007) |
| Alamine 336b | Kerosene |
Ir 50 mg/L [Cl-] 0.2 M, [H+] 0.2 M | Ir 94% |
NaOH + NaCl Ir 97% | Kedari, S. et al. (2005) |
|
Alamine 336b + LIX 54c | Dodecane |
Ir 50 mg/L [Cl-] 1.0 M, [H+] 1.0 M | Ir 93% |
NH3 + NH4Cl Ir 100% | Kedari, S. et al. (2005) |
| Alamine 300b | Dodecane |
Ir 50 mg/L [Cl-] 0.2 M, [H+] 0.2 M | Ir 93% |
NaOH + NaCl Ir 100% | Kedari, S. et al. (2005) |
| Alamine 308b | Kerosene |
Ir(IV) 200 mg/L, Rh(III) 120 mg/L [HCl] 1 M |
Ir > 90% Rh < 20% | – | Nguyen, T. H. et al. (2016) |
| TOAb | Kerosene |
Ir(IV) 200 mg/L, Rh(III) 120 mg/L [HCl] 1 M |
Ir > 95% Rh < 30% | – | Nguyen, T. H. et al. (2016) |
| TEHAb | Kerosene |
Ir(IV) 200 mg/L, Rh(III) 120 mg/L [HCl] 1 M |
Ir > 90% Rh < 20% | – | Nguyen, T. H. et al. (2016) |
Where denotes the chloro-complex anioninc species of Ir(IV).
Kedari, S. et al. reported that acidic extractants such as 15% LIX 54, LIX 860N-1, or Cyanex 272 resulted in extraction efficiencies of less than 2% for Ir(IV) in 0.1–4 M HCl solutions. This suggests that Ir(IV) forms a very strong complex with chloride anions59). However, when LIX 54 was mixed with Alamine 336 or Aliquat 336, the extraction efficiency of Ir(IV) increased59).
A major challenge in Ir solvent extraction is its separation from Rh. Ir(IV) and Rh(III) have similar chemical and physical properties, making their separation particularly difficult24,60). To address this problem, reductive separation methods have been investigated. One approach involves the use of SnCl2 to reduce Ir(IV) to Ir(III) and Rh(III) to Rh(I). Rh(I) forms stable complexes such as [Rh(SnCl3)5]4- in chloride media, which can be selectively extracted by extractants such as Alamine 336 and TBP61). The presence of SnCl2 improves separation selectivity by reducing Rh(III) to Rh(I), thereby increasing Rh extraction and decreasing Ir extraction61).
5. Conclusion
This study reviewed the properties, industrial applications and hydrometallurgical recovery processes of Ir. Ir is widely used in various industries due to its excellent corrosion and heat resistance. However, Ir has limited reserves and high price. Ir leaching processes require strongly acidic environment and high temperatures. However, strong oxidants such as Cl2 can cause equipment corrosion and safety risk. To address these issues, thermal pretreatment methods such as alkali fusion have been employed to increase Ir solubility, enabling leaching under relatively milder acidic conditions. Solvent extraction of Ir mainly follows an anion-exchange mechanism. Neutral and amine-based extractants are usually used for separation Ir from other PGMs. However, due to the chemical similarity between Ir and other PGMs, selective separation remains a major challenge. It is essential to improve extraction selectivity and develop more efficient separation techniques.